In ARAMIS, patients with nmCRPC (n=1509) were randomized 2:1 to darolutamide or placebo with androgen deprivation therapy. The ARAMIS trial was unblinded at the primary analysis, after which all patients could receive open-label darolutamide, and tolerability was assessed every 16 weeks. Pharmacodynamic modeling investigated the association between treatment response (maximum PSA decline from baseline) and OS at 2 years using a Cox proportional hazards model. Landmark sensitivity analyses adjusted for the baseline covariates of PSA, PSADT (>6 vs <=6 months), age, ECOG performance status, and Gleason score (>=7 vs 7) were conducted to confirm the correlation between PSA response at week and subsequent OS in patients with PSA data at week 16.
In patients randomized to darolutamide, the median duration of treatment was 18.5 months (range: 0-48 months) during the double-blind period and 25.8 months (range: 0-59 months) during the double-blind plus open-label periods. For patients randomized to placebo, the median duration of treatment was 11.6 months (range: 0-45 months) during the double-blind period and 11.0 months (range: 1-12 months) for patients who crossed over to darolutamide during the open-label period. Darolutamide was consistently well tolerated during the double-blind and open-label treatment periods. Similar to discontinuation due to adverse events, the discontinuation rate due to disease progression was lower in the darolutamide versus placebo arms during the double-blind period (12.5 vs 25.3%) and remained similar (12.6%) with extended treatment during the double-blind + placebo period. Almost all patients were able to receive the full planned dose of darolutamide during the double-blind and double-blind + open-label periods (98.8%), and the majority of patients who had dose modifications were re-escalated to the full dose:

Most patients randomized to darolutamide (double-blind 84.0%; double-blind + open-label 84.5%) had a PSA response (defined as a >=50% decline from baseline) versus 7.9% of patients randomized to placebo during the double-blind period and 31.6% who crossed over from placebo to darolutamide during the open-label period. Using pharmacodynamic modeling, longer OS was positively associated with maximum PSA decline (defined as >=90% decline from baseline) in darolutamide-treated patients. Additionally, most patients (95%) with a maximum PSA decline >=90% from baseline were still alive after 2 years. Landmark sensitivity analyses of patients with PSA data at week 16 confirmed a positive association between maximum PSA decline at week 16 and subsequent OS:
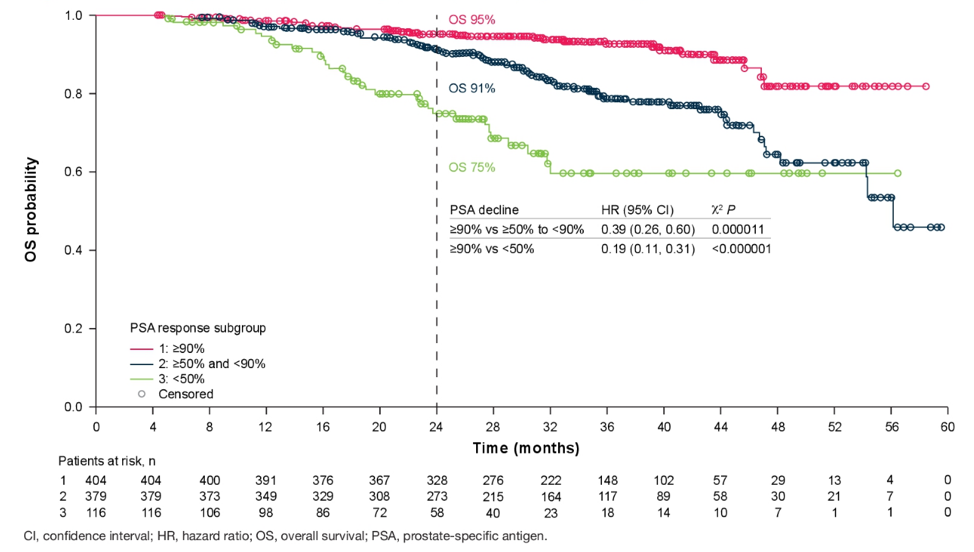
Adjusting for differences in baseline covariates had little impact on OS in the landmark analysis
- Adjusting OS in patients with PSA decline >=90% vs >=50%: HR 0.39, 95% CI 0.25-0.59)
- Adjusting OS in patients with PSA decline >=90% vs <50%: HR 0.18, 95% CI 0.11-0.31
Dr. Fizazi concluded his presentation of ARAMIS with the following take-home messages:
- Darolutamide remained well tolerated with extended treatment at the recommended dose of 600 mg twice daily, which allows patients to maintain their quality of life long term
- Almost all patients with nmCRPC were able to receive the full planned dose, with low rates of dose modifications
- Long-term treatment with darolutamide extends survival and delays disease progression in men with nmCRPC
- The majority of patients randomized to darolutamide (~84%) versus placebo (8%) showed a >=50% decline in PSA from baseline. Pharmacodynamic modeling predicted a positive association between maximum PSA decline from baseline and longer OS
- Darolutamide has a highly favorable tolerability and safety profile, and real-world experience comparing androgen receptor inhibitors will be of great clinical interest
Clinical trial information: NCT02200614
Presented by: Karim Fizazi, MD, PhD, Institut Gustave Roussy and University of Paris Saclay, Villejuif, France
Written by: Zachary Klaassen, MD, MSc – Urologic Oncologist, Assistant Professor of Urology, Georgia Cancer Center, Augusta University/Medical College of Georgia Twitter: @zklaassen_md at the 2021 American Society of Clinical Oncology (ASCO) Annual Meeting, Virtual Annual Meeting #ASCO21, June, 4-8, 2021
References:
- Fizazi K, Shore N, Tammela TL, et al. Darolutamide in nonmetastatic castration-resistant prostate cancer. N Engl J Med. 2019;380(13):1235-1246.
- Fizazi K, Shore N, Tammela TL, et al. Nonmetastatic, Castration-Resistant Prostate Cancer and Survival with Darolutamide. N Engl J Med. 2020 Sep 10;383(11):1040-1049.


