The goal of the presented study was to rapidly assess the effects of AV002, intranasal emulsified vasopressin, in patients with nocturnal polyuria. Researchers were interested in investigating if AV002 would affect length of first uninterrupted sleep period (FUSP) and a number of nocturic voids.
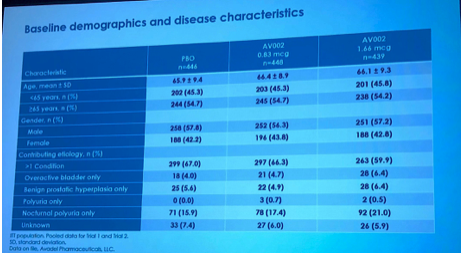
Total number of 1,333 patients ages 50 or older were randomized to 3 groups: 1.66mcg, 0.83mcg, or placebo. Baseline characteristics are refelected in Figure 1.
Rapid assessment of AV002 was measured after the first dose.
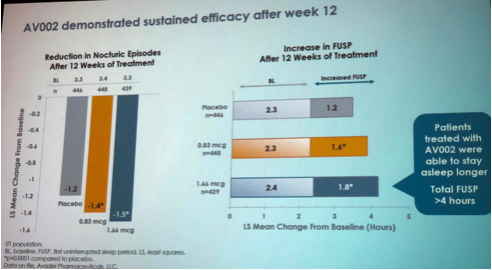
Data indicated a significant decrease in nocturic episodes in both treatment groups. In addition, FUSP has increased to more than 4 hours in people who took 1.66mcg of AV002 (Figure 2).
Researchers assessed adverse events across three study groups (Figure 3). No patients who took 0.83 mcg of vasopressin reported severe hyponatremia. This dose is recommended in people 65 and older. Other adverse events were comparable to the placebo arm.

Data demonstrate that AV002 can be used in treating patients with nocturia because it’s effective after first dose and relatively safe. Rapid effectiveness of the medication allows for the development of a patient-centric treatment plan and evaluation of medication dose and its’ health effects.
Presented by: Diane Newman, DNP, FAAN, Co-Director, Penn Center for Continence and Pelvic Health Division of Urology, University of Pennsylvania Medical Center, Philadelphia, Pennsylvania
Coauthors: Campeau L., Yang A., Francis, L.
Written by: Hanna Stambakio, BS, Clinical Research Coordinator, Division of Urology, University of Pennsylvania, Twitter: @PennUrology at the 2018 ICS International Continence Society Meeting - August 28 - 31, 2018 – Philadelphia, PA USA


