The authors examined 11,741 patients with prostate cancer who underwent comprehensive genomic profiling as part of routine clinical care (Foundation Medicine Inc.). Among these individuals, the authors assessed the genomic landscape including predominant ancestry which was inferred using a SNP-based approach. In addition, the authors utilized a separate dataset, the US-based de-identified Flatiron Health-Foundation Medicine Clinico-Genomic Database (CGDB), of 897 patients with metastatic or castrate-resistant prostate cancer between January 2011 and June 2020. Among these patients, they examined clinical characteristics and treatment selections.

Among the 11,741 patients in the Foundation Medicine cohort, 1,422 (12%) men were of African ancestry and 9,244 (79%) men were of European ancestry. The majority of men had localized disease and presented with adenocarcinoma.
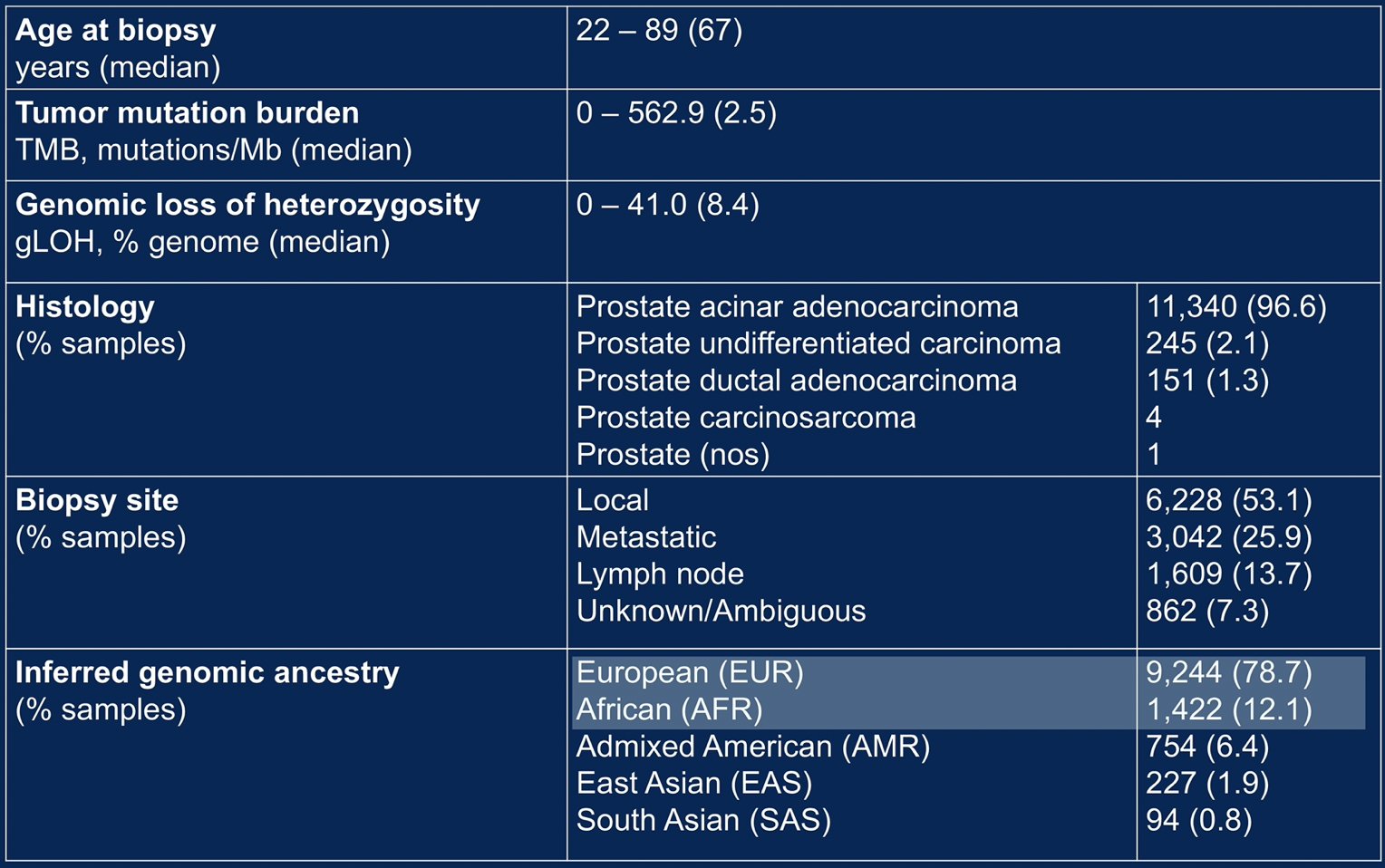
At the time of testing, men of African ancestry were younger than those of European ancestry (median age 64 vs. 67 years, p < 0.001). Additionally, the proportion of men diagnosed before age 50 years was higher among men of African Ancestry (4.2%) than men of European ancestry (2.5%).
The authors then assessed the tumor genomic landscape, stratified by patient SNP-inferred ancestry.
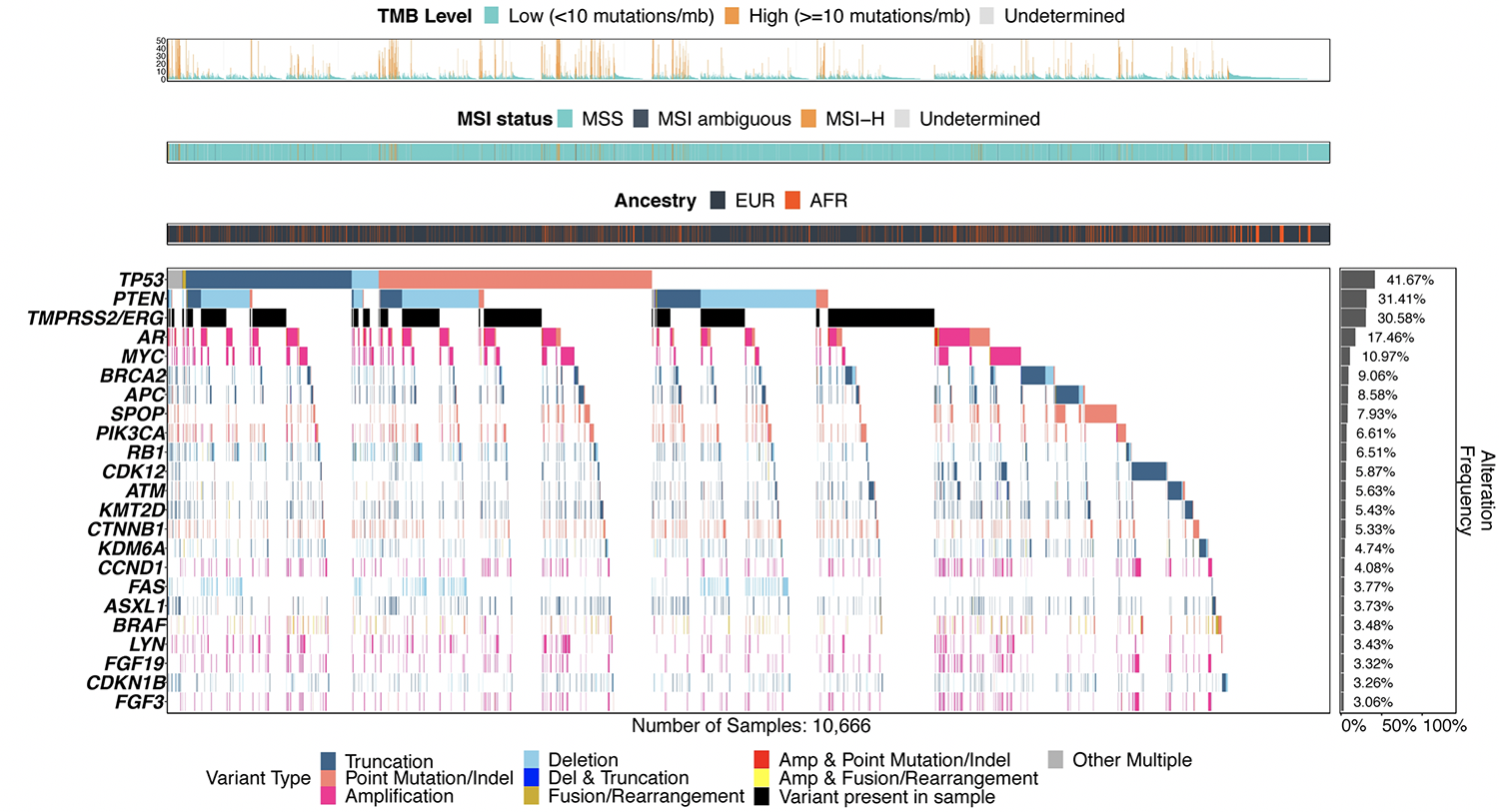
Notably, in terms of the tumor genomic landscape, TP53 and PTEN alterations and TMPRSS2-ERG rearrangements occurred less frequently in men of African ancestry than men of European ancestry (35% vs. 43%, 21% vs. 33%, 15% vs. 33% respectively, p < 0.05). Conversely, alterations in SPOP (11.9% vs. 7.3%), CDK12 (10.0% vs. 5.2%), CCND1 (6.0% vs. 3.8%), KMT2D (7.7% vs. 5.1%), HGF (4.1% vs. 2.5%), and MYC (13.4% vs. 10.6%) were more common in men of African ancestry than those of European ancestry (p < 0.05). Further, BRAF alterations were somewhat more common in men of African ancestry (5.0% vs. 3.2%, p < 0.05).
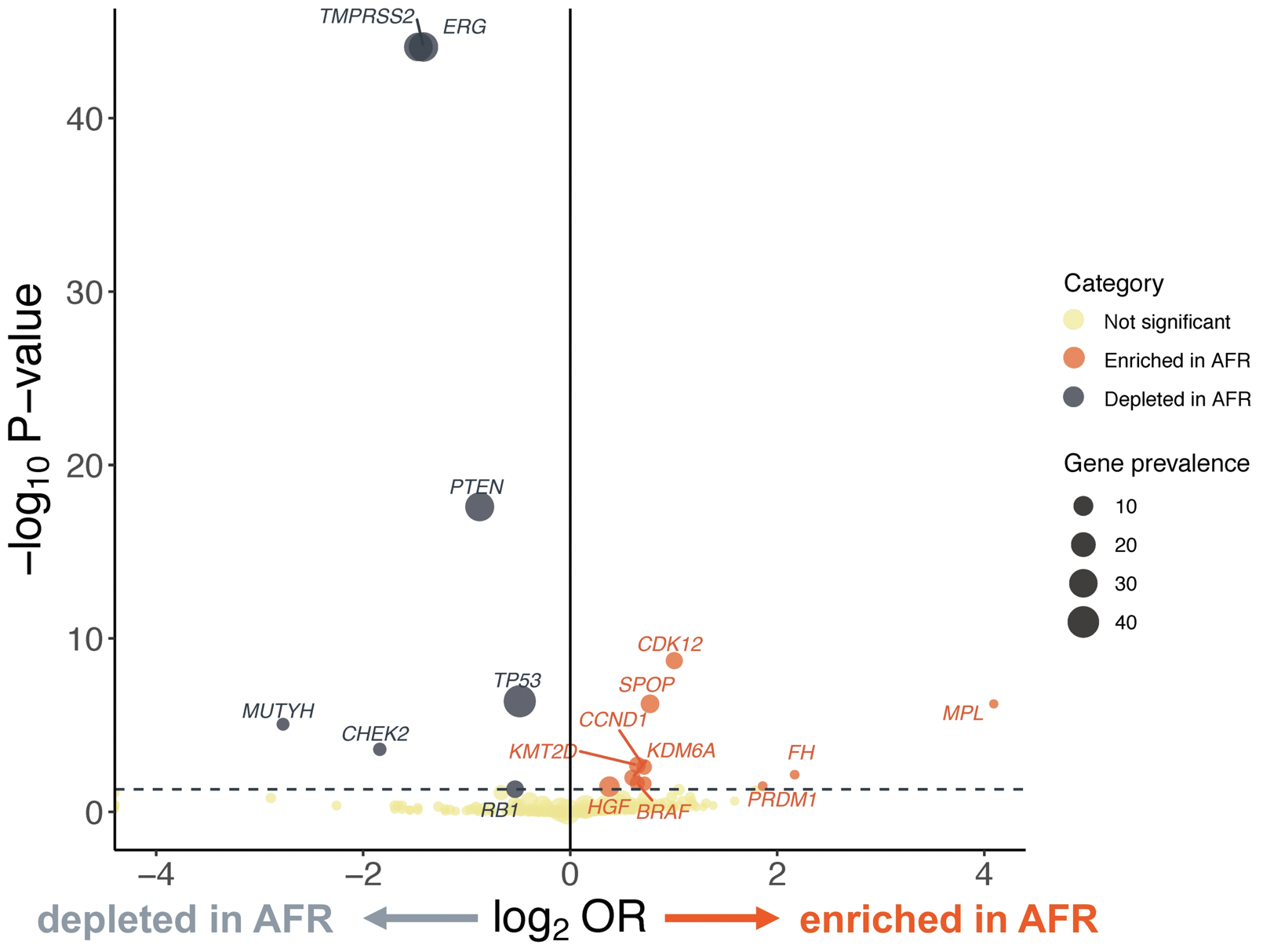
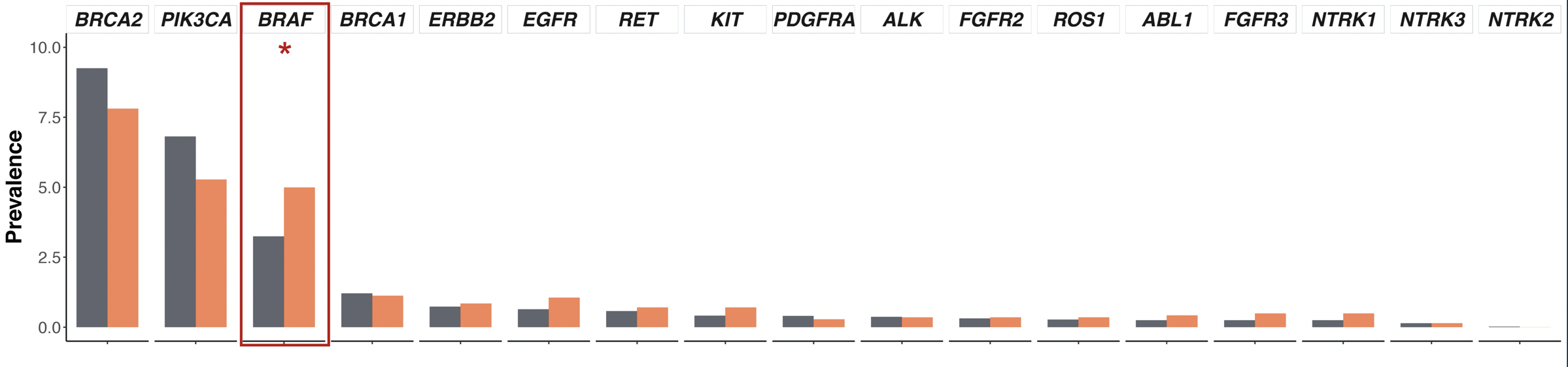
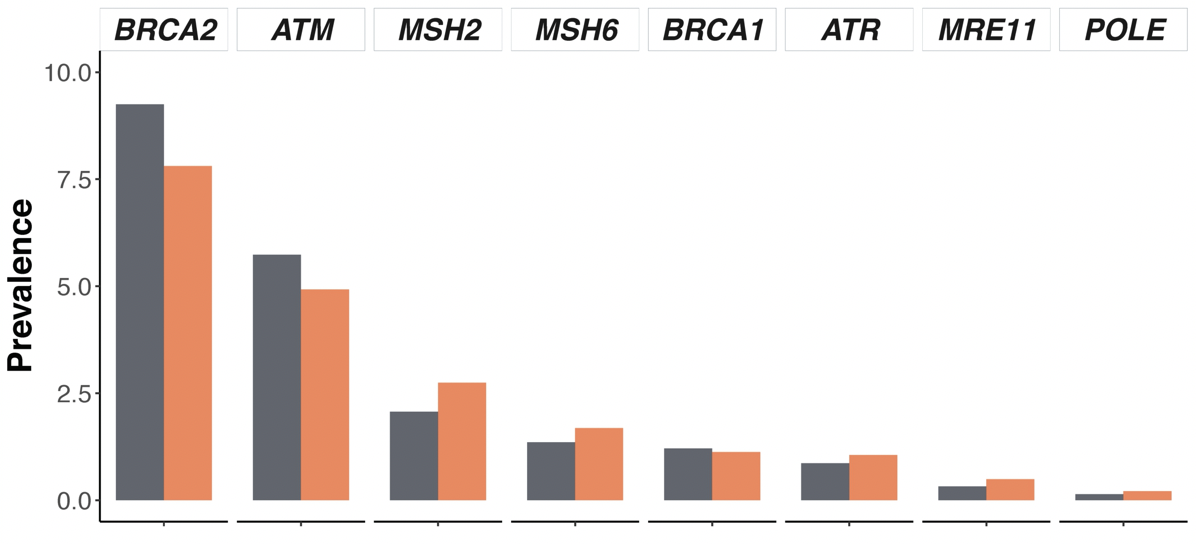
The authors did not find significant differences in alteration frequency for BRCA1/2, AR, DNA damage response pathway genes, and actionable genes with therapy implications between men of African ancestry than those of European ancestry.
Among patients in the Flatiron Health-Foundation Medicine Clinico-Genomic Database, the authors identified 79 men with African ancestry and 762 men with European ancestry. Notably, a substantially higher proportion of men of African ancestry were treated at community institutions (91%) compared to men of European ancestry (63%).
Notably, men of African ancestry received a median of 2 lines of therapy prior to undergoing comprehensive genomic profiling while men of European ancestry had received a single line of therapy. This is particularly relevant as previously used treatment lines may affect observed genomic landscapes.
Further, while, the proportion of patients receiving immunotherapy and PARPi was similar across ancestry, however men of African ancestry were less likely to receive clinical study drug compared with men of European ancestry (11% vs 30%, p < 0.001). This was true even among men with actionable alterations (1% vs 6%, p < 0.001).
The authors conclude that this large analysis demonstrated largely similar rates of actionable gene alterations for men of African and European ancestry. As a result, intrinsic biologic differences are unlikely to explain observed differences in racial outcomes for men with prostate cancer. Despite this, men of African ancestry underwent comprehensive genomic profiling later in their disease course and were less likely to be treated on clinical trials, highlighting differences in care allocation between racial groups. Thus, equitable use of genetic testing, clinical trial enrolment, and subsequent precision medicine may decrease observed racial disparities.
Presented by: Brandon Mahal, MD, Assistant Professor of Radiation Oncology, Assistant Director of Community Outreach and Engagement, Department of Radiation Oncology, University of Miami Health System, Coral Gables, FL.
Written By: Christopher J.D. Wallis, MD, Ph.D., Urologic Oncology Fellow, Vanderbilt University Medical Center, Twitter: @WallisCJD at the 2021 American Society of Clinical Oncology (ASCO) Annual Meeting #ASCO21, June, 4-8, 2021

