Primary care physicians (PCPs) are tasked as the frontline experts on every disease imaginable, and therefore, maintaining the up-to-date education needed for prostate cancer detection may be a challenge. The United States Preventive Services Task Force’s recommendations on prostate-specific antigen (PSA) screening test has added ongoing confusion regarding the screening criteria for men at risk for prostate cancer. Urologists are primarily responsible for diagnosing prostate cancer, staging the disease, and providing an individualized treatment path. PCPs can—and should—play a central role in the initial evaluation and management of potential prostate cancer patients.
PCPs, including internists and family physicians, order approximately 90% of all PSA screening tests. For more than 25 years, the PSA test has been used to screen for prostate cancer, yet it has a low specificity and cannot differentiate between clinically significant and indolent cancer. Over-screening and overdiagnosis of indolent disease—and the added stress of visiting a doctor during a pandemic—can lead to marked patient anxiety as well as physical adverse effects from the biopsy and unfortunately, the potential for overtreatment of prostate cancer. Despite the PSA test’s limitations, we recognize that this screening test followed by a digital rectal exam (DRE) is beneficial for men and is a necessary first step in considering a diagnosis for prostate cancer. That said, additional tests may also be needed.
PSA test results should not automatically sound the alarm for immediate urology referral. Elevated PSA levels can result for several reasons: age-related prostate growth, benign prostatic hyperplasia, urinary tract infection, prostatitis, etc. Fortunately, today, there are biomarker tests that address the unmet clinical need that the PSA test presents. Studies show noninvasive biomarker tests, when combined with PSA screening, can improve patient stratification and help improve the early detection of clinically significant prostate cancer.
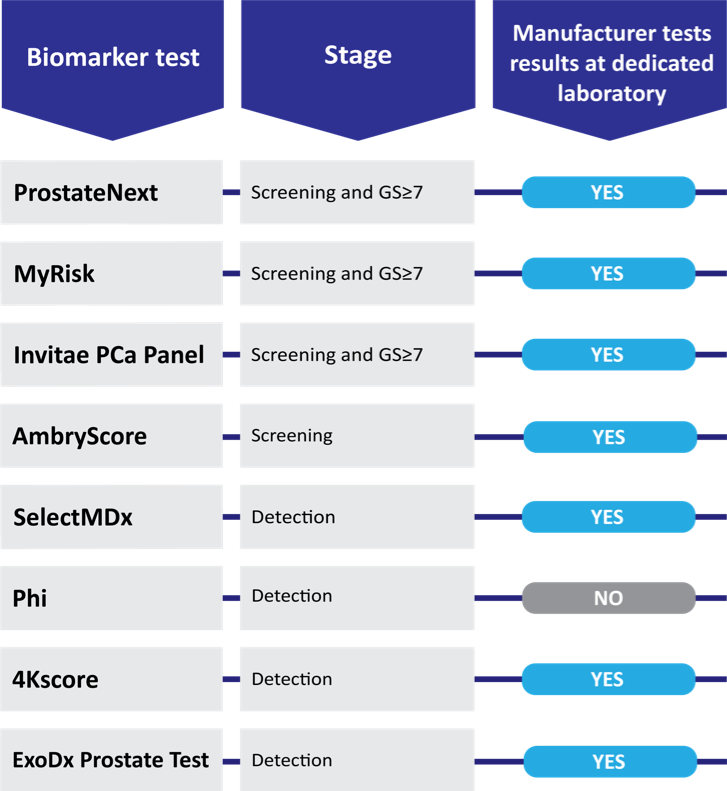
Biomarker tests have a role in primary care, and we need to embrace new technologies to help our patients. Although they are not mandated as first-line screening tests in conjunction with the PSA test, the National Comprehensive Cancer Network’s (NCCN) Guideline for Prostate Cancer Early Detection recognizes that biomarker tests can further define the probability of high-grade cancer before an initial biopsy or following a negative prostate biopsy. For example, SelectMDx is a urine test included in the NCCN guidelines that helps detect cancer after a DRE but before an invasive biopsy. If SelectMDx returns negative, PCPs may confidently continue to monitor for prostate cancer without an immediate referral of their patient to a specialist. With so much controversy surrounding the PSA, implementing biomarker tools would assist shared decision-making with patients and ease concern of missed cancer. Here is a chart of early detection prostate cancer biomarker tests that may benefit PCPs’ shared decision-making with patients:
COVID-19 has galvanized health care professionals’ requirements for both efficiency and safety for patient care, highlighting the need for providers to reassess their specific practices for optimizing care. For urologists and PCPs, an understanding of biomarker options available for appropriately diagnosing prostate cancer is key. We believe that PCP understanding and utilization of prostate biomarker tests could significantly reduce prostate cancer mortality in the United States by ensuring that clinically significant prostate cancers are efficiently detected. Additionally, increased use of biomarker tests may also further reduce the overdiagnosis and overtreatment trends of prostate cancer which have been historically observed.
The bottom line is this: a prostate biopsy should not be performed unless the risk of detecting a clinically significant aggressive tumor is likely. To reduce overtreatment, PCPs can help evaluate patients at risk for prostate cancer before directing them to a specialist by discussing the benefits and use of a prostate biomarker test. Optimizing clinically significant prostate cancer detection will thus achieve improved healthcare outcomes with enhanced resource utilization. Implementing this step will detect significant cancers earlier while avoiding many unnecessary biopsies, ultimately saving time, money, and emotional stress and improving the trajectory of lives saved.
Written by: Neal D. Shore, MD, FACS is the Medical Director for the Carolina Urologic Research Center. He practices with Atlantic Urology Clinics in Myrtle Beach, South Carolina. Dr. Shore has conducted more than 350 clinical trials, focusing mainly on GU Oncology, and serves on the Executive Boards of: Society of Urologic Oncology Board, Bladder Cancer Advocacy Network, and is Immediate Past President, Large Urology Group Practice Association. He is a founder for both: CUSP Clinical Trials Consortium, as well as for DASHKO, large urology practices data registries. He serves as the National Urology Research Director for 21st Century Oncology. He has more than 200 peer-reviewed publications and numerous book chapters; he performs peer review for Lancet Oncology, New England Journal of Medicine, European Urology, Journal Urology, Urology, BJUI, PCPD, and numerous other high-impact scientific journals. A graduate of Duke University and Duke University Medical School, Dr. Shore completed a 6-month clinical research fellowship in Pretoria, South Africa, and then completed his General Surgery/Urology training at New York Hospital Cornell Medical Center and at Memorial Sloan-Kettering Cancer Center in New York City. He is a Fellow of the American College of Surgeons.
Michael Shawn Cookson, MD, MMHC, is Professor and Chairman of the Department of Urology at the University of Oklahoma College of Medicine. He holds the Donald D. Albers Endowed Chair in Urology. Dr. Cookson is one of the most experienced urologic cancer surgeons in the country and has performed more than 1,500 robotic surgeries and more than 3,000 major open surgeries for urologic cancer. He has a special interest in prostate and bladder cancer. He commonly performs both open and robotic radical prostatectomy, radical cystectomy and complex urinary diversions. Dr. Cookson has devoted a significant portion of his academic career to the management of patients with urologic cancers, with a strong emphasis on Clinical Guidelines, Education and Evidenced Based Medicine. In 2018, he received the Presidential Citation Award from the American Urologic Association for educational contributions. For his efforts in teaching at Vanderbilt, he was awarded the Michael Rodriquez Award. He currently serves as the President of the Society of Urologic Oncology and is the President-elect to the AUA’s South Central Section.
Published Date: October 2020
Related Content:


