Athens, Greece (Urotoday.com) Dr. Ischia was asked to describe the strengths and limitations of the most recent imaging modality for prostate cancer introduced to the world – PET PSMA. Prostate-specific membrane antigen (PSMA) is a transmembrane protein expressed on prostate cells. It is overexpressed in virtually all prostate cancers (95%). The expression is increased in poorly differentiated, metastatic and castration-resistant carcinomas.
The strengths of the PSMA include its high sensitivity and specificity, and its use for theranostic purposes. Its weaknesses include the lack of data on its utility, no available guidelines to help us decide how to treat with it, a considerable rate of false-positive and its high cost.
PET PSMA can be used for:
- Primary staging
- Biochemical recurrence
- PSMA theranostics
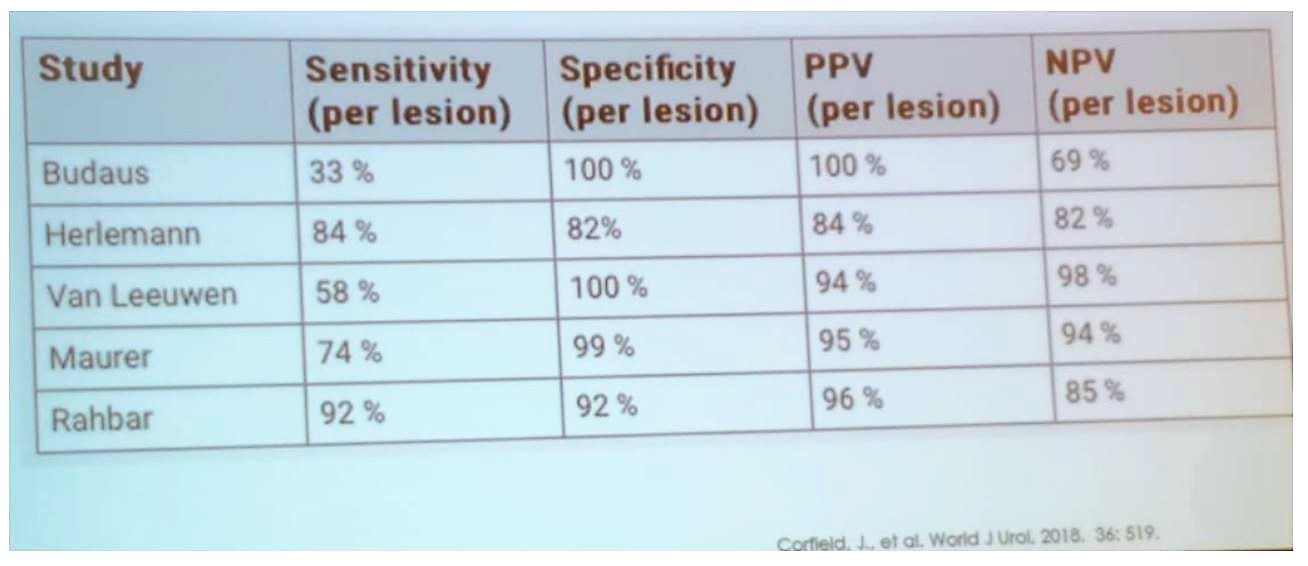
In primary staging, the performance of PSMA is quite impressive with high specificity, sensitivity, and an impressive negative predictive value, as can be seen in table 1. The minimum size of a positive lymph node for diagnosis with PET PSMA is 4 mm.1 It is also important to remember that approximately 5-10% of prostate cancers do not express PSMA.
Table 1 – PSMA performance for primary staging:
In Australia, the proPSMA trial is now accruing in 10 centers across Australia. This is a prospective randomized multi-center study assessing the impact of PSMA PET/CT imaging for staging high-risk prostate cancer prior to curative-intent surgery or radiotherapy, led by Dr. Michael Hofman.2 The researchers aim to accrue 300 men with the endpoints being accuracy of scanning modalities and impact on outcomes, for instance – can adding PSMA to mpMRI reduce unnecessary biopsy in men at risk of prostate cancer.
Next, Dr. Ischia discussed the role of PSMA in PSA recurrence after primary definitive therapy. PSMA shows high sensitivity even at low PSA levels, as can be seen in table 2, with a specificity of over 95%.3 The use of PSMA in this setting has been incorporated into the EAU guidelines supporting its role, as can be seen in Table 3.
Table 2 – PSMA positive scan rate at different PSA levels:
Table 3 – EAU guidelines recommendations regarding PSMA following radical prostatectomy:
Lastly, Dr. Ischia discussed the role of PET PSMA in metastases directed therapy. Dr. Ischia emphasized that this is still regarded as an experimental therapy, but might potentially be able to avoid or delay the initiation of androgen deprivation therapy (ADT), if proved beneficial.
The use of theranostic Lutetium-PSMA PET is still regarded as experimental with a very promising biologic concept of being able to treat the disease locally, using a PSMA targeting ligand radiolabeled with Lutetium-1774,5 (Figure 1).
Dr. Ischia concluded his talk reminding everyone that the role of conventional imaging (CT and bone scans) is still important as the current guidelines depend on it. PSMA PET is gradually changing the games but the “rules of the game” are still not clear.
Figure 1 – Theranostic LU-PSMA PET:
Presented by: Joseph Ischia, MBBS, PhD, University of Melbourne, Australia
Written By: Hanan Goldberg, MD, Urology Department, SUNY Upstate Medical University, Syracuse, New-York, USA @GoldbergHanan at the 39th Congress of the Société Internationale d'Urologie, SIU 2019, #SIUWorld #SIU2019, October 17-20, 2019, Athens, Greece
References:
1. Murphy DG, Hofman M, Lawrentschuk N. Bringing clarity or confusion? The role of prostate-specific membrane antigen positron-emission/computed tomography for primary staging in prostate cancer. 2017; 119(2): 194-5.
2. Hofman MS, Murphy DG. A prospective randomized multicentre study of the impact of gallium-68 prostate-specific membrane antigen (PSMA) PET/CT imaging for staging high-risk prostate cancer prior to curative-intent surgery or radiotherapy (proPSMA study): clinical trial protocol. 2018; 122(5): 783-93.
3. Perera M, Papa N, Roberts M, et al. Gallium-68 Prostate-specific Membrane Antigen Positron Emission Tomography in Advanced Prostate Cancer—Updated Diagnostic Utility, Sensitivity, Specificity, and Distribution of Prostate-specific Membrane Antigen-avid Lesions: A Systematic Review and Meta-analysis. European Urology.
4. Hofman MS, Violet J, Hicks RJ, et al. [(177)Lu]-PSMA-617 radionuclide treatment in patients with metastatic castration-resistant prostate cancer (LuPSMA trial): a single-centre, single-arm, phase 2 study. The Lancet Oncology 2018; 19(6): 825-33.
5. Cimadamore A, Cheng M, Santoni M, et al. New Prostate Cancer Targets for Diagnosis, Imaging, and Therapy: Focus on Prostate-Specific Membrane Antigen. Frontiers in Oncology 2018; 8(653).