(UroToday.com) On Sunday, September 11, 2022, in the Mini Oral session of the European Society for Medical Oncology (ESMO) Annual Congress focusing on non-prostate genitourinary cancers, Dr. Porta provided an updated analysis of the CLEAR trial examining the role of the combination of lenvatinib and pembrolizumab, as compared to sunitinib, as first-line therapy in advanced renal cell carcinoma (aRCC). In the primary analysis, there was significantly improved progression-free survival and overall survival in patients treated with the combination approach.
While previously published, the CLEAR trial enrolled patients with aRCC who had not received any prior systemic therapy and randomized them in a 1:1:1 fashion to receive Lenvatinib 20 mg PO QD + pembrolizumab 200 mg IV Q3W; Lenvatinib 18 mg + everolimus 5 mg PO QD; or sunitinib 50 mg PO QD (4 weeks on/2 weeks off). Randomization was stratified by geographic region and MSKCC prognostic group.
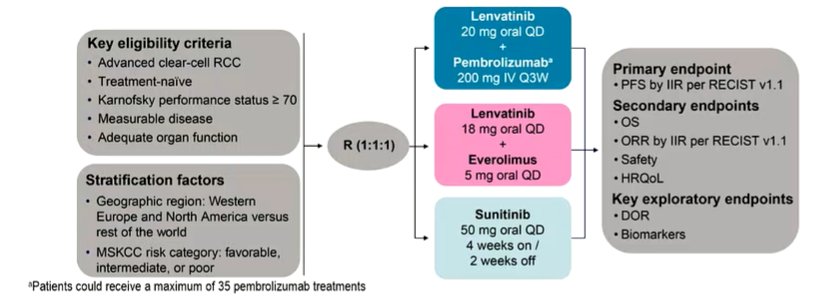
This descriptive follow-up analysis with a data cut-off of March 31, 2021 examines efficacy outcomes including among patients who completed 2 years of pembrolizumab and then continued on Lenvatinib monotherapy.
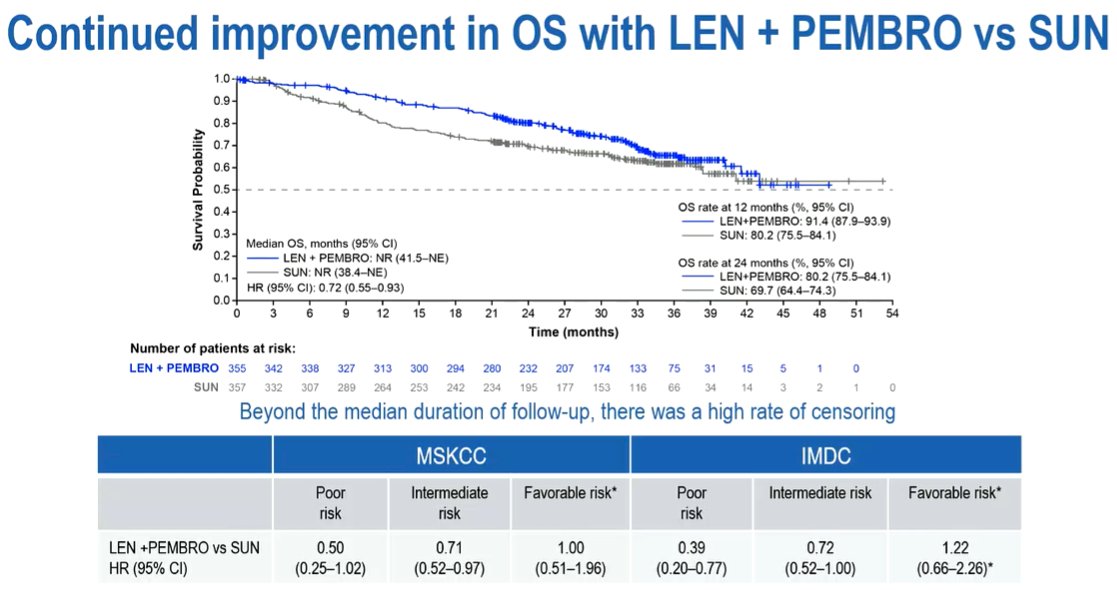
With this prolonged duration of follow-up of a median of 33.7 months among those randomized to Lenvatinib + pembrolizumab (N = 355) and 33.4 months for those randomized to sunitinib (N = 357), progression-free survival (median 23.3 vs 9.2 months) and ORR (71.0% vs 36.1%) continued to be improved with Lenvatinib + pembrolizumab vs sunitinib. Further, the duration of response was longer with Lenvatinib + pembrolizumab, compared to sunitinib (median, 26.0 vs 14.7 months).

Among those patients who completed 2 years of pembrolizumab (n = 101 of 355 pts), most (n = 65) had IMDC intermediate/poor risk disease and fewer (n = 36) had favorable risk disease, in keeping with the overall intention-to-treat population. Further, those patients who completed two-years of pembrolizumab had a 36-mos OS rate of 94.5%.
Notably, 69 (68.3%) of these patients had treatment-related treatment-emergent adverse events.
Thus, Dr. Porta concluded that, with longer durations of follow-up, the combination of Lenvatinib + pembrolizumab continued to show a clinically meaningful benefit compared to sunitinib, consistent with prior initial analyses of the CLEAR study.
Presented by: Camillo G. Porta, MD, Professor, Bari, Italy
Written by: Christopher J.D. Wallis, University of Toronto, Twitter: @WallisCJD during the 2022 European Society of Medical Oncology (ESMO) Annual Hybrid Meeting, Paris, FR, Fri, Sept 9 – Tues, Sept 13, 2022.
Related Content:
ESMO 2022: Updated Results From the CLEAR Trial and the Final Results of TITAN-RCC Discussion


