(UroToday.com) The 2021 American Society of Clinical Oncology (ASCO) program included a session discussing the application of genomic testing treatments for patients with prostate cancer, including a presentation by Dr. Veda Giri discussing genomic medicine in prostate trials and treatments.
In terms of the historical context of genetic testing, this dates back to the late 1990s with the utilization of BRCA testing for breast and ovarian cancer, followed by Lynch Syndrome testing in the mid-2000s, ushering in the era of risk assessment and focused genetic screening. More recently, over the last several years, prostate cancer genetic testing has become more commonplace offering multigene testing and precision medicine. The evolution of prostate cancer germline testing has revolved around several important clinical trials, including TOPARP-A (in 2015), GALAHAD, TRITON2, and PROfound, leading to FDA approval of rucaparib and olaparib in 2020. Multiple novel genomic tests are under development or early clinical use, including RNA testing, transcriptome profiling, etc. As follows is a summary of the current genomic tests: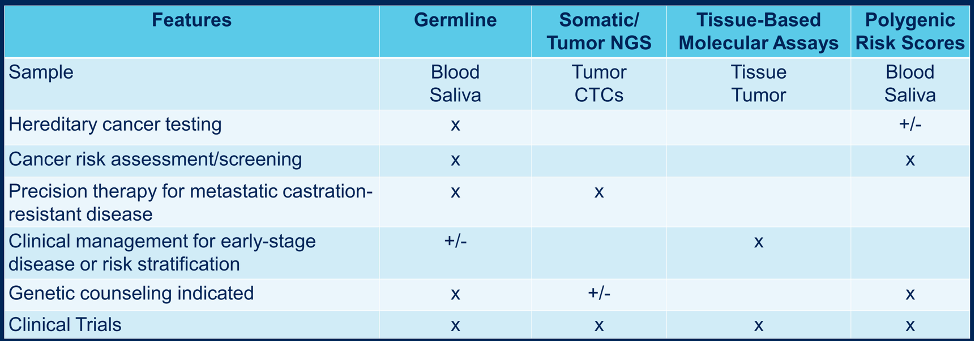
The use of PARP inhibitors capitalizes on the concept of synthetic lethality, with cell death resulting in the setting of HRR deficiency in combination with PARP inhibition. Pivotal clinical trials of PARP inhibitors in mCRPC are summarized as follows1: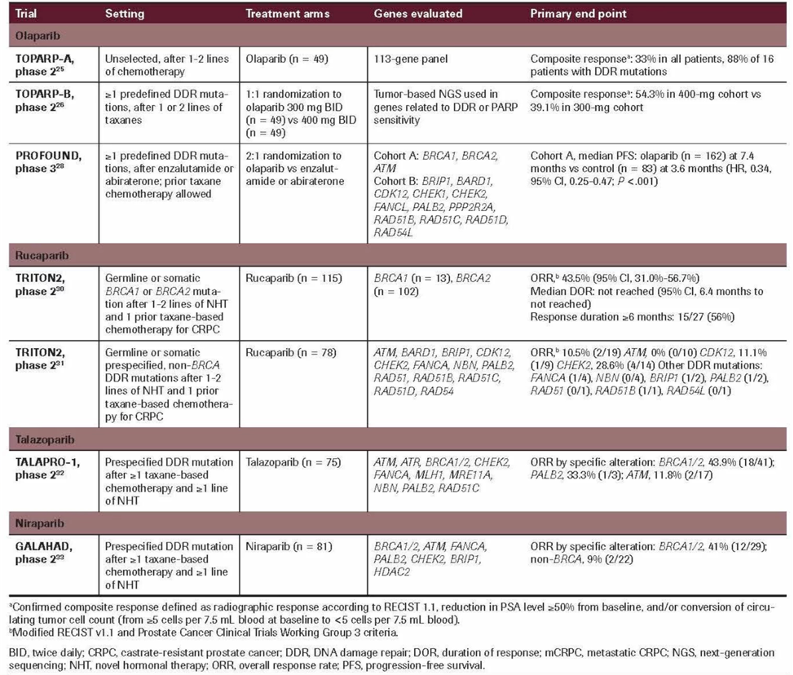
The TRITON2 trial assessed rucaparib 600 mg BID in patients with mCRPC associated homologous recombination repair gene alterations2. For the patients with BRCA1/2 alteration, there was a 44% confirmed overall response rate and 51% confirmed PSA response rate while patients harboring an ATM and CDK12 alteration did not receive significant benefit. The median rPFS among 115 BRCA carriers was 9.0 months (95% CI 8.3-13.5). Based on this data, the FDA granted accelerated approval to rucaparib for men with BRCA-mutated metastatic CRPC (germline or somatic) who had prior treatment with androgen-receptor-directed therapy and prior taxane-based chemotherapy.
The PROfound study recruited men with mCRPC who had progressed on previous abiraterone acetate or enzalutamide administered at the time of non-metastatic castrate-resistant prostate cancer or at the time of metastatic castrate-sensitive prostate cancer3. Alterations were identified in one of 15 pre-specified genes involved in homologous recombination repair (BRCA 1/2, ATM, BRIP1, BARD1, CDK12, CHEK 1/2, FANCL, PALB2, PPP2R2A, RAD51B, RAD51C, RAD51D, RAD54L). Cohort A had alterations in BRCA1, BRCA2, or ATM while Cohort B had alterations in any of the other 15 included genes. In both cohorts, patients were randomized 2:1 to olaparib vs. abiraterone or enzalutamide. The primary analysis was based on imaging-based progression-free survival among patients in Cohort A. In an assessment of the primary outcome, there was a significantly improved progression-free survival in patients with mutations of BRCA1, BRCA2, or ATM (HR 0.34, 95% CI 0.25 to 0.47). Similar results were seen in the combined cohort (HR 0.49, 95% CI 0.38 to 0.63). Based on the results of the PROfound trial, the FDA granted approval for olaparib for deleterious or suspected deleterious germline or somatic homologous recombination repair (HRR) gene-mutated mCRPC that progressed with abiraterone or enzalutamide.
The current NCCN guidelines recommend germline testing for prostate cancer among those with:
• Metastatic prostate cancer
• Ashkenazi Jewish ancestry
• Intraductal/cribriform histology
• Very high-risk disease: T3b-T4 of primary pattern 5 or >4 cores with grade group 4 or 5
• High-risk disease: T3a or grade group 4 or 5 with PSA > 20 ng/mL
• Family history of high-risk germline mutation (pathogenic/likely pathogenic variant)
As follows is a global comparison of prostate cancer germline testing guidelines and statements:
The Philadelphia Prostate Cancer Consensus Conference held in 2019 provides several recommendations for developing a genetic implementation framework, of which Dr. Giri was the lead author4. First, large germline panels and somatic testing were recommended for patients with metastatic prostate cancer. Furthermore, metastatic disease or family history suggestive of hereditary prostate cancer was recommended for germline testing. Priority genes to test for metastatic disease treatment included BRCA2, BRCA1, and mismatch repair genes, with broader testing, such as ATM, for clinical trial eligibility. Additionally, testing for BRCA2 was recommended for men considering active surveillance of low-risk disease. Lastly, screening starting at age 40 years or 10 years before the youngest PCA diagnosis in a family was recommended for BRCA2 carriers, with consideration in HOXB13, BRCA1, ATM, and mismatch repair carriers. As follows is a comprehensive implementation framework for prostate cancer genetic evaluation and management:
Dr. Giri concluded by noting that clinical trials across the entire spectrum of prostate cancer (early stage/low-risk disease, locally advanced/high-risk disease, hormone-sensitive and castration-resistant non-metastatic disease, and hormone-sensitive and castration-resistant metastatic disease) are ongoing, all aiming to improve precision therapy and management.
Presented By: Veda N. Giri, MD, Departments of Medical Oncology, Cancer Biology, and Urology, Cancer Risk Assessment and Clinical Cancer Genetics Program, Sidney Kimmel Cancer Center, Thomas Jefferson University
Written By: Zachary Klaassen, MD, MSc – Urologic Oncologist, Assistant Professor of Urology, Georgia Cancer Center, Augusta University/Medical College of Georgia Twitter: @zklaassen_md at the 2021 American Society of Clinical Oncology (ASCO) Annual Meeting, Virtual Annual Meeting #ASCO21, June, 4-8, 2021
References:
- Gong J, Posadas E, Bhowmick N, et al. Integrating PARP inhibitors into advanced prostate cancer therapeutics. Oncology. 2021 Mar 15;35(3):119-125.
- Abida W, Patnaik A, Campbell D, et al. Rucaparib in Men with Metastatic Castration-Resistant Prostate Cancer Harboring a BRCA1 or BRCA2 Gene Alteration. J Clin Oncol 2020 Nov 10;38(32):3763-3772.
- de Bono J, Mateo J, Fizazi K, et al. Olaparib for Metastatic Castration-Resistant Prostate Cancer. N Engl J Med 2020 May 28;382(22):2091-2102.
- Giri VN, Knudsen KE, Kelly WK, et al. Implementation of Germline Testing for Prostate Cancer: Philadelphia Prostate Cancer Consensus Conference 2019. J Clin Oncol. 2020 Aug 20;38(24):2798-2811.
Related Content:
Implementation of Germline Testing for Prostate Cancer - Veda Giri


