One newer option is the Nuro™ Percutaneous Tibial Neuromodulation System (PTNM) device, which produces an adjustable electrical pulse that travels to the sacral nerve plexus via the tibial nerve. A needle electrode is placed cephalad to the medial malleolus to deliver the electrical pulse, and a typical treatment involves a 30-minute session once a week for a duration of 12 weeks.
Kathleen Kobashi, MD of the Virginia Mason Medical Center in Seattle, WA, reported on the results of the RESET study, a multi-center, single arm, prospective study that evaluated changes in UUI episodes and urinary frequency after treatment with the Nuro™ device in drug-naïve OAB subjects. Patients were eligible for the study if they had 3+ UUI episodes demonstrated on a 3-day voiding diary, as well as UUI symptoms for a minimum of 3 months. Patients were excluded from the study if they had a history of prior pharmacotherapy or advanced therapy for OAB, or if they had either stress urinary incontinence or stress-predominant mixed incontinence.
Change in voiding symptoms were evaluated by bladder diaries from baseline through 12 weeks (PTNM sessions 1, 4, 8, and 12), ad quality of life (QOL) changes were similarly assessed with the OABq questionnaire at the same intervals. Safety was evaluated through adverse events (AE) related to the device, procedure, and therapy.
The study population included 121 OAB treatment-naïve subjects, of whom 116 (96%) completed the full twelve sessions. Subjects were primarily female (86%), with a mean age of 64.8 years. At baseline, patients had a mean duration of OAB of 3.4 years, 3.5±2.5 UUI episodes/day and 11.5±2.9 voids/day. Over the course of the study, the subjects were found to have significant reductions from baseline (p<0.0001) at each follow-up visit (Week 1, 4, 8, 12), with a reduction of 2.4±2.1 episodes at session 12 from baseline (Figure 1). Similarly, subjects with urinary frequency (UF) at baseline also reported successive reductions in frequency over the treatment course. After session 12, a statistically significant reduction of 1.7±2.5 voids/day was observed (p<0.0001; Figure 2). QOL was also reported as significantly improved. There were no serious or unanticipated adverse events (AE), but 4 subjects reported medical device site pain (3.3%) and extremity pain (3.3%).
Figure 1

Figure 2
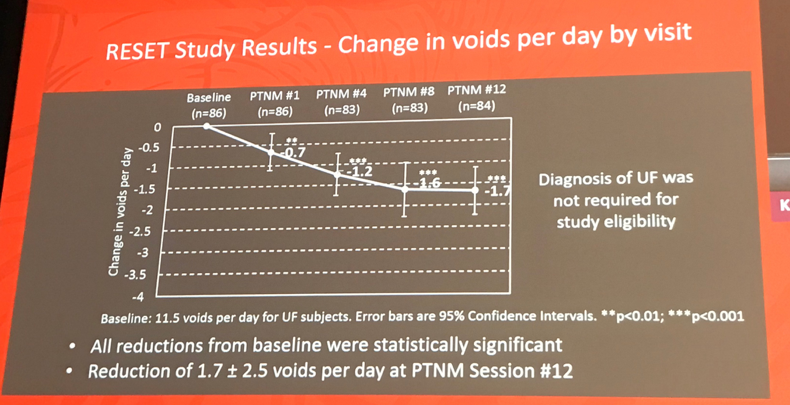
The authors concluded that PTNM using Nuro™ device is an effective and safe treatment for drug-naïve patients with OAB. The data suggest that the Nuro™ device could be used in patients who either opt out of or are not appropriate candidates for pharmacotherapy as an initial treatment of OAB
Presented by: Kathleen Kobashi, MD, Virginia Mason, Seattle, WA,
Co-Authors: Salil Khandwala, MD, Advanced Urogynecology of Michigan, Dearborn, MI, Scott MacDiarmid, MD, Alliance Urology Specialists, Greensboro, NC , Eric Margolis, MD, Urology Center of Englewood, Englewood, NJ, Diane Newman, DNP, University of Pennsylvania, Philadelphia, PA, Victor Nitti, MD, NYU, New York, NY , Peter Sand, NorthShore Medical Group, Evanston, IL, Steven Siegel, Metro Urology Center, Woodbury, MN, Fangyu Kan, Minneapolis, MN, Elizabeth Michaud, Minneapolis, MN
Written by: Judy Choi, MD, Assistant Professor, Department of Urology, University of California, Irvine @judymchoi at the 2018 AUA Annual Meeting - May 18 - 21, 2018 – San Francisco, CA USA


