(UroToday.com) Non-clear cell renal cell carcinoma (nccRCC) encompasses a heterogeneous group of histologies comprising ∼25% of all RCC diagnoses with worse outcomes than clear cell RCC. Cabozantinib inhibits receptor tyrosine kinases that regular tumor cell growth, angiogenesis, and immune cell function, including MET, VEGFR, and the TAM family of kinases (TYRO3, AXL, and MER). Cabozantinib is approved in the United States for the treatment of patients with advanced RCC, and in the European Union for patients with previously untreated advanced RCC of intermediate and poor-risk patients with advanced RCC previously treated with VEGF-targeted therapy. In retrospective studies, single-agent cabozantinib has demonstrated clinically meaningful benefits across all subtypes of nccRCC.1
Both cabozantinib monotherapy and immune checkpoint inhibitors have shown preliminary activity in nccRCC, and several phase 2 trials are evaluating cabozantinib in nccRCC. Cabozantinib promotes an immune-permissive environment that may enhance response to immune checkpoint inhibitors and has shown encouraging activity in combination with immune checkpoint inhibitors in tumor types including ccRCC, urothelial carcinoma, metastatic castration-resistant prostate cancer (mCRPC), and hepatocellular carcinoma. COSMIC-021, a multicenter phase 1b study, is evaluating the combination of cabozantinib with atezolizumab in various solid tumors (NCT03170960). At the virtual European Society of Medical Oncology – (ESMO) 2020 annual meeting, Dr. Bradley McGregor and colleagues presented initial results from cohort 10 of COSMIC-021 in patients with nccRCC.
Eligible patients for cohort 10 of COSMIC-021 had an ECOG performance status of 0-1 and had received ≤1 prior VEGFR-TKI therapy for advanced nccRCC. Prior immune checkpoint inhibitor or cabozantinib therapy was not allowed. Patients received cabozantinib 40 mg PO QD and atezolizumab 1200 mg IV Q3W:
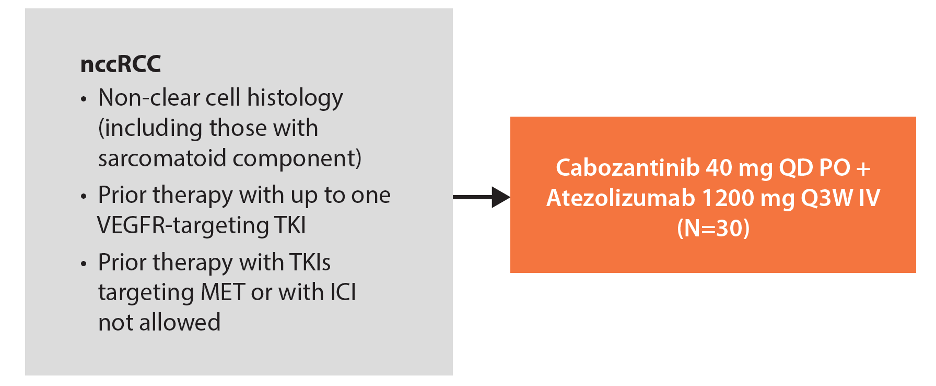
The primary endpoint is objective response rate (ORR) per RECIST v1.1 by the investigator. Other endpoints include safety, duration of response (DOR), PFS, and OS. Tumor assessments by CT or MRI were performed at screening and every six weeks for the first 12 months and every 12 weeks thereafter.
At the data cutoff of July 21, 2020, 30 patients were enrolled with a median follow-up of 13.0 months (range 8-20), including 16 patients (53%) that remained on treatment. Histological subtypes were papillary, n=15; chromophobe, n=7; and other, n=8. The median age was 61.0 years (range: 37-78, 87% were male, 70% had ECOG performance status 0, 80% had prior nephrectomy, and 57% had ≥3 sites of disease. Five patients (17%) had received prior VEGFR-TKI therapy (two in combination with everolimus). There were 37% of patients that had favorable-risk disease, 50% were intermediate, and 13% were poor risk by IMDC criteria.
Confirmed ORR per RECIST v1.1 was 33% (80% CI 22-47), and there were 10 patients with partial responses (papillary, n=6; chromophobe, n=1; ccRCC, n=1; translocation, n=1; and unclassified, n=1); there were no complete responses, although partial responses occurred in all IMDC risk groups. The DCR (CR+PR+SD) rate was 93%. The change in the sum of target lesions over time was as follows:
Median duration of response was 7.9 months (range 1.0+, 8.3+), and summarized as follows:

The median progression-free survival was 9.5 months (95% CI 5.5 to not reached). Increased median levels of activated peripheral cytotoxic T (+13%) and NK (+34%) cells were observed at day 21 with a concomitant decrease in immunosuppressive cells. Grade 3/4 treatment-related adverse events occurred in 30%, with no grade 5 treatment-related adverse events. Hypophosphatemia was the most common grade 3/4 treatment-related adverse event (n=4, 13%).
Dr. McGregor provided the following concluding statements for his presentation of the initial results of cohort 10 of the COSMIC-021 study:
- Cabozantinib plus atezolizumab demonstrated encouraging clinical activity in patients with nccRCC with an acceptable safety profile
- Responses were observed across several subtypes of nccRCC
- Antitumor immunomodulatory effects were observed in peripheral blood with cabozantinib plus atezolizumab
- The CONTACT-03 trial of cabozantinib plus atezolizumab immune checkpoint inhibitor-pretreated RCC patients, including those with papillary or unclassified nccRCC, is currently ongoing
References:
1. Chanza NM, Xie W, Bilen MA, et al. Cabozantinib in advanced non-clear-cell renal cell carcinoma: A multicentre, retrospective, cohort study. Lancet Oncol 2019 Apr;20(4):581-590.
Presented by: Bradley McGregor, MD, Clinical Director, Senior Physician, Instructor in Medicine, Lank Center for Genitourinary Oncology, Harvard Medical School, Dana-Farber Cancer Institute
Written by: Zachary Klaassen, MD, MSc – Assistant Professor of Urology, Georgia Cancer Center, Augusta University/Medical College of Georgia, Twitter: @zklaassen_md at the European Society for Medical Oncology Virtual Congress, ESMO Virtual Congress 2020 #ESMO20, 18 Sept - 21 Sept 2020
Related Content:
ASCO GU 2019: Phase Ib (COSMIC-021) Trial of Cabozantinib in Urothelial Carcinoma or Cabozantinib in Combination with Atezolizumab in patients with Urothelial Carcinoma, Castrate Resistant Prostate Cancer or Renal Cell Carcinoma
ASCO 2020: Cabozantinib in Combination with Atezolizumab in Patients with Metastatic Castration-Resistant Prostate Cancer: Results of Cohort 6 of the COSMIC-021 Study
ASCO 2020: Cabozantinib in Combination with Atezolizumab in Urothelial Carcinoma Previously Treated with Platinum-Containing Chemotherapy: Results from Cohort 2 of the COSMIC-021 Study

