A total of 1,410 testicular cancer survivors ≥6 months post-cisplatin-based chemotherapy completed comprehensive audiometric assessments (0.25-12 kHz) and detailed questionnaires of sociodemographic, clinical, and health behaviors. Audiometrically-defined hearing loss severity was defined using American Speech-Language-Hearing Association (ASHA) criteria. Multivariable multinomial logistic regression identified factors associated with discrepancies (overestimation and underestimation versus concordance), between patient-reported and audiometrically-defined hearing loss, and multivariable ordinal logistic regression evaluated factors associated with the hearing loss severity.
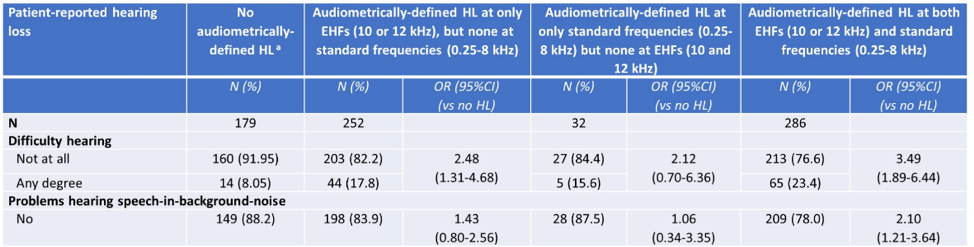
Overall, 34.8% of testicular cancer survivors self-reported hearing loss, while 77.8% had audiometrically-defined hearing loss. Among testicular cancer survivors without tinnitus, those with audiometrically-defined hearing loss at only extended high frequencies (10-12 kHz) (17.8%) or at both extended high frequencies and standard frequencies (0.25-8 kHz) (23.4%) were significantly more likely to self-report hearing loss than those with no audiometrically-defined hearing loss (8.1%) (OR 2.48, 95% CI 1.31-4.68 and OR 3.49, 95% CI 1.89-6.44, respectively):
Older age (OR 1.09; p < 0.0001), absence of prior noise exposure (OR 1.40; p = 0.02), and mixed/conductive hearing loss (OR 2.01; p = 0.0007) were associated with greater underestimation of audiometrically-defined hearing loss severity. Hearing aid use (OR 0.18; p = 0.003) and higher education (p = 0.004) were associated with less underestimation of audiometrically-defined hearing loss severity, while tinnitus was associated with greater overestimation (p < 0.0001). Older age (OR 1.13; p < 0.0001), cumulative cisplatin dose (> 300 mg/m2, OR 1.47; p = 0.0001), and hypertension (OR 1.80; p = 0.0007) were associated with greater ASHA-defined hearing loss severity, whereas post-graduate education (OR 0.58; p = 0.005) was associated with less severe hearing loss.
Dr. Ardeshir-Rouhani-Fard concluded the presentation with the following summary statements:
- Discrepancies between patient-reported and audiometrically-defined hearing loss after cisplatin-based chemotherapy are associated with several factors including age, education, tinnitus, prior noise exposure, use of hearing aids, and conductive hearing loss
- Understanding these factors will help clinicians to better interpret self-reported hearing loss as a surrogate for audiometric assessments
- For survivors who self-report hearing loss, but have normal audiometric findings at standard frequencies, referral to an audiologist for additional testing and inclusion of extended high frequencies in audiometric assessments, should be considered
Presented by: Shirin Ardeshir-Rouhani-Fard, Indiana University School of Medicine, Indianapolis, IN
Written by: Zachary Klaassen, MD, MSc – Urologic Oncologist, Assistant Professor of Urology, Georgia Cancer Center, Augusta University/Medical College of Georgia Twitter: @zklaassen_md at the 2021 American Society of Clinical Oncology (ASCO) Annual Meeting, Virtual Annual Meeting #ASCO21, June, 4-8, 2021


