When examined at a national level in the United States, cancers of the kidney and renal pelvis (despite different histology) are considered in aggregate and represent the sixth most common newly diagnosed tumors in men and eighth most common in women in the United States in 2020,1 representing an estimated 73,750 new diagnoses and 14,830 deaths. The vast majority of these cancers will be renal parenchymal tumors with renal cell carcinoma (RCC) comprising the preponderance of these lesions. As previous UroToday Center of Excellence articles have highlighted, clear cell renal cell carcinoma (ccRCC) is the most common histologic subtype of renal cell carcinoma (RCC). Likely due to its much higher prevalence, the vast majority of systemic therapies for RCC have been investigated among patients with ccRCC. However, there have been important recent advances in treatment for patients with non-clear cell renal cell carcinomas (nccRCC) as well in recent years.
Despite ongoing stage migration as a result of widespread use of axial abdominal imaging for non-specific abdominal complaints,2 a large proportion (up to 35%) of patients present with advanced disease, including metastases.3 Historically, metastatic RCC has been early uniformly fatal, with 10-year survival rates less than 5%.4 However, there has been a transformational change in this disease space over the past fifteen years and, with newer immunotherapy-based approaches, the potential for long-term cure is something that may be considered. Certainly, a significantly longer natural history is feasible given available therapeutic options.
Historical Context
Most clinicians involved in the care of patients with advanced kidney cancer will know that the immunologically active characteristics of this tumor have long been recognized. As a result, modulators of the immune system were among the first therapeutic targets for advanced ccRCC and interferon-alfa and interleukin-2 were the available treatment options prior to 2005. Based on early data suggesting a response rate between 10 to 15%5 and comparative data demonstrating a survival benefit compared to other available systemic therapies available at the time,6 interferon-alfa retained utilization despite significant toxicity. Further, it was among patients with metastatic RCC receiving interferon-alfa that the Motzer prognostic criteria were derived.6 However, even among patients treated at a center of excellence, median overall survival was only 30 months in favorable-risk patients, 14 months in intermediate-risk patients, and 5 months in poor-risk patients.6 Interleukin-2 had similar response rates to interferon-based therapies (~15 to 20%),7 but distinctly had evidence of durable complete responses in approximately 7 to 9% of patients.8 This observation led to the U.S. Food and Drug Administration (FDA) approval of high-dose IL-2 in 1992. However, IL-2 is associated with significant toxicity which has limited its widespread use.
Following the so-called “cytokine era”, treatment of advanced kidney cancer was revolutionized by the introduction of targeted therapies, beginning with sorafenib in 2005, sunitinib in 2006, and temsirolimus in 2007, along with a number of other agents in the years that followed.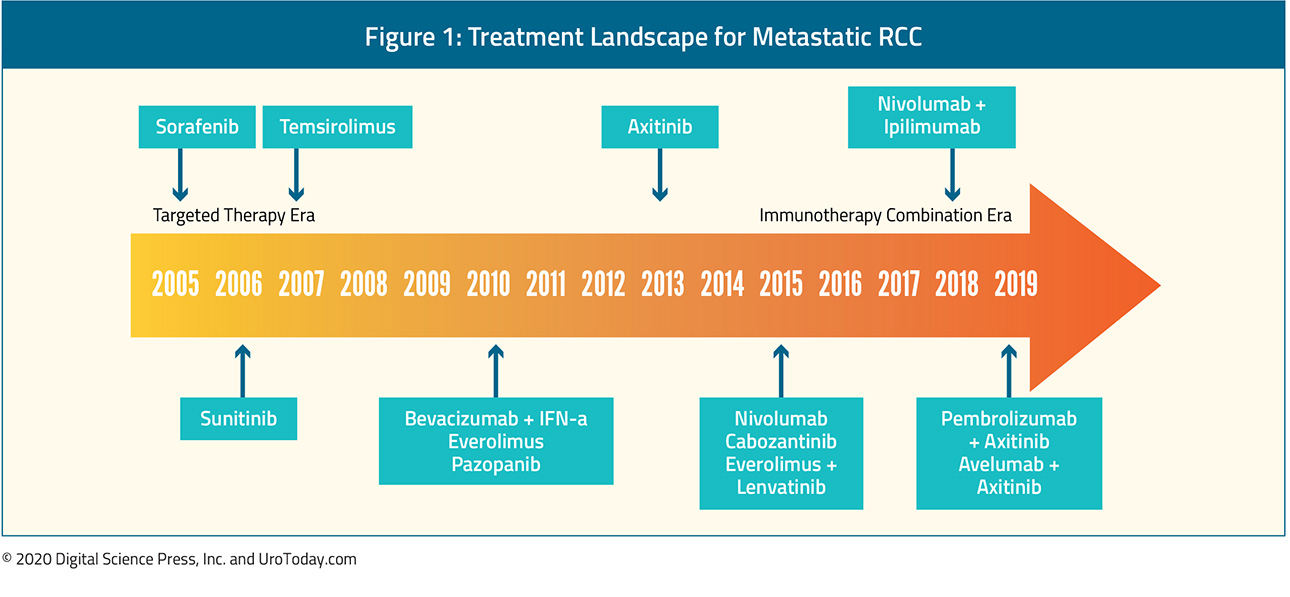
These so-called “targeted therapies” were developed based on work into the molecular biology underlying ccRCC through targeting of the vascular endothelial growth factor (VEGF) pathway and mammalian target of rapamycin (mTOR). This pathway plays a key role in regulating HIF-α, thus modulating the pathway between abnormalities in VHF and proliferation.
Bevacizumab, a humanized monoclonal antibody against VEGF-A, was the first inhibitor of the VEGF pathway used in clinical trials. In head-to-head trials against interferon-alfa, the addition of bevacizumab to interferon resulted in significant improvements in response rate and progression-free survival.9,10 Today, bevacizumab is uncommonly used as monotherapy in untreated patients but is considered as second-line therapy in patients who have failed prior therapy with tyrosine kinase inhibitors.
In contrast, tyrosine-kinase inhibitors (TKIs) quickly became standard of care as first-line monotherapy and, for many years, sunitinib was the standard of care, such that it has formed the control comparison for testing of newer approaches. TKIs also target the VEGF pathway, through inhibition of a combination of VEGFR-2, PDGFR-β, raf-1 c-Kit, and Fit-3 (sunitinib and sorafenib). In 2006, sorafenib was shown to have biologic activity in ccRCC and was one of the first molecularly targeted agents clinically available. However, despite FDA approval, sorafenib was quickly supplanted by sunitinib as a first-line VEGF inhibitor. Sunitinib was first tested among patients who had previously received cytokine therapy and then, in a pivotal Phase III trial, demonstrated superiority (both in terms of progression-free survival and quality of life) in a head-to-head comparison with interferon-α.11 Since the approval of sunitinib and sorafenib, there has been the development and subsequent approval of a number of other tyrosine kinase inhibitors. For the most part, the goal of these agents has been to reduce the toxicity of VEGF inhibitors while retaining oncologic efficacy. Comparative data of pazopanib and sunitinib have demonstrated non-inferior oncologic outcomes with decreased toxicity among patients receiving pazopanib.12 Axitinib was evaluated first as second-line therapy13 and then in the first-line setting compared to sorafenib.14 Finally, tivozanib has been compared to sorafenib among patients who had not previously received VEGF or mTOR-targeting therapies. While this study demonstrated tivozanib’s activity, it was not FDA approved and is therefore not used.
Most recently, a multikinase inhibitor (acting on tyrosine kinases including MET, VEGF receptors), and TAM family of kinases (TYRO3, MER, and AXL), cabozantinib has been approved for the first-line treatment of mRCC. In the Phase II CABOSUN trial, "Cabozantinib-s-malate or Sunitinib Malate in Treating Patients With Previously Untreated Locally Advanced or Metastatic Kidney Cancer (CABOSUN)", cabozantinib was compared to sunitinib in the first-line treatment of patients with intermediate or poor-risk mRCC and demonstrated significantly improved progression-free survival (hazard ratio 0.66, 95% confidence interval [CI] 0.46 to 0.95).15 An updated analysis of these data utilizing independent progression-free survival (PFS) review demonstrated comparable results (hazard ratio 0.48, 95% confidence interval 0.31 to 0.74).16 However, this trial has yet to demonstrate an overall survival benefit to cabozantinib compared to sunitinib (hazard ratio 0.80, 95% confidence interval 0.53 to 1.21).
In parallel to the development, clinical appraisal, and utilization of VEGF inhibitors has come to the development of mTOR inhibitors. For the most part, these agents have not been used in first-line therapy. However, temsirolimus, interferon, and the combination were compared among 626 patients with pre-defined poor-risk metastatic RCC who had not previously received systemic therapy.17 Patients who received temsirolimus had significantly improved overall survival compared to those receiving interferon-alfa (hazard ratio 0.73, 95% confidence interval 0.58 to 0.92). Notably, the combination arm did not offer a benefit compared to interferon alone. Unlike temsirolimus which must be administered intravenously, everolimus is an oral agent.
More recently, the potent, selective, small molecular HIF-2α inhibitor MK-6482 was evaluated among patients with von Hippel-Lindau disease on the basis of germline alterations and measurable solid RCC tumors who had not previously received systemic anti-cancer therapy. In an analysis of 61 patients presented at the 2020 European Society of Medical Oncology (ESMO) Virtual Congress, the objective response rate was 36% (95% confidence interval 24 to 49%) with an additional 11% unconfirmed responses. However, none of these were complete responses. Progression-free survival was 98% (95% confidence interval 89 to 100%) at 52 weeks. MK-6482 also demonstrated benefits in non-RCC tumors including pancreatic lesions and central nervous systemic hemangioblastomas. This novel therapy was relatively well tolerated with 13% experiencing grade 3 treatment-related adverse events and none experiencing grade 4 or 5 treatment-related adverse events.
The Return of Immunotherapy for Advanced RCC
The immunologic basis for the treatment of advanced RCC has been well established, including the aforementioned cytokine therapies. Thus, it should not be surprising that the use of checkpoint inhibitors has demonstrated benefit in patients with metastatic RCC.
First presented at ESMO in the fall of 2017 and subsequently published in the spring of 2018, CheckMate 214, "Nivolumab Combined With Ipilimumab Versus Sunitinib in Previously Untreated Advanced or Metastatic Renal Cell Carcinoma (CheckMate 214)", demonstrated an overall survival (OS) benefit for first-line nivolumab + ipilimumab vs sunitinib.18 This trial randomized 1096 patients to the combination immunotherapy approach of nivolumab + ipilimumab (550 patients) or sunitinib (546 patients). The majority of patients had intermediate or poor-risk disease (n=847). Overall survival was significantly improved in the overall patient population; however, stratified analyses provide more nuanced results. Among the subgroup of patients with intermediate or poor-risk RCC, treatment with nivolumab + ipilimumab resulted in significantly improved overall response rate, comparable progression-free survival, and significantly improved overall survival. In contrast, among patients with favorable-risk disease, progression-free survival and overall response rate were higher among patients who received sunitinib. Subsequently, Escudier and colleagues have assessed the efficacy of nivolumab + ipilimumab according to the number of IMDC risk factors.19 In keeping with the previously reported differences in the comparative benefit of nivolumab + ipilimumab versus sunitinib on the basis of risk category (intermediate/poor vs favorable), the authors demonstrated stable objective response rate (ORR) across increasing numbers of International Metastatic RCC Database Consortium (IMDC) risk factors (from zero to 6) for those who received nivolumab and ipilimumab, while the ORR in patients treated with sunitinib decreased with an increasing number of IMDC risk factors.
At ESMO 2020, long-term follow-up from CheckMate 214 was reported. In a poster presentation, Dr. Albiges presented results among patients with at least four years of follow-up data. In this group, in the intention-to-treat population, results were very similar to the initial analysis previously published with the combined nivolumab + ipilimumab approach continuing to demonstrate superiority (hazard ratio 0.69, 95% confidence interval 0.59 to 0.81). In sub-groups defined according to IMDC criteria, those with intermediate or poor-risk had improved survival with nivolumab/ipilimumab (hazard ratio 0.65, 95% confidence interval 0.54 to 0.78) while there continued to be no appreciable difference between treatment approaches among those with favorable-risk disease (hazard ratio 0.93, 95% confidence interval 0.62 to 1.40). These authors then presented a posthoc analysis of patients without a history of nephrectomy, demonstrating that while primary tumor responses were more common among patients receiving nivolumab + ipilimumab, no complete responses were seen in either group.
Also at ESMO 2020, Dr. Regan and colleagues relied upon long term data from CheckMate214 to assess a novel outcome metric, treatment-free survival with and without toxicity. The rationale for this approach is that conventional measures may not fully capture the effects of immuno-oncology (IO) approaches. Previous work has shown that patients may have long periods of disease control without subsequent anticancer therapy following discontinuation of IO regimes, and my experience toxicity during this time. The authors defined treatment-free survival (TFS), as the time between protocol therapy cessation and subsequent systemic therapy or death. They stratified this as TFS with or without toxicity by counting the number of days with ≥1 grade ≥3 treatment-related adverse events reported. As of 42-months of follow-up, 56% of patients randomized to nivolumab + ipilimumab and 47% of those randomized to sunitinib were alive with 13% and 7%, respectively, remaining on their original therapy. A further 31% of patients randomized to nivolumab + ipilimumab and 12% of those randomized to sunitinib were surviving free of subsequent, second-line therapy. Fourty-two-month restricted TFS was higher for patients randomized to nivolumab + ipilimumab (7.8 months) than those randomized to sunitinib (3.3 months). Toxicity-free TFS was 7.1 months and 3.0 months, respectively. In each case, the 95% confidence interval of the difference in median TFS excluded unity demonstrating that these are significant differences. Unlike the differences in PFS and OS which appear to be restricted to patients with intermediate and poor-risk disease, Dr. Regan and colleagues showed that the benefits in TFS were dramatic in patients with both IMBC intermediate and poor-risk disease (median TFS 6.9 vs 3.1 months) and favorable-risk disease (median TFS 11.0 vs 3.7 months).
The BIONIKK trial, an open-label, Phase II biomarker-driven randomized trial, "A BIOmarker Driven Trial With Nivolumab and Ipilimumab or VEGFR tKi in Naïve Metastatic Kidney Cancer (BIONIKK)", was also presented as ESMO 2020. Previous analyses have demonstrated that immune and angiogenic signatures can allow for the differentiation of four groups of patients (ccrcc1-4) with immune and angiogenic high/low features which could allow to better identify responders to either nivolumab, nivolumab + ipilimumab, or TKI. ccrcc1 “immune-low” and ccrcc4 “immune-high” tumors have been associated with the poorest outcomes, whereas ccrcc2 “angio-high” and ccrcc3 “normal-like” tumors have been associated with the best outcomes. In this biomarker-driven trial, patients with ccrcc1 and ccrcc4 signatures were randomized to nivolumab versus nivolumab + ipilimumab, whereas those with ccrcc2 and ccrcc3 signatures were randomized to receive nivolumab + ipilimumab versus TKI.
As a Phase II trial, the primary endpoint for this study was objective response rate (ORR, RECIST1.1) per treatment and group. Secondary endpoints included PFS, OS, and tolerability. Two-hundred and two patients were randomized of a targeted 187.
Among patients with the ccrcc1 signature, objective response rates were higher among those who received combination therapy with nivolumab + ipilimumab (39.4%; 6.1% complete response rate) than those who received nivolumab alone (20.7%; 0% complete response rate) whereas among those with a ccrcc4 signature, objective response rates were 50.3% in those receiving the combination approach (11.8% complete response rate) as compared to 50% in those receiving nivolumab alone (7.1%). Median progression-free survival among patients with the ccrcc1 signature was 8.0 months in those receiving nivolumab + ipilimumab and 4.6 months among those receiving nivolumab alone. In the ccrcc4 group, median progression-free survival was 12.2 months in the combination arm and 7.8 months in the nivolumab monotherapy arm.
In patients with the ccrcc2 signature, objective response rates were 48.3% in the nivolumab + ipilimumab arm (13.8% complete response rate) and 53.8% in the TKI arm (0% complete response rate), whereas among patients with the ccrcc3 signature, 25% receiving nivolumab + ipilimumab had objective responses (0% complete response rate) and 0% receiving TKI had an objective response.
These are the first randomized data based on molecular risk group assessment to guide first-line therapy in metastatic ccRCC. In particular, among patients with the ccrcc4 signature, the use of combination therapy may not be required and thus ipilimumab may be spared.
Combination Approaches: Targeted Therapy and Immunotherapy
Combination therapy has been well established in the treatment of advanced RCC, including the use of interferon-alfa and bevacizumab.9,10 Following the data from CheckMate 214 demonstrating the role of immune checkpoint blockade in advanced RCC, data began to emerge on the combination of targeted therapies with checkpoint inhibitors.
The first of these studies was IMmotion151, "A Study of Atezolizumab in Combination With Bevacizumab Versus Sunitinib in Participants With Untreated Advanced Renal Cell Carcinoma (RCC) (IMmotion151)", first presented at the 2018 American Society of Clinical Oncology Genitourinary (ASCO GU) Cancers Symposium and subsequently published, which compared first-line atezolizumab + bevacizumab vs sunitinib among 915 patients with previously untreated metastatic RCC.20 The combined approach demonstrated a significant benefit in progression-free survival (11.2 months versus 7.7 months; hazard ratio 0.74, 95% confidence interval 0.57 to 0.96) among the whole cohort of patients and had lower rates of significant (grade 3-4) adverse events (40% vs 54%).
Subsequently, two further combination approaches have been approved on the basis of published Phase III trials: pembrolizumab + axitinib (KEYNOTE-426), "Study to Evaluate the Efficacy and Safety of Pembrolizumab (MK-3475) in Combination With Axitinib Versus Sunitinib Monotherapy in Participants With Renal Cell Carcinoma (MK-3475-426/KEYNOTE-426)", and avelumab + axitinib (JAVELIN Renal 101), "A Study of Avelumab With Axitinib Versus Sunitinib In Advanced Renal Cell Cancer (JAVELIN Renal 101)". In KEYNOTE-426, 861 patients with metastatic clear cell RCC, predominately with intermediate or poor-risk disease, who had not previously received systemic therapy were randomized to pembrolizumab + axitinib or sunitinib and followed for the co-primary endpoints of overall survival and progression-free survival.21 While median survival was not reached, patients who received pembrolizumab + axitinib had improved overall survival (hazard ratio 0.53, 95% confidence interval 0.38 to 0.74) and progression-free survival (hazard ratio 0.69, 95% confidence interval 0.57 to 0.84), as well as overall response rate. These results were consistent across subgroups of demographic characteristics, IMDC risk categories, and PD-L1 expression level. Grade 3 to 5 adverse events were somewhat more common among patients getting pembrolizumab and axitinib, though rates of discontinuation were lower. Similarly, JAVELIN Renal 101 randomized 886 patients to avelumab + axitinib or sunitinib.22 Again, the preponderance of patients had IMDC intermediate or poor-risk disease. In this analysis, the primary endpoints were PFS and OS in patients with programmed death ligand 1 (PD-L1) positive tumors. Notably, 560 of the 886 patients had PD-L1 positive tumors. Among the PD-L1 positive subgroup, progression-free survival (hazard ratio 0.61, 95% confidence interval 0.47 to 0.79) was improved in patients receiving avelumab + axitinib compared to sunitinib while overall survival did not significantly differ (hazard ratio 0.82, 95% confidence interval 0.53 to 1.28). In the overall study population, progression-free survival was similarly improved, as compared to the PD-L1 positive population (hazard ratio 0.69, 95% confidence interval 0.56 to 0.84).
New Combination Therapies: Data from ESMO 2020
While not yet published, three impactful analyses in this disease space were presented at ESMO 2020. These rely on combinations with the multi-kinase inhibitor cabozantinib, which acts on tyrosine kinases including MET, VEGF receptors, and TAM family of kinases (TYRO3, MER, and AXL). Cabozantinib, a standard-of-care for the treatment of advanced RCC, is potentially particularly well suited to combination therapy with immune checkpoint inhibitors as it promotes an immune-permissive environment which may enhance response to immune checkpoint inhibitors. In combination with immune checkpoint inhibitors, cabozantinib has shown promising activity for other tumor types including urothelial carcinoma, castration-resistant prostate cancer, lung cancer, and hepatocellular carcinoma.
First, as presented in the Presidential I session, Dr. Choueiri provided the first results of the CheckMate-9ER trial (NCT03141177), "A Study of Nivolumab Combined With Cabozantinib Compared to Sunitinib in Previously Untreated Advanced or Metastatic Renal Cell Carcinoma (CheckMate 9ER)", a Phase III randomized controlled trial comparing nivolumab + cabozantinib to sunitinib, in the first-line treatment of patients with advanced or metastatic renal cell carcinoma. Included patients had previously untreated, histologically confirmed advanced or metastatic renal cell carcinoma with a clear cell component and were randomized in a 1:1 fashion to receive nivolumab 240mg IV q2 weeks + cabozantinib 40mg PO daily or sunitinib 50mg PO daily for 4 weeks in repeating 6-week cycles, with randomization was stratified by IMDC risk score, tumor PD-L1 expression, and region. The primary outcome was progression-free survival with overall survival, objective response rate, and toxicity comprising important secondary outcomes.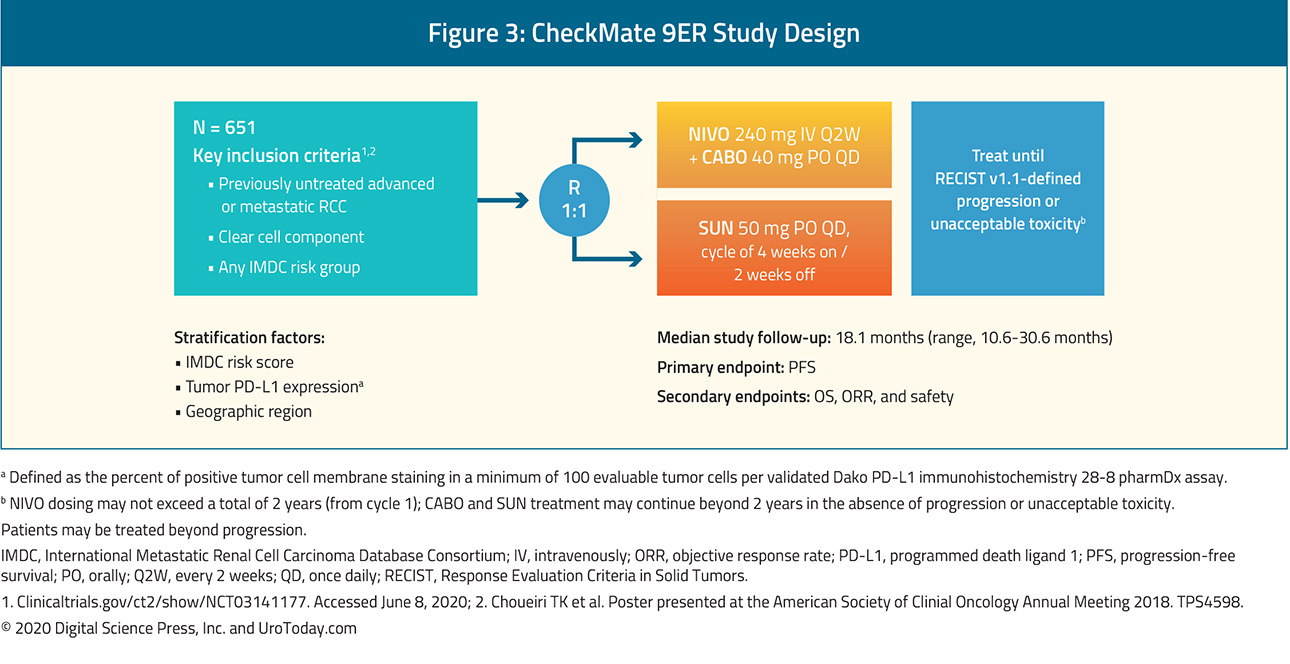
Among 651 randomized patients (22.6% favorable-risk disease, 57.6% intermediate-risk disease, and 19.7% poor-risk disease), 323 received nivolumab + cabozantinib and 328 received sunitinib. Over a median follow-up of 18 months, median progression-free survival was significantly longer among those randomized to nivolumab + cabozantinib (16.6 months) than those randomized to sunitinib (8.3 months), with a relative difference of 49% (hazard ratio 0.51, 95% confidence interval 0.41 to 0.64) as was overall survival (medians not reached; hazard ratio 0.60, 98.89% confidence interval 0.40 to 0.89). Notably, these benefits were seen consistently across pre-specified subgroups defined according to IMDC risk categories and PD-L1 expression. Any grade treatment-related adverse events were common in both groups: 96.6% among those receiving nivolumab + cabozantinib and 93.1% among those receiving sunitinib. High-grade events (grade 3 or greater) were somewhat higher among those receiving nivolumab + cabozantinib (60.6% vs 50.9%). One grade 5 event occurred in the nivolumab + cabozantinib arm while two occurred in the sunitinib treated group. Notably, quality of life was maintained for those receiving nivolumab + cabozantinib while there was a decline in quality of life among those receiving sunitinib. On the basis of these data, a supplementary new drug application has been filed for this combination as first-line treatment in patients with advanced RCC.
The second important trial in this disease space to be presented at ESMO 2020 is COSMIC-021, "Study of Cabozantinib in Combination With Atezolizumab to Subjects With Locally Advanced or Metastatic Solid Tumors (COSMIC-021)", a multicenter phase 1b study, evaluating the combination of cabozantinib + atezolizumab in various solid tumors (NCT03170960), including first-line treatment of clear cell renal cell carcinoma (RCC). In the subset of included patients with ccRCC, 10 patients were enrolled in the dose-escalation and 60 in the expansion stage of the study. Eligible patients had to have an ECOG performance status of 0-1 and not have received prior systemic anticancer therapy for advanced RCC. Patients were enrolled sequentially to receive atezolizumab 1200 mg IV every three weeks with either cabozantinib 40 mg (dose level 40 [DL40], n=34) or cabozantinib 60 mg (DL60, n=36) PO daily in each stage. The primary endpoint for this trial is the ORR per RECIST v1.1 by the investigator, the secondary endpoint was safety, and exploratory endpoints include PFS and correlation of biomarkers with outcomes.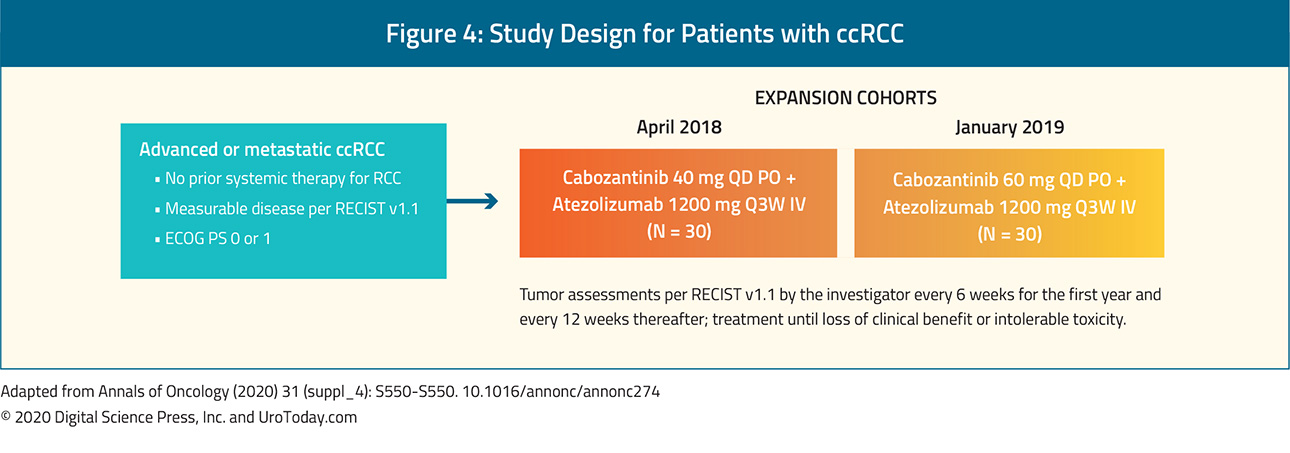
In the COSMIC-021 ccRCC subset, For DL40, the ORR was 53% (80% CI 41-65), with one complete response (3%) and 17 partial responses (50%), the disease control rate was 94%, duration of response was not reached (range: 12.4 months to not reached), and the median time to objective response was 1.4 months (range: 1-19). For DL60, the ORR was 58% (80% CI 46-70), with four complete responses (11%) and 17 partial responses (47%), the disease control rate was 92%, the median duration of response was 15.4 months (range: 8.1 to not reached), and the median time to objective response was 1.5 months (range: 1-7). For DL40, the median PFS was 19.5 months (95% CI 11.0 to not reached) compared to 15.1 months (95% CI 8.2-22.3) for DL60. This approach is currently being further investigated in the CONTACT-03 trial, "A Study of Atezolizumab in Combination With Cabozantinib Compared to Cabozantinib Alone in Participants With Advanced Renal Cell Carcinoma After Immune Checkpoint Inhibitor Treatment (CONTACT-03)", (NCT04338269), a phase III RCT comparing atezolizumab + cabozantinib to cabozantinib alone in patients who had previously received immune checkpoint therapy.
Non-Clear Cell Histology
In general, randomized trials in advanced RCC have focused on patients with clear cell histology. As a result, there have been little direct data to guide care and we have had to rely on extrapolation from data derived among patients with clear cell histology. However, at this year’s ESMO meeting, Dr. McGregor reported on the use of cabozantinib + atezolizumab in a subcohort of patients with non-clear cell histology the COSMIC-031 trial, "Study of Cabozantinib in Combination With Nivolumab and Ipilimumab in Patients With Previously Untreated Advanced or Metastatic Renal Cell Carcinoma (COSMIC-313)". Notably, in this cohort, patients were allowed up to one previously line of TKI (but not previous checkpoint inhibitor therapy or cabozantinib). At the time of data cut-off, 30 patients had been enrolled and followed for a median of 13.0 months. The cohort included 15 patients with papillary, seven patients with chromophobe, and eight patients with other histology. Five patients had received previous systemic therapy while 25 (83%) were treatment-naïve. Confirmed objective response rate per RECIST v1.1 was 33% (80% confidence interval 22 to 47%), and there were 10 patients with partial responses (papillary, n=6; chromophobe, n=1; ccRCC, n=1; translocation, n=1; and unclassified, n=1) but there were no complete responses, although partial responses occurred in all IMDC risk groups. The median progression-free survival was 9.5 months (95% CI 5.5 to not reached). Notably, patients with nccRCC will be included in the previously mentioned CONTACT-03 trial.
Treatment Selection
As highlighted above, there are a number of treatment approaches which have, in Phase III randomized controlled trials, demonstrated superiority to sunitinib in first-line treatment of mRCC including atezolizumab + bevacizumab, nivolumab + ipilimumab, pembrolizumab + axitinib, avelumab + axitinib, and nivolumab + cabozantinib. As highlighted in the BIONNIKK trial, a tumor-derived signature may allow for rational treatment selection, however, prior to this, IMDC risk categories and PD-L1 testing may provide some guidance. Additionally, authors have considered cost-effectiveness analyses to help guide treatment selection.23,24 However, as may be expected, varying the assumptions of these models may change the preferred treatment options.
Written by: Christopher J.D. Wallis, MD, PhD, Instructor in Urology, Vanderbilt University Medical Center, Nashville, Tennessee; Zachary Klaassen, MD, MSc, Assistant Professor of Urology, Georgia Cancer Center, Augusta University/Medical College of Georgia, Atlanta, Georgia
References:
2. Welch, H. Gilbert, Jonathan S. Skinner, Florian R. Schroeck, Weiping Zhou, and William C. Black. "Regional variation of computed tomographic imaging in the United States and the risk of nephrectomy." JAMA internal medicine 178, no. 2 (2018): 221-227.
3. Motzer, Robert J., Madhu Mazumdar, Jennifer Bacik, William Berg, Alison Amsterdam, and Joseph Ferrara. "Survival and prognostic stratification of 670 patients with advanced renal cell carcinoma." Journal of clinical oncology 17, no. 8 (1999): 2530-2530.
4. Negrier, S., B. Escudier, F. Gomez, J-Y. Douillard, A. Ravaud, C. Chevreau, M. Buclon, D. Perol, and C. Lasset. "Prognostic factors of survival and rapid progression in 782 patients with metastatic renal carcinomas treated by cytokines: a report from the Groupe Francais d’Immunotherapie." Annals of Oncology 13, no. 9 (2002): 1460-1468.
5. Motzer, Robert J., Jennifer Bacik, Barbara A. Murphy, Paul Russo, and Madhu Mazumdar. "Interferon-alfa as a comparative treatment for clinical trials of new therapies against advanced renal cell carcinoma." Journal of clinical oncology 20, no. 1 (2002): 289-296.
6. Coppin, Chris, Franz Porzsolt, Michael Autenrieth, Julia Kumpf, Andrew Coldman, and Timothy Wilt. "Immunotherapy for advanced renal cell cancer." Cochrane database of systematic reviews 3 (2004).
7. Dutcher, Janice P., Michael Atkins, Richard Fisher, Geoffrey Weiss, Kim Margolin, Fred Aronson, Jeffrey Sosman et al. "Interleukin-2-based therapy for metastatic renal cell cancer: the Cytokine Working Group experience, 1989-1997." The Cancer Journal From Scientific American 3 (1997): S73-8.
8. Rosenberg, Steven A., James C. Yang, Donald E. White, and Seth M. Steinberg. "Durability of complete responses in patients with metastatic cancer treated with high-dose interleukin-2: identification of the antigens mediating response." Annals of surgery 228, no. 3 (1998): 307.
9. Rini, Brian I., Susan Halabi, Jonathan E. Rosenberg, Walter M. Stadler, Daniel A. Vaena, San-San Ou, Laura Archer et al. "Bevacizumab plus interferon alfa compared with interferon alfa monotherapy in patients with metastatic renal cell carcinoma: CALGB 90206." Journal of Clinical Oncology 26, no. 33 (2008): 5422.
10. Escudier, Bernard, Anna Pluzanska, Piotr Koralewski, Alain Ravaud, Sergio Bracarda, Cezary Szczylik, Christine Chevreau et al. "Bevacizumab plus interferon alfa-2a for treatment of metastatic renal cell carcinoma: a randomised, double-blind phase III trial." The Lancet 370, no. 9605 (2007): 2103-2111.
11. Motzer, Robert J., Thomas E. Hutson, Piotr Tomczak, M. Dror Michaelson, Ronald M. Bukowski, Olivier Rixe, Stéphane Oudard et al. "Sunitinib versus interferon alfa in metastatic renal-cell carcinoma." New England Journal of Medicine 356, no. 2 (2007): 115-124.
17. Hudes, Gary, Michael Carducci, Piotr Tomczak, Janice Dutcher, Robert Figlin, Anil Kapoor, Elzbieta Staroslawska et al. "Temsirolimus, interferon alfa, or both for advanced renal-cell carcinoma." New England Journal of Medicine 356, no. 22 (2007): 2271-2281.
18. Escudier, B., N. M. Tannir, D. F. McDermott, O. A. Frontera, B. Melichar, E. R. Plimack, P. Barthelemy et al. "LBA5CheckMate 214: Efficacy and safety of nivolumab+ ipilimumab (N+ I) v sunitinib (S) for treatment-naïve advanced or metastatic renal cell carcinoma (mRCC), including IMDC risk and PD-L1 expression subgroups." Annals of Oncology 28, no. suppl_5 (2017).
23. Su, Yingjie, Jie Fu, Jiangyang Du, and Bin Wu. "First-line treatments for advanced renal-cell carcinoma with immune checkpoint inhibitors: systematic review, network meta-analysis and cost-effectiveness analysis." Therapeutic advances in medical oncology 12 (2020): 1758835920950199.
24. Bensimon, Arielle G., Yichen Zhong, Umang Swami, Allison Briggs, Joshua Young, Yuan Feng, Yan Song et al. "Cost-effectiveness of pembrolizumab with axitinib as first-line treatment for advanced renal cell carcinoma." Current medical research and opinion 36, no. 9 (2020): 1507-1517.
Download Issue


