This was a 59-year-old male with no history of smoking or alcohol and a medical history that was significant for hypertension only, with a Charlson comorbidity score of 1 and a BMI of 23. The patient had no prior surgical history and no known family history of prostate cancer.
The patient underwent a digital rectal examination demonstrating a T3 disease, with a total PSA of 8 ng/ml, and a prostatic health index of 62. The patient also underwent a 1.5 Tesla multiparametric MRI in another institution, which demonstrated a PIRADS 5 lesion with evidence of left seminal vesicle involvement at T2-weighted sequences with concomitant restricted diffusion at the ADC map. Furthermore, there was evidence of enlarged lymph nodes in the T1 sequence, at the right obturator (12 mm) and right external iliac (14 mm).
Dr. Schaeffer began his presentation discussing the biopsy strategy of such a patient. A prostate biopsy would establish the diagnosis in this case, while a lymph node biopsy would establish the pathological stage. There are various prostate biopsy techniques, including a transrectal systemic, saturated or image-guided biopsy, and a transperineal limited, saturated or image-guided biopsy. Cancer usually develops in the peripheral zones of the prostate, which constitutes a relatively small portion of the prostate gland. This is opposed to the central zone of the prostate, which is the location of benign prostatic hypertrophy (Figure 1).
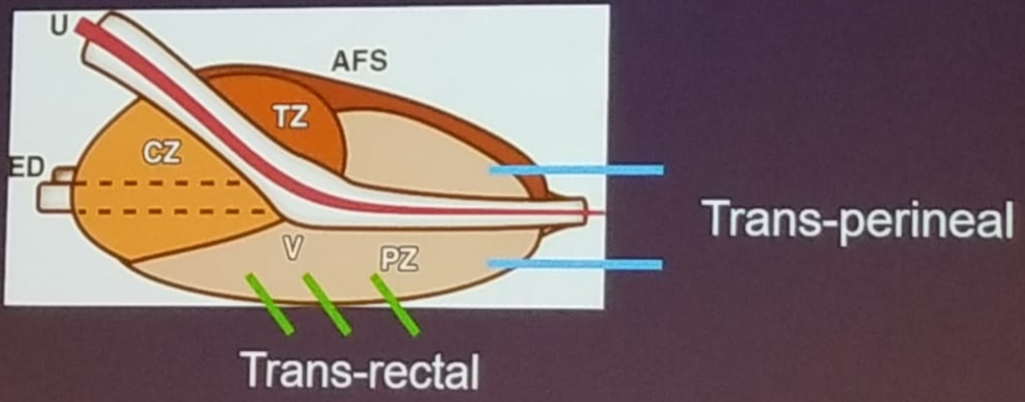
The disadvantage of the transrectal biopsy is the risk of missing tumors in the anterior zone of the prostate. This disadvantage does not exist with the transperineal technique (Figure 2). The transperineal saturation approach entails the most comprehensive sampling with no need for prebiopsy antibiotics. However, it is costly and time-consuming. Image-guided transrectal or transperineal biopsies offer improved detection of clinically significant cancer, and an MRI before a biopsy can reduce the number of biopsies performed.
Figure 1 – Prostate anatomy:
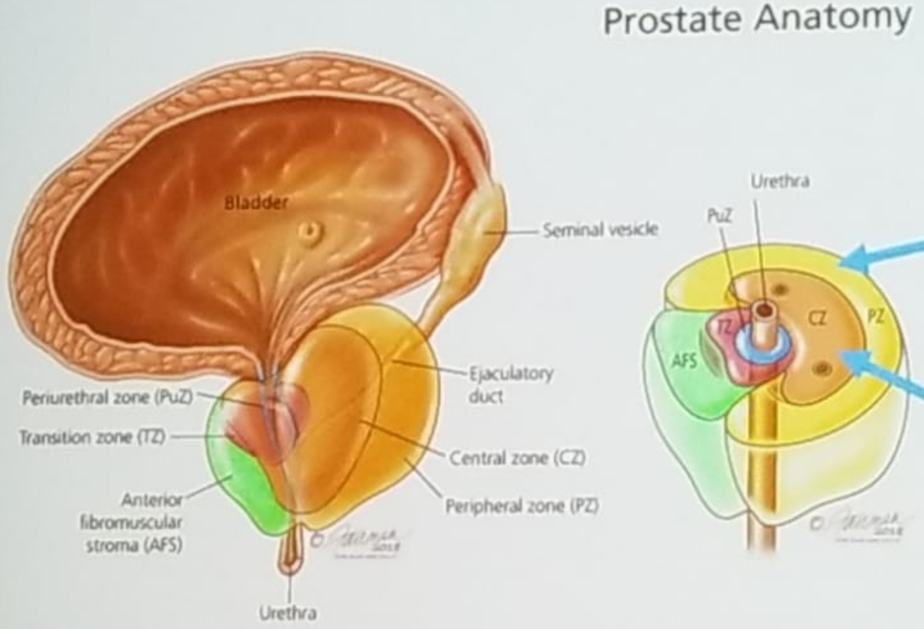
Figure 2 – Transrectal vs. Transperineal prostate biopsy:
The PRECISION trial1 compared MRI targeted biopsy only to systematic 10-12 core standard transrectal ultrasound-guided biopsy. This trial demonstrated an increased detection of significant cancer (10-15%), and reduced detection of insignificant cancer with the MRI targeted biopsy only. However, Dr. Schaeffer believes that 12 systematic cores should also be obtained at the time of targeted prostate biopsy. This is due to the fact that 5-10% of clinically significant cancers are found outside of the MRI target. Furthermore, systematic biopsy adds to the understanding of the complete geography of the tumor.
Dr. Montorsi continued at this point to present what had occurred with the patient later on. The patient went on to undergo a transrectal targeted and systematic biopsy which revealed 10/15 cores positive for Gleason 7 (4+3) disease. His bone scan was negative, and his CT scan revealed two suspicious lymph nodes at the right obturator, measuring 12 mm, and a right external iliac node measuring 14 mm. Various treatment options were presented at this time, including radical prostatectomy, external beam radiotherapy + androgen deprivation therapy (ADT), or systemic therapies.
Dr. Schaeffer continued to discuss the option of radical prostatectomy for this specific patient. The goals of surgery include optimal local control with extirpation of the entire tumor. The surgical option also offers optimal consolidation with extended lymph node dissection, with a possible cure. The type of pelvic lymph node dissection that should be performed was discussed next.
According to the EAU and NCCN guidelines, an extended pelvic lymph node dissection is indicated in patients with a higher risk of lymph node invasion (>5%).2 The extended template according to the NCCN guidelines includes nodes between the external iliac vein/pelvic side wall/ bladder medially/ pelvic floor, and the proximal extent is to the internal iliac artery. According to the EAU guidelines, the extended template includes the obturator, external iliac, and internal iliac nodes, as can be seen in figure 3.
Figure 3 – Extended pelvic lymph node dissection:

Dr. Schaeffer addressed the important point of whether the extent of pelvic lymph node dissection matters when the nodes are all negative (pN0). In a study published in 2018, 28,000 men with NCCN intermediate or high-risk disease were identified using the SEER database. All these men underwent radical prostatectomy with pelvic lymph node dissection and had no evidence of lymph node invasion.3 The study showed that if more than 11 nodes are removed, there is an improved 6-years prostate cancer-specific survival rate (99.5% vs. 98.1%, p=0.014). On multivariable analysis, the risk of prostate cancer-specific mortality decreased by 4.5% for every additional removed lymph node (Figure 4).
Figure 4: Extent of pelvic lymph node dissection with no evidence of pelvic lymph node involvement (pN0):
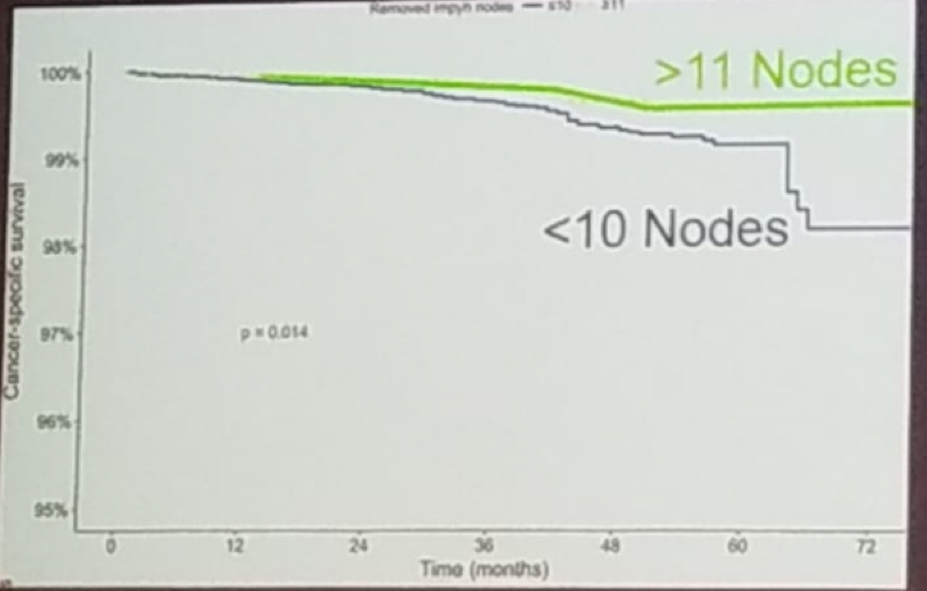
* Preisser et al. Prostate 2018
The extent of pelvic lymph node dissection also matters when the nodes are positive (pN1). The prognosis is affected by the absolute number of lymph nodes involved. Patients with more than four positive lymph nodes have significantly worse cancer-specific survival (HR 2.7, 1.1-6.8, p=0.03) and overall survival (HR 2.3, 1.2-4.6, p=0.01) than patients with less than four positive nodes. (4) The prognosis is also affected by the absolute lymph node yield. There is a stepwise improvement in survival with increase in the lymph node count. Less than 15% of lymph node involvement is associated with improved metastasis-free survival, and extensive lymph node dissection is a key oncologic principle.
Dr. Montorsi continued to describe what had happened to the patient at this time. The patient underwent robotic radical prostatectomy with extended pelvic lymph node dissection in June 2015. The pathology demonstrated a pT3b disease, with a Gleason score of 8 (4+4), 38 nodes removed and four positive nodes, two on the right obturator and two on the right external iliac area. Surgical margins demonstrated 1 mm positive base margin (Gleason pattern 4). The final pathology was pT3b Gleason 4+4, R1pN1. One month following surgery, the first PSA was undetectable, the patient required one pad per day for mild incontinence, and the patient had erectile dysfunction.
Dr. Schaeffer concluded his talk summarizing the functional impact of postoperative radiotherapy in patients such as this. The functional recovery is impacted by postoperative radiotherapy and androgen deprivation therapy (ADT). If only RT is given, there is a 4% higher incontinence rate, while if ADT + radiotherapy is given, there is an 11% risk of greater incontinence. The sexual function and quality of life are also impacted. Lastly, it is important to remember that all outcomes that are reported are usually from centers of excellence, and are not necessarily similar in other centers.
Presented by: Edward Schaeffer, MD, Ph.D. is Chair of the Department of Urology at Feinberg School of Medicine and Program Director of the Genitourinary Oncology Program at the Robert H. Lurie Comprehensive Cancer Center of Northwestern University
Written By: Hanan Goldberg, MD, Urologic Oncology Fellow (SUO), University of Toronto, Princess Margaret Cancer Centre @GoldbergHanan
References:
- Kasivisvanathan V et al. MRI-Targeted or Standard Biopsy for Prostate-Cancer Diagnosis, May 10, 2018 N Engl J Med 2018; 378:1767-1777 DOI: 10.1056/NEJMoa1801993
- Mottet etl al. EAU-ESTRO-SIOG Guidelines on Prostate Cancer. Part 1: Screening, Diagnosis, and Local Treatment with Curative Intent. Eur Urol. 2017 Apr;71(4):618-629. doi: 10.1016/j.eururo.2016.08.003. Epub 2016 Aug 25. Eur Urol 2017
- Preisser et al. Racial disparities in lymph node dissection at radical prostatectomy: A Surveillance, Epidemiology and End Results database analysis. Int J Urol. 2018 Nov;25(11):929-936. doi: 10.1111/iju.13780. Epub 2018 Aug 26.
- Moris et al.Impact of Lymph Node Burden on Survival of High-risk Prostate Cancer Patients Following Radical Prostatectomy and Pelvic Lymph Node Dissection. Front Surg. 2016; 3: 65.Published online 2016 Dec 16. doi: 10.3389/fsurg.2016.00065
Further Related Content:
Oligometastatic Disease: Tumor Board, A Radiation Oncologist Perspective by Bridget Koontz, MD
Oligometastatic Disease: Tumor Board - Medical Oncologist Perspective by Silke Gillessen, MD
Oligometastatic Disease: Tumor Board - A Medical Oncologist Perspective by Michael Morris, MD
Oligometastatic Disease: Tumor Board - A Radiation Oncologist Perspective by Gert de Meerleer, MD


