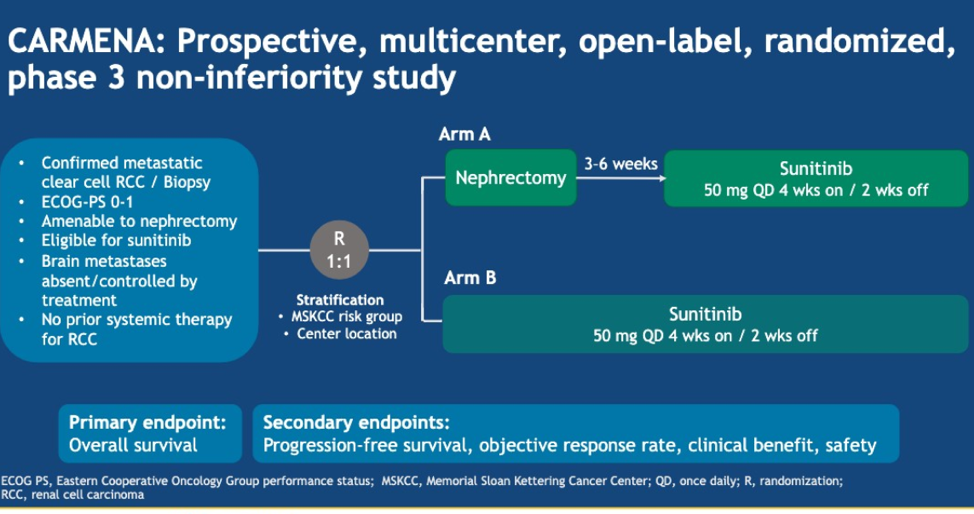
This abstract provides data with an updated median follow up of 61.5 months. Patients in CARMENA randomly assigned to either cytoreductive nephrectomy followed by sunitinib (arm A) or sunitinib alone (arm B). The original stratification for CARMENA was done with MSKCC risk groups and this abstract, the analysis is re-run based on IMDC risk groups. The primary endpoint was overall survival, which was also analyzed based on number of metastatic sites and whether or not patients in arm B received a secondary nephrectomy.
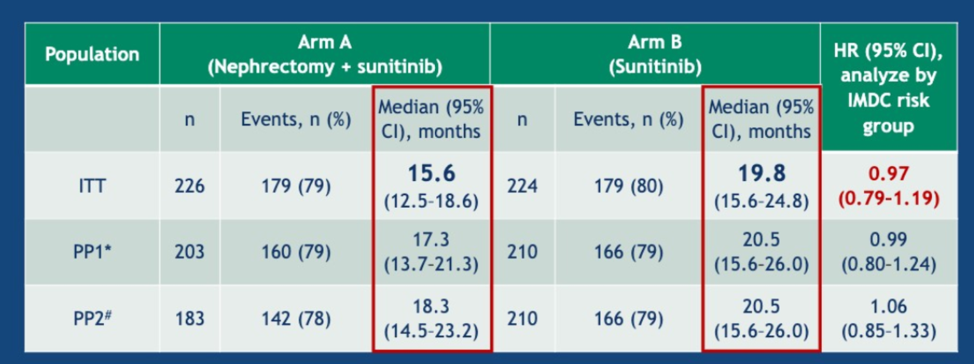
First, with respect to patients with one IMDC risk factor, there was no significant difference between Arm A and Arm B (31.4 vs 25.2 months, p=0.232).

For those with two risk factors, the median OS was 17.6 months in arm A and 31.2 months in arm B (HR 0.63, p = 0.033).
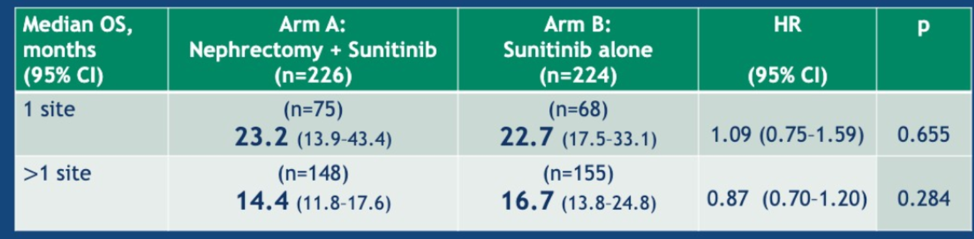
With regards to metastatic sites, there was no difference in overall survival between arm A and Arm B for those with 1 site or more than 1 site (data below).
Lastly, for the 40 patients who received a secondary nephrectomy in arm B compared with those without, there was a large difference in median OS, 48.5 mo [CI 95%: 27.9-64.4] vs 15.7 mo [CI 95%: 13.3-20.5].
Using the IMDC criteria, there was no significant difference in overall survival for patients in the intermediate group receiving cytoreductive nephrectomy. In the poor risk group, patients receiving sunitinib alone had better overall survival compared with patients receiving cytoreductive nephrectomy. The number of metastatic sites did not correlate with overall survival outcomes. Lastly, for patients who were candidates for a secondary nephrectomy, these patients tended to have longer OS than those who did not receive a secondary nephrectomy. This is not surprising as these are non-random patients who likely had a great treatment effect with sunitinib and were deemed, good surgical candidates. Now that several different combinations of immunotherapies being used in the front line setting, the decision regarding cytoreductive nephrectomy is becoming more complicated, especially since many patients receiving immunotherapy may obtain a CR. Secondary nephrectomies may play a greater role in the future as we change the sequencing of therapies from VEGF inhibitors in the first line to combination VEGF/IO or IO/IO. The data presented during this talk supports delayed nephrectomy in patients who have a good response to initial therapy.

Presented by: Arnaud Mejean, MD, PhD, Professor of Surgery, Chair of the Department of Urology, HEGP and Hopital Necker, APHP, Université Paris Descartes, Paris, France
Written by: Jason Zhu, MD. Fellow, Division of Hematology and Oncology, Duke University, Twitter: @TheRealJasonZhu at the 2019 ASCO Annual Meeting #ASCO19, May 31- June 4, 2019, Chicago, IL USA
References:
- Méjean A, Ravaud A, Thezenas S, et al. Sunitinib alone or after nephrectomy in metastatic renal-cell carcinoma. New England Journal of Medicine 2018;379:417-27.
- Motzer RJ, Russo P. Cytoreductive Nephrectomy — Patient Selection Is Key. New England Journal of Medicine;0:null.


