Background
While there have been dramatic changes in treatment options for patients with advanced prostate cancer over the past 5 years, perhaps the greatest change has been for patients with non-metastatic castration-resistant prostate cancer (nmCRPC). Prior to February 14, 2018, there were no agents approved by the United States Food and Drug Administration (FDA) for men with nmCRPC. Since then, three agents have been approved (apalutamide, enzalutamide, and darolutamide, in chronologic sequence of approval). While approval was initially based on improvements in metastasis-free survival, the seminal phase III trials for each of these agents have now reported overall survival data.
Before launching into a discussion of these overall survival data, it is worthwhile to consider the context of nmCRPC. Since the seminal work of Huggins and Hodges1 seventy years ago, androgen deprivation therapy (ADT) has formed the cornerstone of management for advanced prostate cancer with indications including concurrent therapy with primary curative-intent radiotherapy, salvage therapy after recurrence following local therapy, and in the treatment of metastatic disease. While efficacious, nearly all patients will eventually develop castration resistance with disease progression despite castrate levels of testosterone. Among patients who receive ADT for biochemical recurrence following radical prostatectomy or radiotherapy, the development of castration resistance typically occurs prior to identification of metastasis on conventional imaging, the treatment-induced disease state known as non-metastatic castration-resistant prostate cancer. NmCRPC is typically identified on the basis of the PCWG3 consensus definition for PSA progression on ADT, namely a 25% PSA increase from nadir (starting PSA ≥1.0 ng/mL), with a minimum rise of 2 ng/mL in the setting of castrate testosterone (< 50 ng/dL).2 In these patients, treatment is aimed at delaying the development of metastasis, preserving quality of life, and increasing overall survival.
Study Design – ARAMIS, PROSPER, and SPARTAN
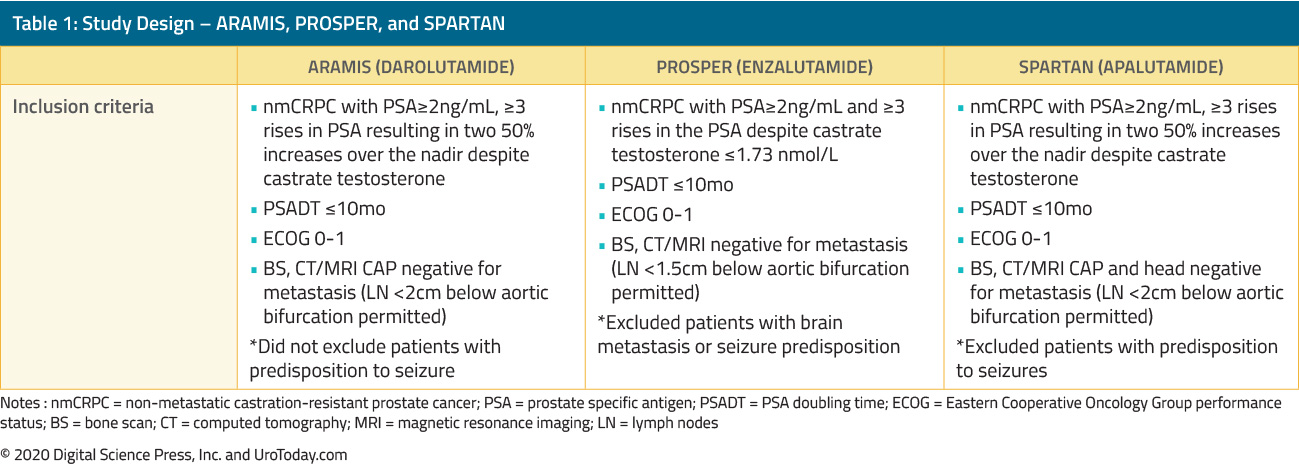
As may be expected on the basis of a generally similar mechanism of action of the agents under study and same treatment indication, the study methodology and inclusion criteria for each of these three trials were broadly similar. Recruited patients had nmCRPC and high-risk features for metastasis including an absolute PSA ≥ 2ng/mL with sequential rises despite castrate testosterone3-5. The SPARTAN and ARAMIS trials also required two 50% increases in the PSA over the nadir. Further, each trial mandated that patients must have a PSA doubling time of less than 10 months.All men had baseline staging imaging consisting of a bone scan and CT chest with either a CT or MRI of the abdomen and pelvis confirming the absence of metastatic disease. Local lymphadenopathy below the level of the aortic bifurcation was permitted if it was less than 2cm in size for patients in ARAMIS and SPARTAN and less than 1.5cm in size for those in PROSPER. SPARTAN also required patients to have a CT head confirming the absence of intracranial metastasis.
ARAMIS uniquely allowed patients with seizure-predisposing conditions, whereas such patients were excluded from the other two trials. All three trials excluded patients with prior exposure to non-steroidal anti-androgens. PROSPER did not permit enrollment if the patient had any prior chemotherapy exposure, whereas SPARTAN and ARAMIS permitted chemotherapy if it had been given in the neoadjuvant or adjuvant setting.
Each of the three trials used a 2:1 randomization schema with allocation to the experimental agent and placebo, respectively. Randomization was stratified according to PSA doubling time and the use of bone-targeting (osteoclast-targeting) agents. In each trial, patients continued on ADT throughout whether randomized to active agent or placebo.
The primary endpoint for each study was metastasis-free survival, defined as distant metastases or death from any cause. Previously, the ICECaP Working Group demonstrated that metastasis-free survival is a strong surrogate of OS among men with localized prostate cancer6. Key secondary and exploratory endpoints included overall survival, time to first subsequent anti-neoplastic therapy, time to first cytotoxic chemotherapy, time to pain progression, and adverse events.
Primary analysis – metastatic-free survival and toxicity
The primary analyses of PROSPER and SPARTAN were published in 20184,5 while ARAMIS was published in 20193 and have been extensively examined previously. At the time of primary analysis and publication, each of these studies demonstrated significant prolonged metastasis-free survival with the use of enzalutamide (hazard ratio 0.29, 95% confidence interval 0.24-0.35), apalutamide (hazard ratio 0.28, 95% confidence interval 0.23-0.35), or darolutamide (hazard ratio 0.41, 95% confidence interval 0.34-0.50).While overall survival data at the time of primary analysis was immature within each study, pooled analysis in a meta-analysis demonstrated a signal for improved overall survival based on these data (hazard ratio 0.74, 95% confidence interval 0.61-0.90)7.
Updated analysis – overall survival
The first trial to report a secondary analysis with updated overall survival data was SPARTAN. These data were presented at ESMO 2019 and concurrently published in Annals of Oncology8. At that time, with 67% of required overall survival events observed, there was evidence of improved overall survival (hazard ratio 0.75, 95% confidence interval 0.59 to 0.96) though this didn’t reach statistical significance on the basis of the prespecified O’Brien–Fleming boundary of 0.0121.However, at the 2020 ASCO Virtual Annual Meeting, updated overall survival data for all three studies were presented and data for PROSPER were concurrently published in the New England Journal of Medicine9. In each trial, these updated data demonstrated improved overall survival for patients treated with novel oral anti-androgens for nmCRPC. Since that time, updated overall survival data from SPARTAN were published in European Urology10 on September 6th and from ARAMIS were published in the New England Journal of Medicine11 on September 10th.
Overall survival – ARAMIS
Beginning with the most recently published data, the recent publication of data from ARAMIS was based on a data cut-off of November 15, 2019, with a median follow-up of 29.0 months11. At this point, 49% of patients who were randomized to receive darolutamide were still receiving study agent and 86% of those randomized to placebo who crossed over without progression at the time of unblinding were still receiving study agent.Overall survival analysis in the ARAMIS trial was performed after 254 deaths were noted (of an initially planned 240 deaths): 148 among those randomized to darolutamide and 106 among those randomized to placebo. In this cohort, with 29.0 months median follow-up, median overall survival was not reached in either the darolutamide or placebo arm. However, the proportion of patients who were alive at 3 years was significantly higher among those randomized to darolutamide (83%, 95% confidence interval 80 to 86) than those randomized to placebo (77%, 95% confidence interval 72 to 81). Cox proportional hazard modeling demonstrated a 31% reduction in the risk of death for those randomized to darolutamide (hazard ratio 0.69, 95% confidence interval 0.53 to 0.88).

This beneficial effect was observed across subgroups defined according to PSA doubling time (≤6 months vs >6 months), geographic region, presence of lymph node involvement, baseline ECOG status.
Notably, prostate cancer-related mortality (80 of 149 in the darolutamide group and 56 of 106 in the placebo group) was more common than other causes of mortality among patients in this cohort.
In the ARAMIS trial, 307 of 554 patients randomized to placebo received subsequent treatment with darolutamide or other proven life-prolonging therapy at the time of progression. The most commonly used of these agents were docetaxel, abiraterone acetate, and enzalutamide.
Overall survival – SPARTAN
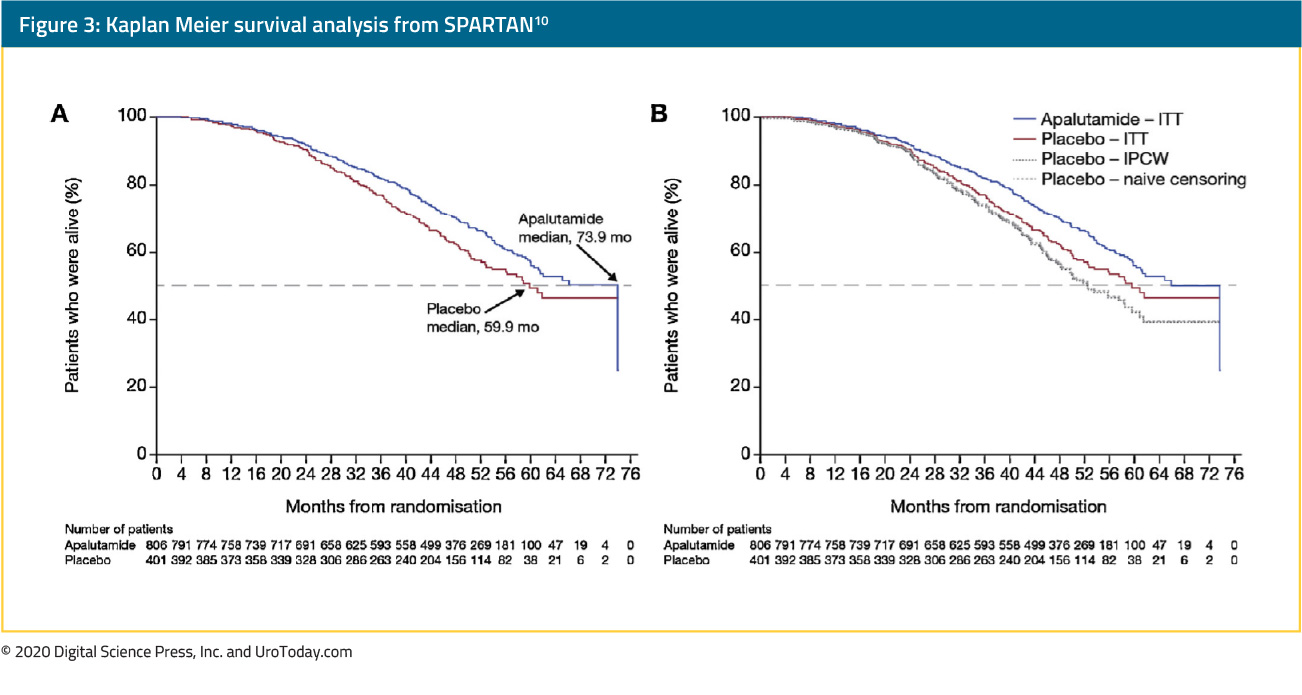
Just prior to the publication of these data from ARAMIS, the final analysis of SPARTAN was published in European Urology10. Following publication of the primary analysis, 76 patients randomized to placebo crossed over to apalutamide without disease progression. At the time of final data cut-off with a median follow-up of 52 months, 237 of 803 patients originally randomized to apalutamide continued on experimental therapy and 46 of 76 patients who crossed over remained on therapy.At the time of data cut-off, there were 428 deaths (of a required 427 events), of which 274 occurred among those randomized to apalutamide and 154 among those randomized to placebo. Median overall survival was significantly longer among men receiving apalutamide (73.9 months, 95% confidence interval 61.2 to not reached) than placebo (59.9 months, 52.8 to not reached), corresponding to a relative reduction of 21.6% in the risk of death (hazard ratio 0.784, 95% confidence interval 0.64 to 0.96, p-value 0.0161; O’Brien–Fleming threshold of 0.046 for statistical significance).
The authors conducted further analyses, accounting for those patients randomized to placebo who crossed over to apalutamide therapy without disease progression.
As with the results of the ARAMIS trial, the observed beneficial effect of apalutamide in SPARTAN was consistent across subgroups based on patient age, race, region, performance status, PSA, PSA doubling time, use of a bone-sparing agent, and presence of locoregional disease.
Overall survival – PROSPER
While final results for each of these three trials were presented at the 2020 ASCO Virtual Annual Meeting, the data from PROSPER were the first to be published, in the New England Journal of Medicine9.With a data cut off of October 15, 2019, median follow-up in the PROSPER trial was approximately 48 months. There was a total of 466 deaths, of which 288 occurred among those randomized to enzalutamide and 178 among those randomized to placebo. As with patients in the ARAMIS trial, prostate cancer-related deaths were more common than non-prostate cancer-related deaths in this cohort, both among patients randomized to enzalutamide (178 and 110 deaths, respectively) and those randomized to placebo (136 and 42 deaths, respectively).
Median overall survival was 67.0 months (95% confidence interval 64.0 to not reached) among patients receiving enzalutamide and 56.3 months (95% confidence interval 54.4 to 63.0) among patients receiving placebo, corresponding to a 27% relative reduction in the risk of death for patients receiving enzalutamide (hazard ratio 0.73; 95% confidence interval 0.61 to 0.89; p = .001). The authors utilized O’Brien–Fleming-type alpha-spending function on the basis of three planned analyses. As a result of adjustment for multiplicity of testing, the p-value threshold for significance was 0.021. Thus, the analysis of overall survival met the threshold for statistical significance.

Again, these results were consistent across subgroups defined based on region, age, performance status, PSA doubling time, PSA value, use of bone-targeting agents, Gleason score, LDH at baseline, and hemoglobin at baseline.
Toxicity considerations
These updated analyses did not demonstrate any new safety or toxicity signals, as compared to the initial study reports. A previous systematic review and network meta-analysis demonstrated that patients treated with apalutamide, darolutamide, or enzalutamide were significantly more likely to experience grade 3 or 4 adverse events than those receiving placebo (OR: 1.47, 95% CI:1.27-1.71)7. Among the novel oral anti-androgens, there was a 61% chance that darolutamide was the preferred agent (SUCRA 0.50) and 55% chance that apalutamide was the least preferred agent (SUCRA 0.19) with respect to rates of grade 3 and 4 adverse events.At the ASCO 2020 virtual meeting, new data was presented comparing the safety outcomes of darolutamide versus abiraterone or enzalutamide using matching-adjusted indirect comparison. Individual patient-level data from ARAMIS were selected and re-weighted to match the inclusion criteria and baseline characteristics published in SPARTAN and PROSPER (the authors did not have access to these trial’s individual patient-level data). Darolutamide showed a numeric advantage over apalutamide on adverse events leading to treatment discontinuation and adverse events leading to death, whereas apalutamide showed a numeric advantage over darolutamide on any serious adverse events. Among individual adverse events, darolutamide generally exhibited favorable results compared to apalutamide, showing advantages for mental-impairment disorder, diarrhea, nausea, fatigue, and severe fatigue, as well as reaching statistical significance for falls, rash, and fracture. In comparison to enzalutamide, darolutamide showed a numeric advantage over enzalutamide on any serious adverse events, adverse events leading to treatment discontinuation, and adverse events leading to death. Among individual adverse events, darolutamide generally exhibited favorable results compared to enzalutamide, showing an advantage for nausea, asthenia, and headache, as well as reaching statistical significance for falls, dizziness, mental-impairment disorder, hypertension, fatigue (all grades), and severe fatigue.
The ongoing DaroAct trial seeks to assess differences in physical and cognitive toxicity related outcomes for patients with both non-metastatic and metastatic CRPC. Patients are randomized in a 2:1 fashion to either darolutamide or enzalutamide, with stratification based on age (≤75 years vs >75 years). This will allow for the first randomized, direct comparison of these treatment approaches.
Conclusions
The treatment landscape for patients with nmCRPC has changed dramatically since 2018. There are now three FDA approved agents that were initially approved on the basis of improvements in metastasis-free survival which have now demonstrated benefits in overall survival. An editorial published in European Urology12 in conjunction with the publication of the final SPARTAN data suggests some caution, however. First, the authors cautioned that all of these data were among men with a PSA doubling time of <10 months and thus should not be extrapolated to all men with nmCRPC. Second, ongoing advances in imaging approaches (including the use of PSMA-based imaging) will likely continue to refine the definition of nmCRPC.Beyond these considerations of the nature of the study population, the authors highlight potential complications of the use of the novel oral anti-androgens in nmCRPC with a focus on the increased risk of bone density loss and cardiovascular toxicity12. This is particularly important as the duration of treatment exposure is typically much longer in patients who receive these agents of nmCRPC than those who receive them for mCRPC, either before or following cytotoxic chemotherapy.
The editorial authors highlight the importance of a baseline evaluation of cardiac risk factors before prescribing these agents. They also speculate regarding the role for use of GnRH antagonists (rather than GnRH agonists), the use of intermittent novel oral anti-androgens, and the use of cardioprotective agents (including statins, aspirin, beta-blockers, and angiotensin II-receptor blockers).
Written by: Zachary Klaassen, MD, MSc, Assistant Professor of Urology, Georgia Cancer Center, Augusta University/Medical College of Georgia, Atlanta, Georgia
Published Date: September 17th, 2020


