Phase 2 Study of the Oral HIF-2α Inhibitor MK-6482 for Von Hippel-Lindau (VHL) Disease–Associated Clear Cell Renal Cell Carcinoma (ccRCC): Update on RCC and Non-RCC Disease
Von Hippel-Lindau disease, a condition associated with inherited alterations in the VHL gene, gives rise to benign and malignant tumors including clear cell renal cell carcinoma (RCC). Given its relative rarity, recruitment to clinical trials is difficult, and there is significant heterogeneity in terms of response to different therapies due in part to heterogeneity in the expression of various associated endothelial receptors at different organ sites. Management of VHL disease-associated kidney cancer is additionally challenging because of the need to balance the risk of disseminated disease with risk of renal morbidity from treatment. Currently, localized renal masses in patients with VHL disease are managed under the “3 cm rule”, which aims to limit the risk of metastatic disease but also avoid for as long as safely possible the multiple renal resections and risk of renal insufficiency that result from multiple tumors arising in the kidney.
Inactivation of the VHL gene is thought to exert its oncogenic influence in part through activation of the HIF-2a transcription factor. MK-6482 is an oral inhibitor of HIF-2a. Dr. Suarez summarized available data from 61 patients treated with MK-6482, with an overall response rate of 36.1%, 91.8% rate of tumors experiencing size reduction, and no patients to date having progressive disease. This response rate is comparable to previously reported outcomes with other targeted agents in RCC such as sunitinib and pazopanib. MK-6482 is also active in other VHL disease-associated tumors such as pancreatic lesion (ORR 63.9%, no progressive disease to date), and CNS hemangioblastomas (ORR 30.2%, 1 patient with progressive disease to date). Importantly, these response rates are the best reported to date for VHL disease-associated pancreatic lesions and CNS hemangioblastomas. While promising, further study is required to confirm the benefit of this agent in randomized clinical trials, understand its efficacy in advanced disease, explore the appropriate duration of therapy, and identify is single-agent therapy is optimal or if patients are better served with combination therapies involving MK-6482.
An integrated study of spatial dynamics and genomic alterations in renal cell carcinoma evolution
Renal cell carcinoma is a unique therapeutic challenge in part because of the lack of predictive and prognostic biomarkers, a fact-driven likely by substantial intratumoral genomic heterogeneity. In a prior publication (Turajlic, et al, Cell 2018), seven different evolutionary subtypes were identified in localized ccRCC tumors that correlated with clinical phenotypes (shown below).1
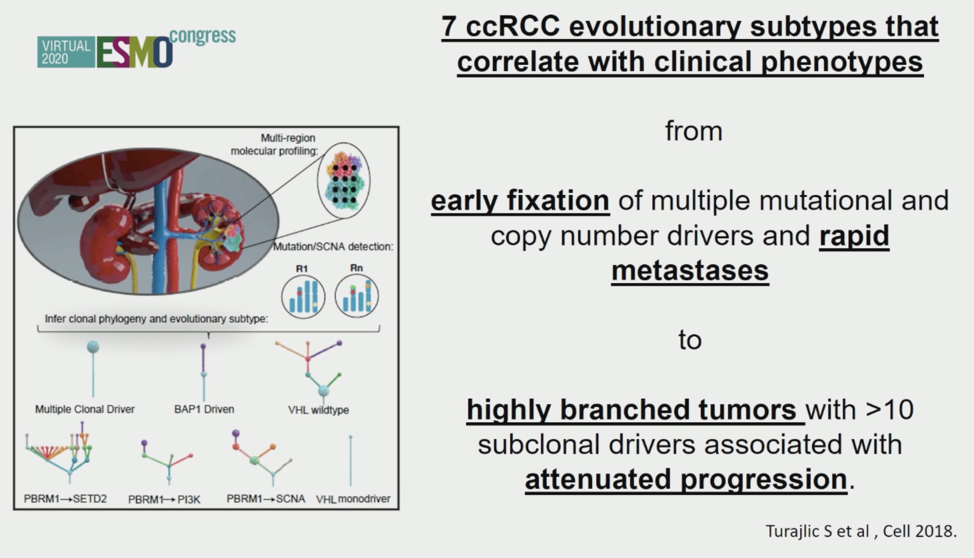
Building on this previous work, presentation 703MO leveraged spatial genomics in 79 clear cell RCC samples to find that more aggressive features such as higher proliferation, necrosis, higher Fuhrmann grade, higher copy number alteration burden, and subclones that subsequently metastasized were present in the centers of the tumors. Based on an MRI study from 2018 and pre-clinical models, it is known that the center of ccRCC tumors is more hypoxic, which could be the stressor leading to the more aggressive features discovered in these regions.
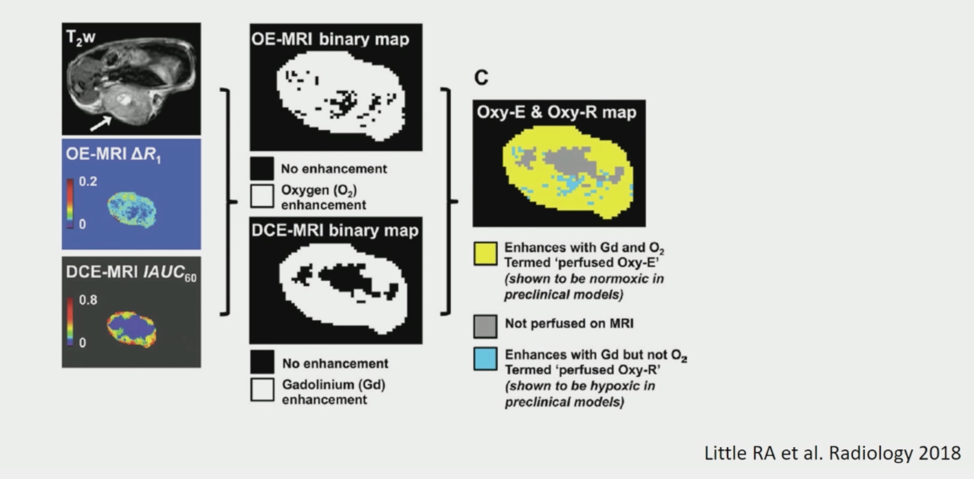
This study highlights the importance of considering subclonal and spatial architecture within tumors, especially those in the center, in understanding tumor metastatic potential.
Presented by: Cristina Suarez, MD, Ph.D., Medical Oncologist at the Hospital Universitari Vall D’Hebron and Vall D’Hebron Institute of Oncology
Written by: Alok Tewari, MD, Ph.D., Medical Oncologist at the Dana-Farber Cancer Institute, at the 2020 European Society for Medical Oncology Virtual Congress (#ESMO20), September 19th-September 21st, 2020.
References:
1. Turajlic, et al. Deterministic Evolutionary Trajectories Influence Primary Tumor Growth: TRACERx Renal. Cell. 2018. doi: 10.1016/j.cell.2018.03.043.
Related Content:
ESMO Virtual Congress 2020: An Integrated Study of Spatial Dynamics and Genomic Alterations in Renal Cell Carcinoma Evolution

