Prostate cancer, while commonly diagnosed as localized disease, remains the second leading cause of cancer mortality in the United States and Europe.1 For patients who die of prostate cancer, some will be initially diagnosed and treated for metastatic hormone-sensitive disease (mHSPC).
Others will progress from an initial diagnosis of localized disease through non-metastatic castration-resistant disease (nmCPRC) following initial local therapy with subsequent androgen deprivation therapy (ADT) for biochemical recurrence. In either case, nearly all men who die of prostate cancer will develop metastatic castration-resistant prostate cancer (mCRPC) prior to death.
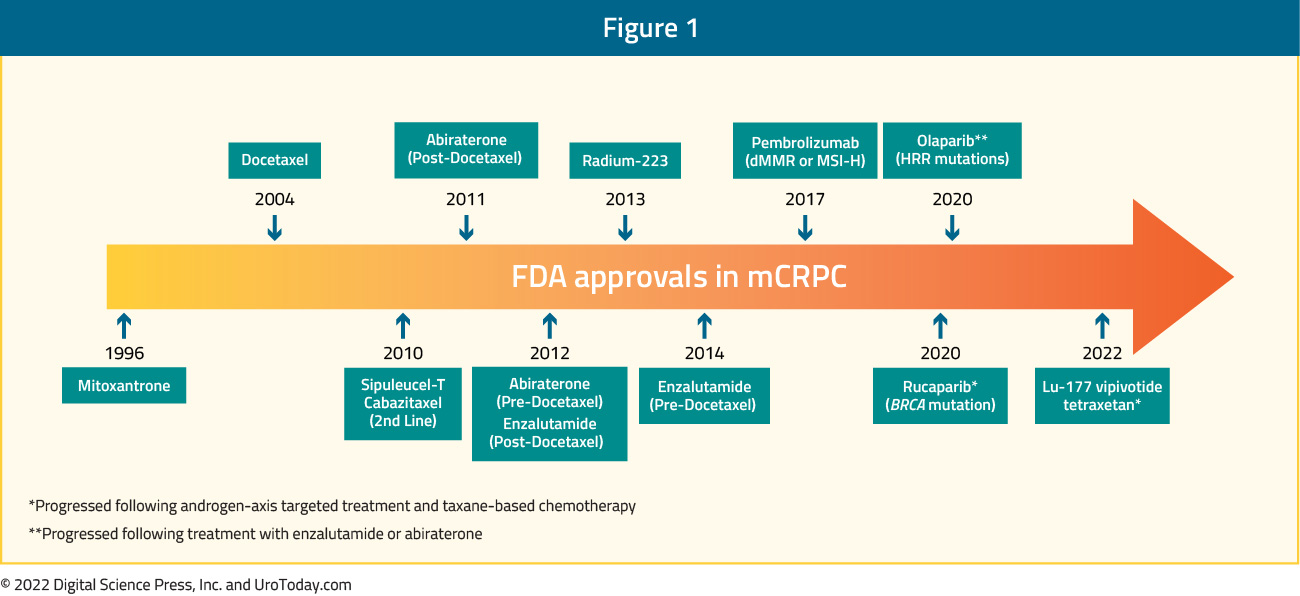
The treatment story for mCRPC starts in 1996, when mitoxantrone was approved by the U.S Food and Drug Administration (FDA) based on a palliative benefit with improvement in bone pain and effect on analgesic use. In the seminal trial leading to FDA approval, 161 mCRPC men with pain were randomized to receive mitoxantrone + prednisone or prednisone alone (10 mg daily). The primary endpoint for this trial was a palliative response defined as a two-point decrease in pain as assessed by a six-point scale completed by patients without an increase in analgesic medication and maintained for 2 consecutive evaluations at least 3 weeks apart. This trial found that a palliative response was observed in 29% of patients (95% CI 19% to 40%) who received mitoxantrone + prednisone and in 12% of patients (95% CI 6% to 22%) of men who received prednisone alone (p = 0.01).2 As such, chemotherapy with mitoxantrone and prednisone was deemed to provide palliation for men with symptomatic mCRPC.
Since the approval of mitoxantrone in 1996, over the last two decades, the treatment landscape of patients with mCRPC has rapidly evolved. Following the FDA approval of docetaxel in 2004, nine further agents have been approved in this disease space:
With the wealth of treatment options, clinicians and patients are in both the enviable but difficult position of making treatment choices and sequencing decisions in the mCRPC disease space. Additionally, following approval in mCRPC, many of these agents (including docetaxel, abiraterone, and enzalutamide) have now been approved in mHSPC and nmCRPC:
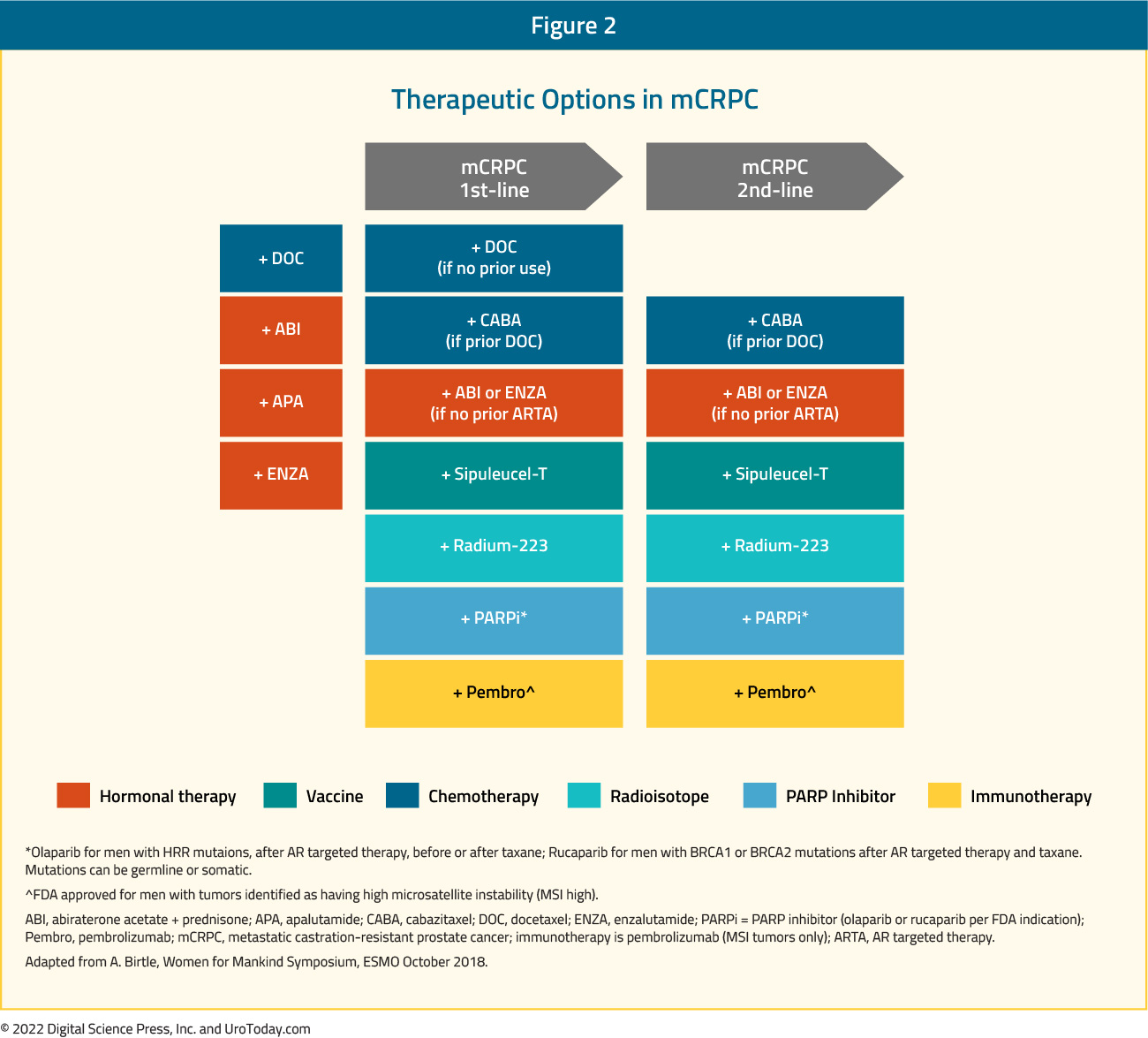
Thus, treatment decisions in the mCRPC disease space may be influenced by prior therapies. However, this series of articles will narratively review the evidence supporting the current FDA approved agents in mCRPC space and recent advances in mCRPC. Given the chronology of progress in advanced prostate cancer, many of these trials were performed while ADT alone was the standard of care in mHSPC.
TAXANES AND OTHER MICROTUBULE ASSEMBLY INHIBITORS
Docetaxel
FDA Approved in 2004 (TAX-327 Trial)
The first major advances leading to survival gains in systemic therapy for patients with advanced prostate cancer began with the introduction of docetaxel for men with castration resistant disease in 2004 (TAX-327). In the pivotal trial by Tannock et al, 1,006 men with mCRPC were randomized to receive: mitoxantrone (12 mg/m2 every 3 weeks), docetaxel (75 mg/m2 every 3 weeks), or docetaxel (30 mg/m2 weekly).1 Mitoxantrone use in patients with mCRPC had previously demonstrated improvements in pain and quality of life measures,2,3 thus forming the basis for its FDA approval in 1996 and inclusion as a control arm in this trial. TAX-327 met its overall survival (OS) primary endpoint, with men receiving docetaxel 75 mg/m2 every 3 weeks having a hazard ratio (HR) for death of 0.76 (95% CI 0.62 to 0.94; p = 0.009) compared to those receiving mitoxantrone. The OS benefit for the weekly docetaxel regimen was non-significantly superior compared to the mitoxantrone arm (HR 0.91, 95% CI 0.75 to 1.11; p = 0.36):

The median overall survival was improved from 16.5 months in the mitoxantrone group to 18.9 months in the docetaxel 75 mg/m2 every 3 weeks arm. Docetaxel 75 mg/m2 use was further associated with improvements in PSA responses, pain outcomes and quality of life metrics.1 These results formed the basis of the FDA’s approval of docetaxel usage in mCRPC patients in 2004.
Cabazitaxel
FDA Approved in 2010 (TROPIC Trial)
The next agent to be approved in the mCRPC space was cabazitaxel. At that time, there was no agent yet approved in docetaxel pre-treated mCRPC patients. De Bono et al. compared the efficacy and safety of cabazitaxel plus prednisone with those of mitoxantrone plus prednisone in men with progressive mCRPC after docetaxel-based treatment. In this phase III trial, 755 men were treated with 10 mg oral prednisone daily and were randomized to either mitoxantrone (12 mg/m2 IV) or cabazitaxel (25 mg/m2 IV) every 3 weeks. Patients in the cabazitaxel arm had a superior median OS (15.1 versus 12.7 months; HR 0.70, 95% CI 0.59 to 0.83, p<0.001)
and median progression-free survival (PFS) (2.8 versus 14 months; HR 0.74; 95% CI 0.64 to 0.86, p<0.001):
Figure 4: OVERALL SURVIVAL/PROGRESSION-FREE SURVIVAL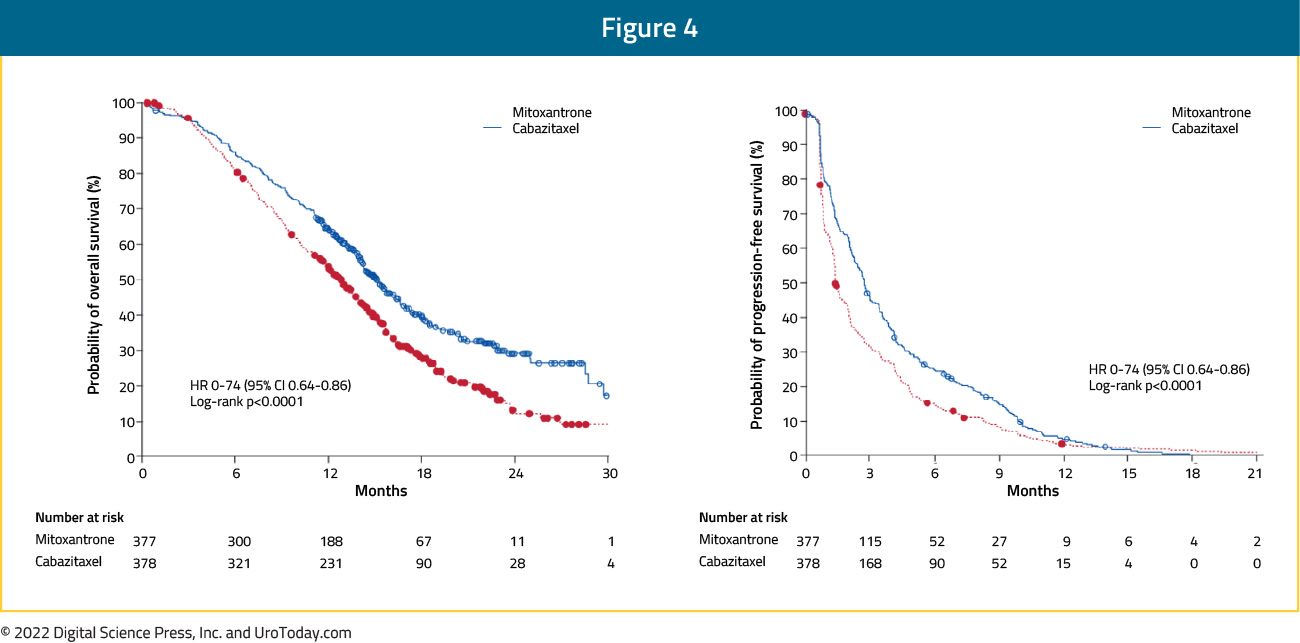
The most common clinically significant grade 3 or higher adverse events (AEs) were neutropenia (cabazitaxel: 82% versus mitoxantrone: 58%) and diarrhea (6% versus <1%).4
CARD Trial (2019)
Between publication of the TAX-327 and TROPIC trials, a number of trials demonstrated benefit of androgen receptor-targeted agents (ARTAs) including abiraterone acetate and enzalutamide in patients in both the pre-chemotherapy and post-chemotherapy setting. As a result, these agents became standard of care. However, there remained an unmet need to assess treatment approaches for patients who have previously received docetaxel and an androgen receptor-targeted agent (ARTA). Published in 2019, the CARD trial randomized 255 patients who had previously received both docetaxel and an ARTA (either abiraterone or enzalutamide) to either cabazitaxel or the other androgen axis targeting agent. The primary outcome of interest was imaging-based PFS with OS, response, and toxicity considered secondarily. Over a median follow-up of 9.2 months, patients who received cabazitaxel had a significantly lower risk of imaging-based progression or death (95 of 129, 74%; median PFS 8 months) compared to those who underwent androgen axis inhibitor switch (101 of 126, 80%; median PFS 3.7 months) (HR 0.54, 95% CI 0.40 to 0.73):
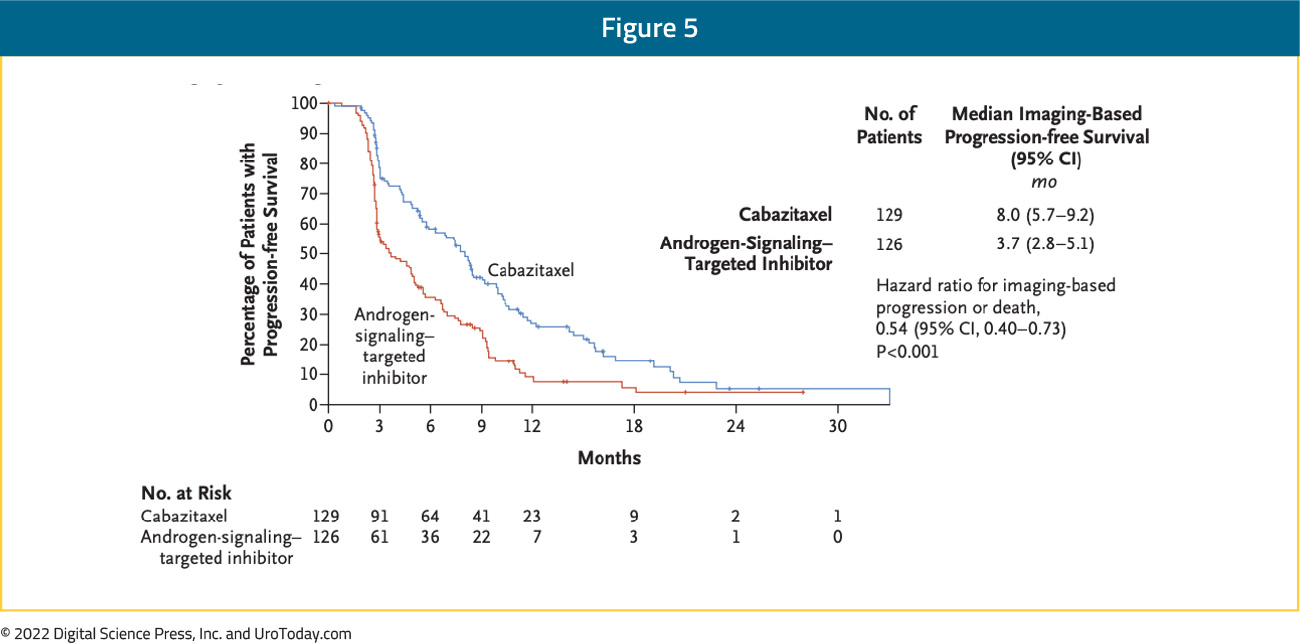
OS was similarly improved with the use of cabazitaxel (13.6 months vs 11.0 months; HR: 0.64, 95% CI: 0.46 – 0.89):
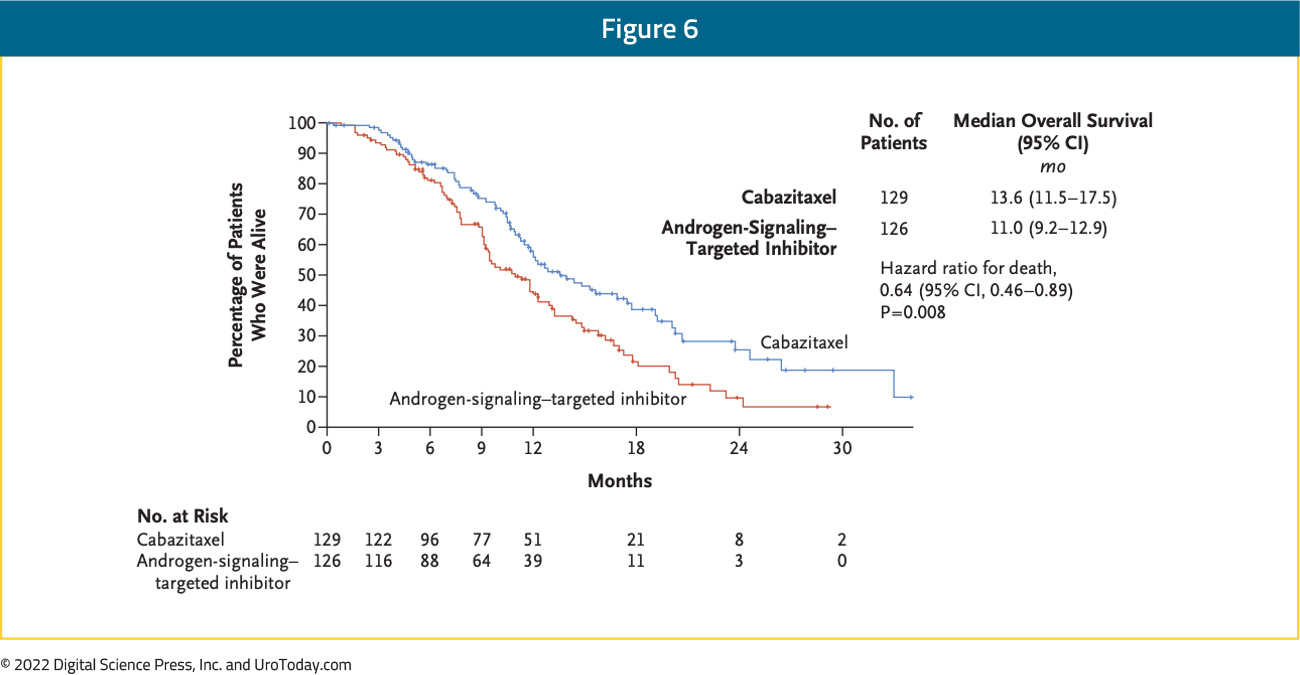
AEs were in keeping with previously identified safety profiles with AEs ≥grade 3 occuring in 56% of patients receiving cabazitaxel and 52% of those receiving ARTAs.5
A comparative cost analysis by Morgans et al. of cabazitaxel versus a second androgen axis inhibitor in mCRPC patients previously treated with docetaxel and an alternative androgen axis inhibitor (similar to CARD setting) demonstrated that the use of cabazitaxel in a US health care setting would be associated with a 17% decrease in overall costs, accounting for over $300,000 USD saved per 100 treated patients.6
OSTRICh Trial
The OSTRICh trial (NCT03295565) is an open-label, multicenter phase IIb study that compared cabazitaxel to an ARTA (abiraterone or enzalutamide) in poor-prognosis mCRPC patients (defined as visceral metastases or < 12 months responsive to androgen deprivation or progression during or within 6 months after docetaxel completion), previously presented at ASCO 2021. Overall, 34% of patients had received docetaxel in the mHSPC setting, while 39% had received an ARTA. The primary endpoint was clinical benefit rate defined as: no radiotherapy, no ECOG PS increase ≥2, no change of therapy, and no radiological progression at 12 weeks in the study arms. A total of 106 patients were randomized, with 53 patients allocated to each arm. There was no significant difference in 12-week clinical benefit rate (60% in cabazitaxel arm versus 51% in AR-targeted agent arm). 12-week rPFS was superior in the cabazitaxel arm (88% versus 67%, p=0.046). However, median rPFS was non-significantly different at 6.0 months in the cabazitaxel arm and 5.8 months in the AR-targeted agent arm (p = 0.5), while median OS was also not statistically significantly different: 15.3 months and 13.8 months in the cabazitaxel and ARTA treated patients, respectively (p = 0.8). Grade ≥3 AEs were more common in patients treated with cabazitaxel (29%) versus those treated with ARTAs (15%).
Sabizabulin
Sabizabulin (VERU-111) is a first-in-class, oral agent that inhibits microtubule polymerization disrupting the cytoskeleton and arresting cellular proliferation with a favorable toxicity profile. While sabizabulin has gained prominence in the COVID-19 and breast cancer disease spaces, to date it has only been formally evaluated in a Phase 1b/2 study in the mCRPC setting. In prostate cancer, targeted inhibition of microtubule polymerization further blocks the transport of the androgen receptor into the nucleus
Presented at the 2022 GU ASCO meeting, this Phase 1b/2 clinical study (NCT03752099) evaluated the minimum tolerated dose and preliminarily efficacy in men with mCRPC who progressed on at least one ARTA. Treatment was relatively well tolerated with the most common AEs being mild to moderate diarrhea, fatigue, nausea, and vomiting with no clinically relevant neurotoxicity or neutropenia. In terms of efficacy, in this Phase 1b/2 study, the median rPFS was 11.4 months (range 6-36+ months) for patients receiving a 63mg dose formulation (n = 55). Treatment has subsequently been reformulated with improved bioavailability. Thus, the Phase 3 dose was subsequently set at 32mg PO daily.
At this dosage, sabizabulin is being assessed in the treatment of patients with mCRPC who have progressed on at least one ARTA in the ongoing VERACITY trial, a phase 3 multicenter, randomized, active-control study (NCT04844749). To be eligible for inclusion, patients must have received at least one ARTA for mCRPC but not have received chemotherapy. Enrolled patients are being randomized in a 2:1 fashion to receive sabizabulin (32 mg/day orally) or active control (alternative ARTA) and will remain on study until rPFS. Randomization will be stratified by a number of characteristics including RECIST measurable disease vs bone-only disease and by prior exposure to ARTA (progressed on one vs more than one prior ARTA):
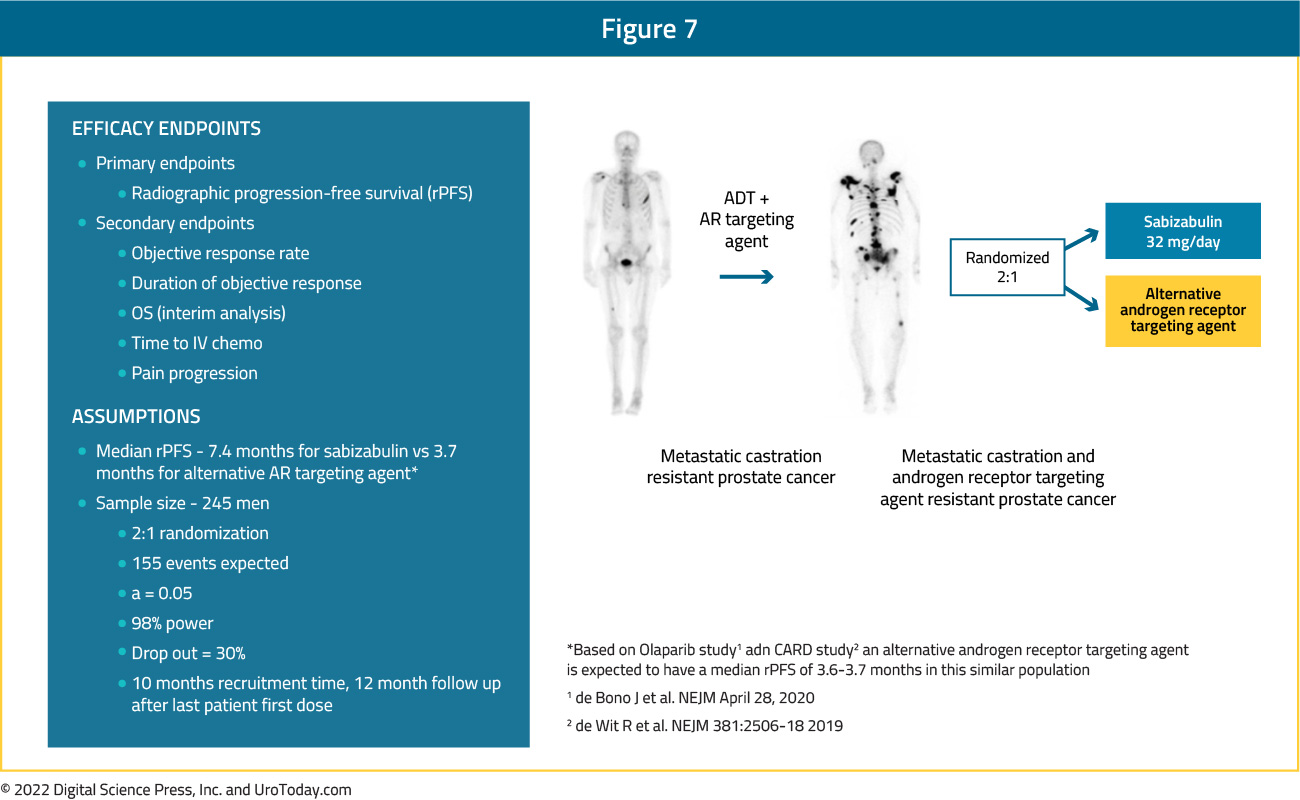
The primary efficacy endpoint of the study is median rPFS with a number of secondary endpoints including ORR, duration of objective response, OS, and time to IV chemotherapy. The target enrollment is 245 patients, and this trial is currently ongoing in approximately 45 clinical sites with enrollment anticipated to be completed in 2022.
Conclusions
Since 2004, chemotherapy represents the backbone of therapy for advanced prostate cancer, including patients with mCRPC. Undoubtedly, chemotherapy has improved quality of life and survival in these men. Over the course of the next mCRPC Center of Excellence articles, we will delve into the other treatment options for mCRPC beyond (and in combination with) chemotherapy.
Written by:Zachary Klaassen, MD MSc, Medical College of Georgia, Augusta, Georgia, USA
Rashid Sayyid, MD MSc, University of Toronto, Toronto, ON


