(UroToday.com) The bladder cancer poster discussion session at American Society of Clinical Oncology (ASCO) 2021 featured a discussant presentation by Dr. Bishoy Faltas examining novel immunotherapy approaches across the spectrum of urothelial cancer stages.
Dr. Faltas notes that immunotherapy is expanding into new clinical spaces in urothelial carcinoma treatment, including neoadjuvant immunotherapy. The rationale for neoadjuvant systemic therapy is that genomic data suggest that subclinical micro-metastatic clones already spread at the time of initial presentation. There are advantages of neoadjuvant systemic therapy, including (i) neoadjuvant cisplatin-based chemotherapy improves OS, (ii) it is often well tolerated, (iii) there is a potential for maximizing impact on patient outcomes by administering drug at the earliest point in the natural history of the disease, (iv) tissue availability from TURBT and radical cystectomy offers opportunities to study biomarkers of response in clinical trials, and (v) surrogate endpoints of responsiveness to therapy enable early risk-stratification to select patients who could benefit from additional therapy. At ASCO 2021, Funt et al. presented “Neoadjuvant atezolizumab with gemcitabine and cisplatin in patients with muscle-invasive bladder cancer: A multicenter, single-arm, phase 2 trial”

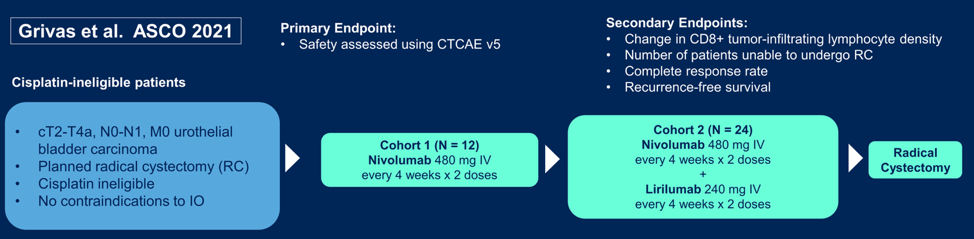
and Grivas et al. presented “PrE0807: A phase Ib feasibility trial of neoadjuvant nivolumab without or with lirilumab in cisplatin-ineligible patients with muscle-invasive bladder cancer”
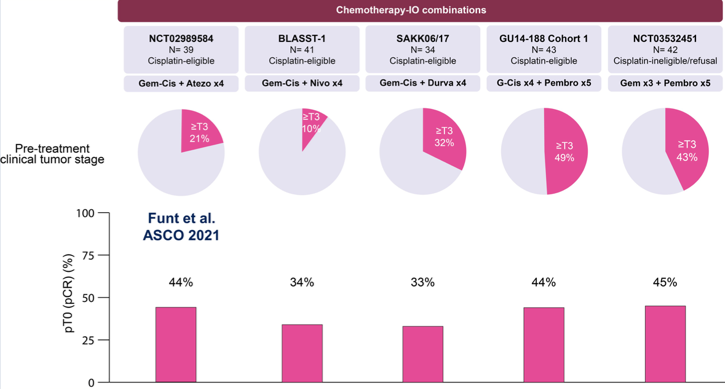
There is a precedent for immunotherapy-chemotherapy neoadjuvant combinations for muscle-invasive bladder cancer, including gemcitabine-cisplatin + nivolumab x 4, gemcitabine-cisplatin + durvalumab x 4, gemcitabine-cisplatin + pembrolizumab x 5, and gemcitabine x 3 + pembrolizumab x 5. pT0 rates range from 33%-45%, with the Funt et al. trial of gemcitabine-cisplatin + atezolizumab x 4 reporting a pT0 rate of 44%:
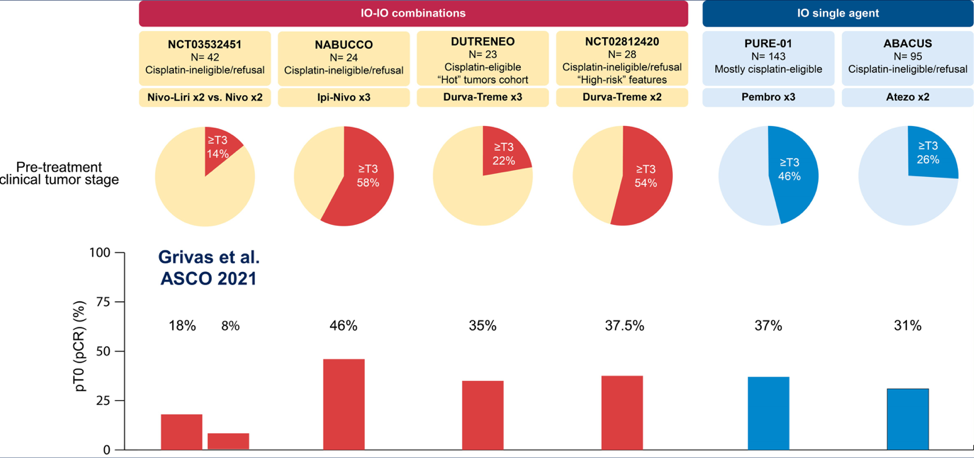
Furthermore, there is context for neoadjuvant immunotherapy single agent and combination therapy for muscle-invasive bladder cancer, including the PURE-01 and ABACUS trials (IO single-agent trials), and the NABUCCO, DUTRENEO, and NCT02812420 (durvalumab/tremelimumab x 2) trials (IO-IO combination trials). The Grivas et al. nivolumab + lirilumab trial is the latest in the IO-IO combination neoadjuvant trials:
There are several ongoing phase 3 trials in this disease space, including (i) NCT04209114 (nivolumab + NKTR214 vs nivolumab vs cystectomy), (ii) KEYNOTE-905/EV-303, (iii) ENERGIZE, (iv) NIAGARA, and (v) KEYNOTE-866.
Dr. Faltas notes that there are several important unanswered questions for perioperative immunotherapy:
- What is the optimal schedule, duration and time to cystectomy with these regimens?
- What are the most accurate surrogate clinical endpoints to predict OS?
- Are IO + chemotherapy, IO + IO, IO + ADCs more effective than single agents in RCTs?
- Can we select patients who are likely to benefit from neoadjuvant immunotherapy based on biomarkers?
- What is the best sequence of neoadjuvant, adjuvant and maintenance lines of therapy?
Dr. Faltas then discussed maintenance immunotherapy in the context of immunotherapy expanding into new clinical spaces in urothelial carcinoma treatment. At ASCO 2021, Dr. Tom Powles presented “Avelumab first-line (1L) maintenance for advanced urothelial carcinoma: Analysis of clinical and genomic subgroups from the JAVELIN Bladder 100 trial” a subgroup analysis of the transformative JAVELIN Bladder 100 trial that showed avelumab first-line maintenance + best supportive care significantly prolonging overall survival versus best supportive care alone in patients with advanced urothelial carcinoma that had not progressed on first-line platinum-based chemotherapy (HR 0.69, 95% CI 0.56 to 0.86; 1-sided p = 0.0005):1

Based on Dr. Powles presentation, Dr. Faltas notes that the efficacy of avelumab is consistent across the majority of these subgroups and as such, maintenance avelumab should be offered to the majority of patients in this disease space. There have been several molecular subtypes of urothelial carcinoma as potential biomarkers of IO clinical benefit, including the classification from UNC, MD Anderson, LUND, TCGA, and the Consensus group:
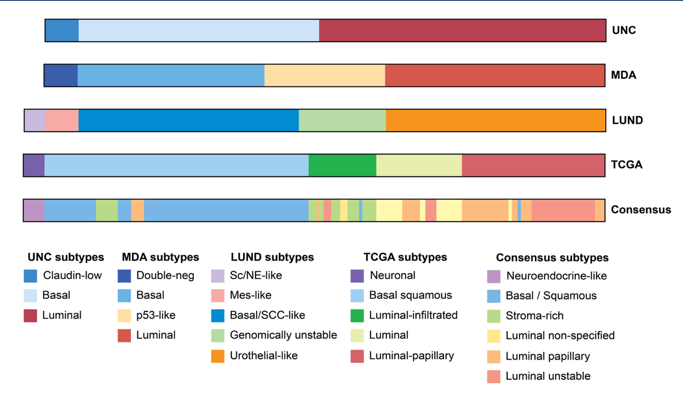
Dr. Faltas then highlighted the molecular subtypes deriving the highest clinical benefit:
- IMvigor-210 (atezolizumab in platinum resistant metastatic urothelial carcinoma, k=4): TCGA v1 subtype II → luminal-infiltrated subtype
- CheckMate 275 (nivolumab in platinum-resistant metastatic urothelial carcinoma, k=4): TCGA v1 subtype III → basal subtype
- JAVELIN Bladder 100 (avelumab maintenance in metastatic urothelial carcinoma, k=5): TCGA v2 subtype → basal subtype
- IMvigor-210 (atezolizumab in platinum resistant metastatic urothelial carcinoma, k=5): TCGA v2 subtype → neuroendocrine subtype
Dr. Faltas notes that the muscle-invasive bladder cancer molecular subtypes and immunotherapy benefit should be viewed as a continuum, however it is important to note that there is classifier-dependent variability among luminal subtypes:
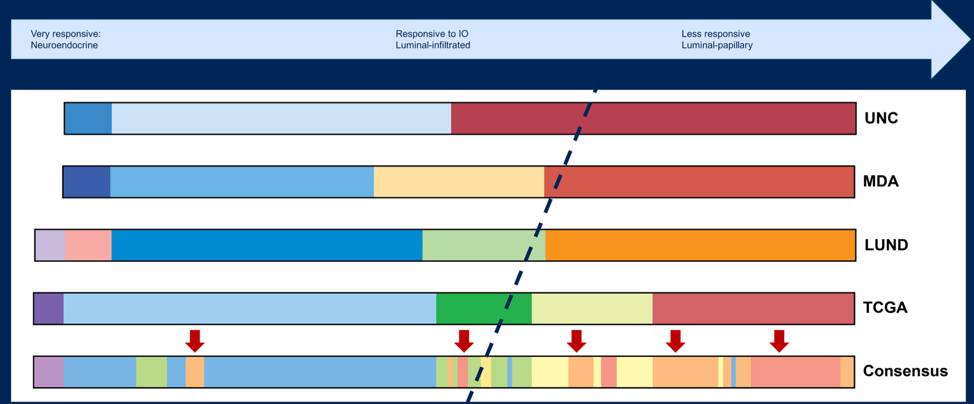
Dr. Faltas’ group has previously shown that luminal-papillary and T-cell depleted phenotype in upper tract urothelial carcinoma is less responsive to immunotherapy, which fits the narrative for lower tract disease, given that the subgroup analysis in Dr. Powles trial did not show a significant benefit of maintenance avelumab for upper tract urothelial carcinoma.
Dr. Faltas highlights that there are several additional challenges that persist:
- Molecular subtype membership switching over the natural history of the disease
- Bulk RNAseq-derived subtypes are byproducts of cancer and stroma cell types
- Subtype plasticity at the single-cell level
Dr. Faltas concluded his presentation with the following take-home messages:
- Immunotherapy is making inroads into the neoadjuvant and maintenance settings
- Neoadjuvant immunotherapy approaches are promising for cisplatin-ineligible patients
- Neoadjuvant immunotherapy combinations are safe and efficacy signals are promising
- Most clinical and molecular subtypes derive clinical benefit from maintenance avelumab
- Molecular subtyping offers a framework for understanding urothelial carcinoma biology
- Challenges for biomarker development require addressing knowledge gaps in urothelial carcinoma biology
Presented by: Bishoy M. Faltas, MD, Director of Bladder Cancer Research, Englander Institute for Precision Medicine, Weill Cornell Medicine, New York City, New York
Written by: Zachary Klaassen, MD, MSc – Urologic Oncologist, Assistant Professor of Urology, Georgia Cancer Center, Augusta University/Medical College of Georgia Twitter: @zklaassen_md at the 2021 American Society of Clinical Oncology (ASCO) Annual Meeting, Virtual Annual Meeting #ASCO21, June, 4-8, 2021
References:
1. Powles T, Park SH, Voog E, et al. Avelumab Maintenance Therapy for Advanced or Metastatic Urothelial Carcinoma. N Engl J Med 2020 Sept 24;383(13):1218-1230.

