While these agents have proven beneficial in clinical trials, improvement in population-level outcomes depends on their widespread adoption. In the Prostate, Testicular, Penile Poster session at the American Society of Clinical Oncology (ASCO) 2021 Annual Meeting, Dr. Daniel George and colleagues assessed the real-world utilization of effective combination therapies in the first-line treatment of mCSPC in oncology practice.
To do so, the authors utilized the ConcertAI Oncology Dataset to examine electronic medical record (EMR) data from a network of U.S.-based oncology practices. They identified patients with mCSPC who began first-line therapy between 2014–2019. Notably, they excluded the use of first-generation anti-androgen where these were utilized for less than 90 days. They further examined the proportion of mCSPC patients receiving each first-line regimen, as well as characterizing the duration of therapy until initiation of next regimen and trends in first-line regimen utilization. They further considered treatment patterns according to race/ethnicity.
The authors identified 858 men with mCSPC, of whom 70% were White, 16% were Black, 3% were Hispanic, and 11% had unknown or other race categorization. The median age at diagnosis of mCSPC was 69 years, and 63% presented with de novo metastases.

Examining treatment practice patterns, the most common first-line treatment approaches in mCSPC were ADT + first-generation anti-androgen (26.3%, mainly bicalutamide), ADT monotherapy (20.5%), ADT + novel hormonal therapy (with or without first-generation anti-androgen) (19.2%), and ADT + docetaxel (with or without first-generation anti-androgen) (16.4%). Novel hormonal therapy included abiraterone, apalutamide, and enzalutamide. The remaining patient received a variety of treatment approaches including first-generation anti-androgen monotherapy (5.9%) or novel hormonal therapy (with or without first-generation anti-androgen) (5.5%).
Assessing trends over time, the authors observed increasing utilization of the combination of ADT with novel hormonal therapy. In contrast, utilization of ADT with docetaxel increased from 2014 to 2017 and then subsequently declined, with the increasing utilization of ADT + novel hormonal therapy. In contrast, while ADT + first-generation anti-androgen was the most commonly used approach in 2014, its utilization progressively decreased over time.
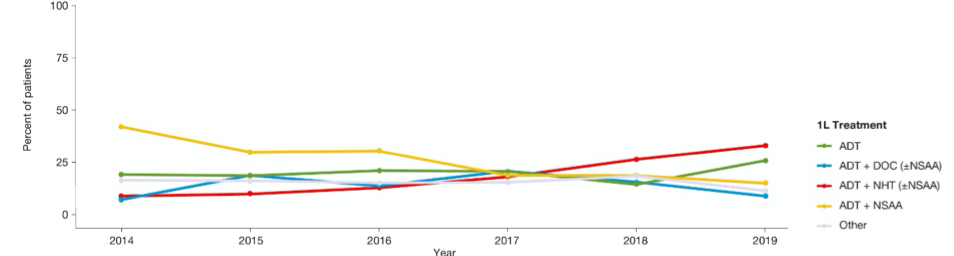
The authors found no statistically significant differences in first-line treatment patterns in mCSPC between White and Black patients.
Finally, the authors assessed the duration of treatment, defined from the start of first-line therapy to the initiation of a subsequent regime. The median duration was 14.3 months for ADT + novel hormonal therapy (with or without first-generation anti-androgen) and 10.8 months for ADT + docetaxel (with or without first-generation anti-androgen).
These authors conclude that, even in 2019, the majority of patients with mCSPC treated in real-world settings do not receive intensified treatment regimes that have been prove to improve overall survival in randomized controlled trials. Further, those that did receive these treatments had shorter durations of treatment than were seen in registration trials.
Presented by: Daniel J. George, MD, Professor of Medicine Professor in Surgery Member of the Duke Cancer Institute
Written by: Christopher J.D. Wallis, Urologic Oncology Fellow, Vanderbilt University Medical Center Contact: @WallisCJD on Twitter at the 2021 American Society of Clinical Oncology (ASCO) Annual Meeting, Virtual Annual Meeting #ASCO21, June, 4-8, 2021

