In addition to these treatments, it is recognized that both androgen-deprivation therapy for prostate cancer and metastatic disease itself is associated with risk of fracture, due to the osteoporotic events and skeletal-related events, respectively. Bone protective agents, with differing doses for these differing indications, may reduce both bone loss and skeletal-related events.
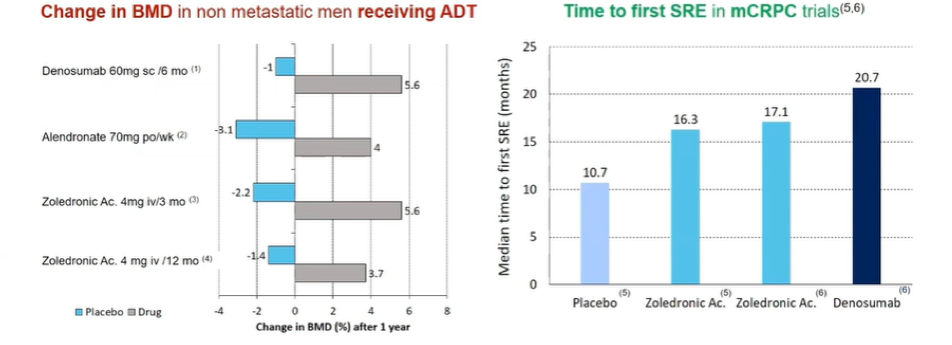
For patients treated with androgen deprivation therapy and an androgen receptor pathway inhibitor, up to 11% of patients may have fragility or osteoporotic fractures and up to 40% of patients may have disease-related skeletal-related events. As a result, guidelines routinely recommend bone protective agents.
As suggested by the success of monotherapy in mCRPC, there has been interest in combination therapies. The ERA-223 trial of abiraterone acetate and radium-223 was launched. However, the trial was unblinded prematurely due to a higher-than-expected rate of fractures and deaths with the combination approach.
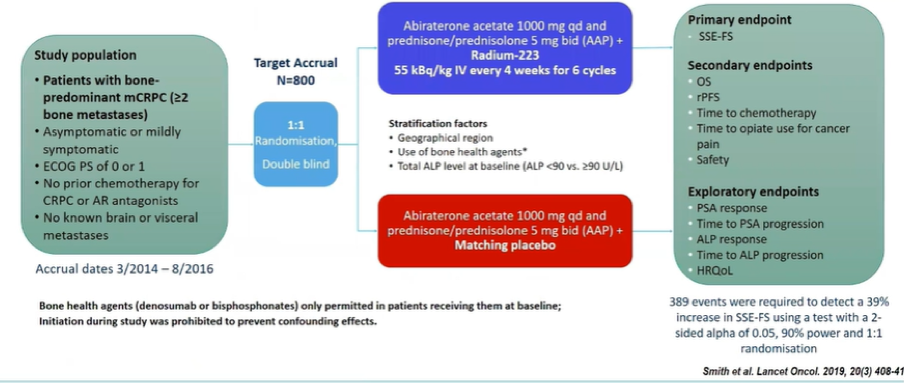
Additionally, in the ERA-223 trial, most fractures occurred at sites without evidence of metastatic disease, suggesting an osteoporotic mechanism. Further, use of bone protective agents at study entry was low in this cohort (40%). However, post hoc analyses suggested that bone protective agents reduced fractures in both the experimental and control arms.
In parallel to the ERA-223 trial, the randomized phase III EORTC-1333-GUCG (NCT02194842) trial compares enzalutamide vs. a combination of Radium 223 and enzalutamide in men with asymptomatic or mildly symptomatic metastatic castration resistant prostate cancer (mCRPC).

As noted above, ERA-223 (NCT02043678) was prematurely unblinded in November 2017 due to a significant increase in the rate of fractures in the combination of abiraterone and radium-223. This led to an amendment in the EORTC-1333-GUCG trial to mandate use of bone protecting agents (BPA).
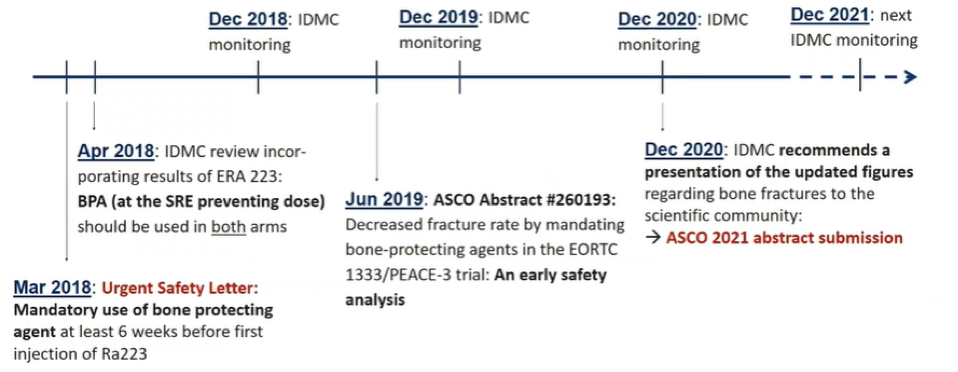
As skeletal fractures, pathological or not, are a frequent and underestimated adverse event of systemic treatment of advanced prostate cancer, the authors sought to assess whether this mandated use of BPA (zoledronic acid or denosumab) would mitigate the risk of fractures in this patient population was unclear. Building on preliminary data previously reported, in the Prostate, Testicular, and Penile Oral Abstract Session at the 2021 American Society of Clinical Oncology 2021 Annual Meeting held on Tuesday, June 8th, 2021, Dr. Gillessen presented updated safety results from the EORTC 1333 / PEACE III trial of radium-223 and enzalutamide, specifically examining the effect of mandated bone protective agents on fracture risk with longer follow-up.
As of the data cut-off of January 28, 2021, the EORTC-1333-GUCG trial had enrolled 253 patients in total of whom 134 were enrolled after BPA were mandated and 119 who were enrolled prior. Patients were randomized as planned to enzalutamide + radium-223 or enzalutamide alone.
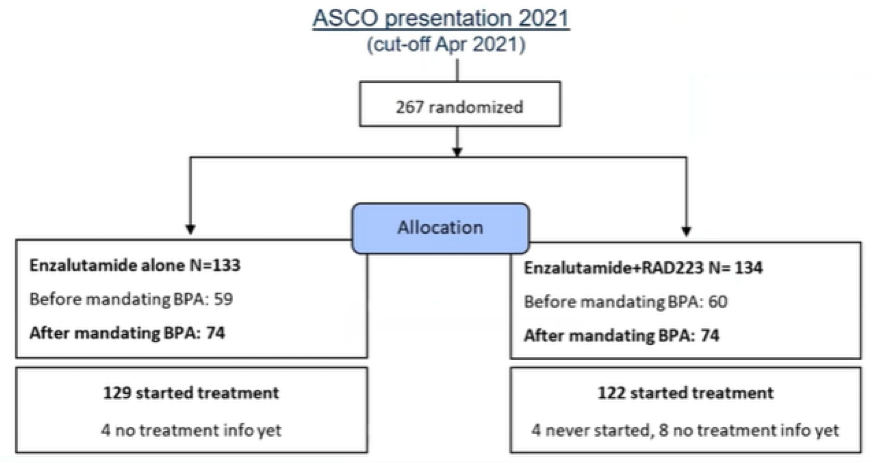
In this analysis focusing on skeletal safety, the authors estimated the
fracture rate using the cumulative incidence method in the safety population of 237 (122 after making BPA mandatory) treated patients. The authors used competing risk models utilizing death in absence of fracture as competing event with time-to-event models using censoring at last follow-up.
Utilization of BPA was significantly increased by the amendment: among those in the combination enzalutamide + radium-223 arm, overall use of 69.5% and use after the amendment was 95.2%. Similarly, overall use in the enzalutamide arm was 73.1% with 95% of patient enrolled after the amendment receiving BPA. Notably, 13.6% of those in the combined arm and 21.8% in the enzalutamide alone arm were not receiving BPA at enrollment but initiated treatment during the on-protocol treatment with 55.9% and 51.3%, respectively, receiving BPA since study entry.
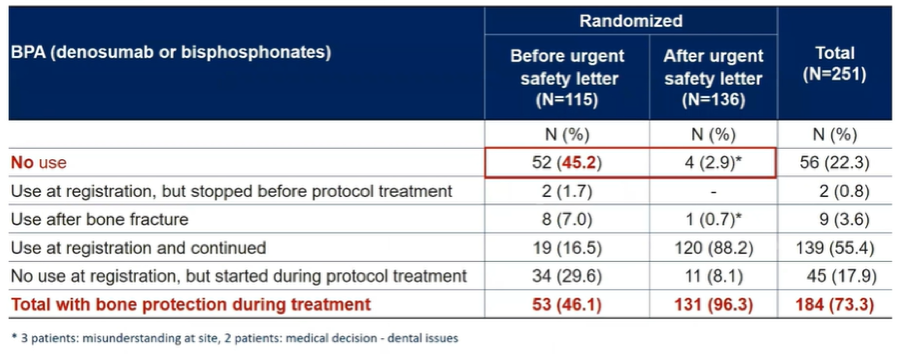
With a median follow-up of 36.7 months in patients prior to the amendment mandating BPA and 23.1 months among those patients receiving BPA, a total of 39 patients reported a fracture. The vast majority of these events (30 patients: 20 in the enzalutamide + radium-223 and 10 in the enzalutamide only arm) occurring in patients not receiving BPA while 9 events occurred on those receiving BPA (4 of which occurred in patients receiving enzalutamide and radium-223).
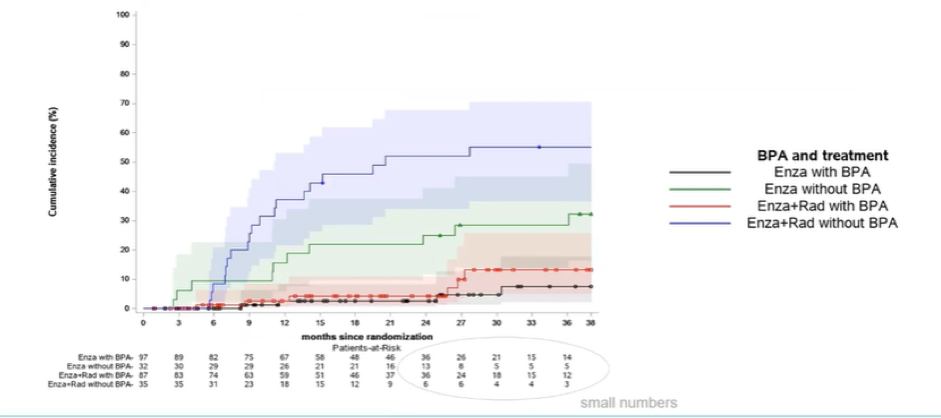
The cumulative incidence of fracture was similar among patients who received BPA (with a numerically lower incidence among those receiving the combination of enzalutamide and radium-223) while, among those who did not receive BPA, rates of fractures were higher in patients receiving the combination of enzalutamide.
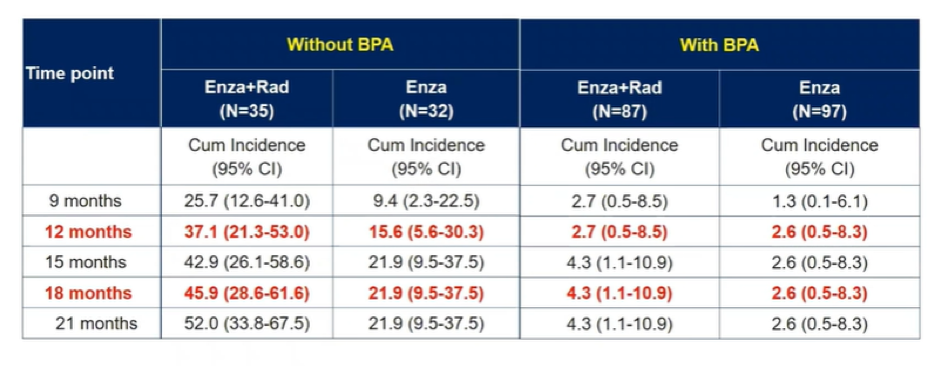
This updated analysis of the EORTC-1333-GUCG trial demonstrates evidence of effect modification for the risk of fracture with the combination of enzalutamide and radium-223: without BPA, this risk in increased while, with BPA, the cumulative incidence is very low for patients treated with either combination therapy or enzalutamide alone. This analysis further emphasizes the importance of administering bone protective agents to prevent skeletal complications.
Presented by: Silke Gillessen, MD, senior consultant in the Medical Oncology-Hematology Department at the Kantonsspital St. Gallen, St Gallen, Switzerland and Co-founder of the Advanced Prostate Cancer Consensus Conference (APCCC), previously of the University of Manchester and the NHS Christie Trust, Manchester, United Kingdom
Written by: Christopher J.D. Wallis, Urologic Oncology Fellow, Vanderbilt University Medical Center Contact: @WallisCJD on Twitter at the 2021 American Society of Clinical Oncology (ASCO) Annual Meeting, Virtual Annual Meeting #ASCO21, June, 4-8, 2021


