Case: 62F presents with gross hematuria and undergoes cystoscopy.
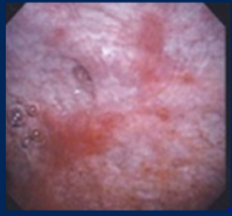
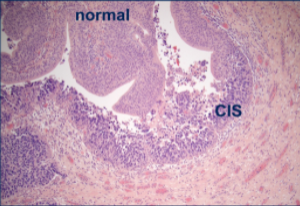
Dr. Lee discussed the cystoscopy findings as a couple of areas of erythema that appear released. In this patient, she would be initially concerned for carcinoma in situ. In the pathology slide, we have labeled carcinoma in situ (CIS). This is an important diagnosis to make earlier because we think it is a precursor to invasive disease, and so I would be interested in talking with her about intravesicular therapy. We still look to intravesical BCG for our first-line treatment, roughly 2/3 of patients will respond initially, and about ½ of patients will have a long-term benefit. After induction BCG, we know that maintenance therapy reduces the risk of recurrence and disease progression. Most urologists follow the SWOG schedule for high-risk patients (such as patients with carcinoma in situ), which provides a course of induction BCG with six doses and then maintenance therapy at 3, 6, 12, 18, 24, 30, and 36 months post-induction so long as the patient tolerates therapy and has no recurrence.
Case continued: She is diagnosed with carcinoma in situ and receives intravesicular BCG for six treatments followed by maintenance therapy. After six months of maintenance therapy, her surveillance cystoscopy reveals recurrence carcinoma and situ and a new invasive T1 urothelial carcinoma.
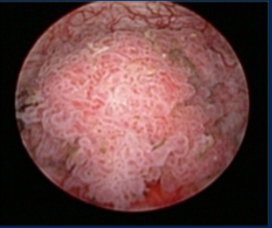

Dr. Lee commented that this appears to be a more concerning tumor based on size with neovascularity around the edges and erythema at the 6 o’clock position. Based on the appearance with some suggestion of papillation, she would worry about an invasive component, and so would suggest transurethral resection of the bladder tumor (TURBT), examination under anesthesia, and upper tract imaging. The degree of invasion seen on the pathology slide (more extensive, not focal) is concerning for an even higher risk of future prediction.
The first question posed was “What treatment option would you proceed with?”. (A) Intravesical chemotherapy, (B) Intravenous pembrolizumab, (C) Radical Cystectomy, or (D) Other.
Dr. Lee discussed that based on her early timing of relapse, if the patient has reasonable performance status, then she would be talking about radical cystectomy. Not everyone will agree with this and many will remain committed to bladder preservation at this point. If she does not agree to cystectomy then I think about second-line therapies including pembrolizumab or intravesical chemotherapy. More and more provocative data is emerging from studies looking at gemcitabine and docetaxel sequentially with intravesical chemotherapy, which in recent data is well-tolerated and showing 50% recurrence-free survival at two years. Though this is retrospective data, it is provocative and likely warrants a clinical trial.
I would pursue re-resection here if the patient does not opt for cystectomy to take a deeper resection.
The patient is not interested in radical cystectomy or intravenous pembrolizumab due to travel requirements. She is willing to receive intravesical chemotherapy with gemcitabine and docetaxel.
Dr. Lee commented that for a long time valrubicin was the only FDA-approved option for intravesical chemotherapy in BCG-unresponsive patients. Unfortunately, this was not durable, as less than 10% of patients were free of disease after two years. Also, for this patient who has travel concerns, it is possible to think of every 6-week infusion schedule for pembrolizumab. However, it is important to monitor these patients on pembrolizumab with labs, so in her practice, she typically throws in a telehealth visit. She also discussed the potential role of nadofaragene firadenovec, which is a non-replicating adenoviral vector that induced interferon alfa-2B destruction of tumors. It’s delivered every three months and not yet approved but appears promising. She also discussed the potential emerging role of oportuzumab monatox (single-chain monoclonal antibody fragment against EpCAM conjugated to a truncated form of Pseudomonas exotoxin that inhibits protein synthesis in target cells), and FGFR inhibitors in BCG-unresponsive disease.
After intravesical chemotherapy, surveillance cystoscopy performed 6 months later reveals invasive disease obscuring the bilateral ureteral orifices, resulting in kidney injury. CT imaging reveals bilateral hydronephrosis but no significant lymphadenopathy.
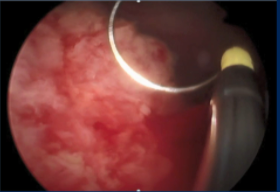
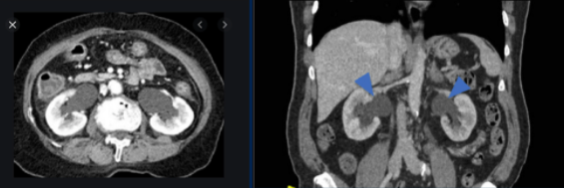
Dr. Lee discussed that this bladder view, which appears to be in the operating room, is very concerning for recurrence which encompasses most of the visual field and appears more nodular than papillary. The presence of bilateral hydronephrosis and is suggestive of extravesicular disease and potentially vaginal invasion. An exam under anesthesia will be important to fully stage the patient. Given the likelihood that the disease is obstructing both ureteral orifices, it would likely be difficult to place internalized stents, so she would probably opt for percutaneous nephrostomy tubes in this patient to improve her renal function. It is important to remember that patients are never excited about external tubes and drains that they have to manage as it impacts their quality of life.
Bilateral percutaneous nephrostomy tubes are recommended, and the patient wants to start treatment as soon as possible. Her ECOG PS is 1 and she has no other major comorbidities. What options do you consider? (A) Radical cystectomy, (B) Split-dose cisplatin-combination chemotherapy followed by cystectomy, (C) carboplatin-combination chemotherapy followed by cystectomy, (D) pembrolizumab followed by cystectomy, (E) Bladder preservation with cisplatin-based chemoradiation, (F) Bladder preservation with 5FU based chemoradiation.
Dr. Lee discussed the promising data from RTOG 0712 exploring the role of trimodality therapy for bladder preservation. However, patients were not recruited to this trial if they had prognostic factors that suggest they might fail, and so it is important to consider what the most efficacious therapy might be in the context of whatever prognostic factors are present.
Although primary cystectomy is possible, we have level 1 evidence to show improved survival outcomes with neoadjuvant cisplatin-based chemotherapy as well as provocative early data showing reasonable pT0 rates in neoadjuvant pembrolizumab. Her preference would be to avoid primary cystectomy unless no neoadjuvant therapy can be administered.
With bilateral nephrostomy tubes, the creatinine clearance improves to 61 ml/min and becomes cisplatin eligible. She prefers surgery but would be willing to receive neoadjuvant chemotherapy if recommended. The patient undergoes accelerated MVAC and then radical cystectomy with an ileal conduit. Pathology reveals ypT2N0 disease.
The goal of neoadjuvant therapy, dating back to the SWOG-8710 trial, is pathologic complete response (pCR). Patients who had pCR had overall survival improvement, with 85% of patients surviving at 5 years, which was very impressive. We know survival outcomes for patients who do not achieve pCR are worse, and also retrospective data showing that patients with the residual muscle-invasive disease do worse than those with residual non-muscle invasive disease.
What course of action would you recommend next? (A) Adjuvant carboplatin with gemcitabine, (B) Adjuvant taxane-combination chemotherapy regimen, (C) Adjuvant checkpoint inhibitor, (D) Observation
The most effective context for adjuvant therapy appears to be in patients with an extravesical component of residual disease. With an organ-confined state after neoadjuvant chemotherapy and surgery, I might favor observation.
The patient undergoes observation. Six months later, surveillance CT imaging reveals the following image. Creatinine clearance is now 48 ml/min and ECOG performance status is 1. Would you perform PD-L1 IHC on archival tissue to aid in treatment decision-making?
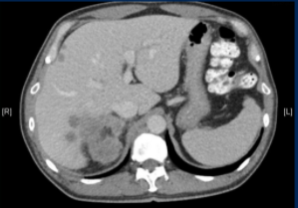
The remainder of the session involved input from medical oncology as the patient developed the systemic disease. The take-home points below were provided at the end.

Presented by: Cheryl T. Lee, MD, Urologic Oncologist and Chair of the Department of Urology at the Ohio State University, Colombus, OH
Written by: Alok Tewari, MD, Ph.D., Medical Oncologist at the Dana-Farber Cancer Institute, at the virtual 2021 American Society of Clinical Oncology Annual Meeting Congress (#ASCO21), June 4th-June 8th, 2021


