(UroToday.com) There has been a dramatic and rapid proliferation of systemic therapy options in advanced prostate cancer since the introduction of docetaxel for metastatic castration resistant prostate cancer (mCRPC) in 2004. These newer treatments have included a number of novel hormonal therapies (including abiraterone acetate and enzalutamide), second-line chemotherapy (cabazitaxel), bone-targeting agents (radium-223), and other targeted agents (including olaparib, rucaparib, and pembrolizumab), each of which has proven survival benefits. However, none of these agents have proven to be curative and there is, as a result, an ongoing need to identify novel treatment approaches. One of the approaches gaining the most interest is molecularly-targeted radionuclide therapy, leveraging molecularly targeting of cancer cells using prostate-specific membrane antigen (PSMA)-directed radioligands.
In a Late-Breaking Special Session at the 2021 Society of Nuclear Medicine & Molecular Imaging Annual Meeting, moderated by Dr. Thomas Hope, Dr. Michael Morris presented the first results of the VISION trial, a phase III study assessing lutetium-177-PSMA-617 in patients with metastatic castration-resistant prostate cancer. These data were first presented in a plenary session of the American Society of Clinical Oncology (ASCO) Annual Meeting.
Dr. Morris began by highlighting PSMA biology. He emphasized that PSMA is expressed across disease states in prostate cancer, and regardless of disease distribution (including bony disease, nodal disease, and visceral disease. Further, PSMA expression is present regardless of treatment with hormone directed therapies and may even increase as androgen-receptor signaling decreases. Further adding to its specificity in prostate cancer, PSMA has relatively restricted expression in normal tissues. As PSMA is highly expressed in prostate cancer and mCRPC lesions, the combination of PSMA-617 with the beta-emitter lutetium allows for the targeted delivery of ß-particle radiation to PSMA-expressing cells and surrounding microenvironment.
He emphasized that there has been a trend in prostate cancer to use therapeutic options earlier in the disease pathway. This has created the dilemma about what treatments can therefore be used in patients with more advanced disease. He highlighted limitations in available options, including cross-resistance of further androgen-receptor targeted therapy, the relatively limited patient populations for PARP inhibitors, sipuleucel-T, and immune checkpoint inhibitors, the restriction to bone-only disease for radium-223, and cabazitaxel among cytotoxic chemotherapies.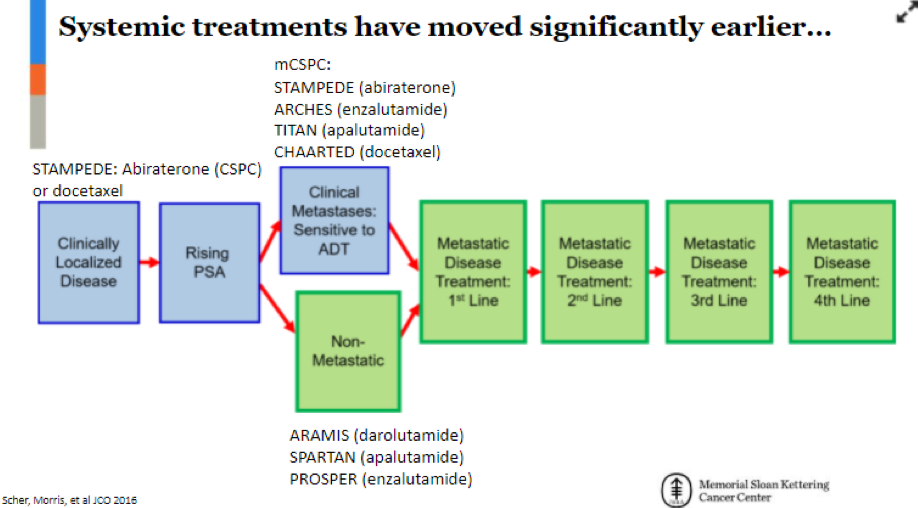
He emphasized that the initial data regarding Lu-PSMA were initially generated from compassionate use in Europe. Subsequently, work in Australia lead by Dr. Hofman provided the first prospective data supporting the use of Lu-PSMA in heavily pretreated patients with mCRPC. Following the LuPSMA trial, the ANZUP-led TheraP trial compared 177Lu-PSMA to cabazitaxel in patients who had progressed following docetaxel for mCRPC.
This randomized phase II trial relied on the selection with both PSMA- and FDG-PET prior to treatment. The TheraP trial primarily assessed PSA response, demonstrated a significant benefit for patients receiving Lu-PSMA.
These data formed the basis for VISION. The VISION trial is an international, randomized, open-label phase III study evaluating 177Lu-PSMA-617 in men with PSMA-positive mCRPC who had previously received treatment with at least one next-generation androgen receptor signaling inhibition (abiraterone, enzalutamide, etc) and one or two prior lines of taxane chemotherapy (NCT03511664). Additionally, patients must have had an ECOG performance status of 0-2 and a life expectancy of at least 6 months.
Importantly, patients must have had PSMA-positive disease on the basis of a central review of 68Ga-PSMA-11 staging scans. PSMA positivity was defined as uptake greater in metastatic lesions than the liver. Further, they could have no PSMA-negative metastatic lesions. Compared to TheraP, Dr. Morris highlighted that this imaging criterion was somewhat more liberal.
Following enrollment, patients were randomized in a 2:1 fashion to receive either 177Lu-PSMA-617 (7.4 GBq every 6 weeks x 6 cycles) plus standard of care (SOC) or SOC alone. SOC was determined for each patient prior to randomization. SOC treatments were at the discretion of the treating investigator; however, cytotoxic chemotherapy, immunotherapy, and radium-223 were explicitly excluded. Most patients received alternative androgen-directed therapies while others received palliative radiotherapy and glucocorticoids.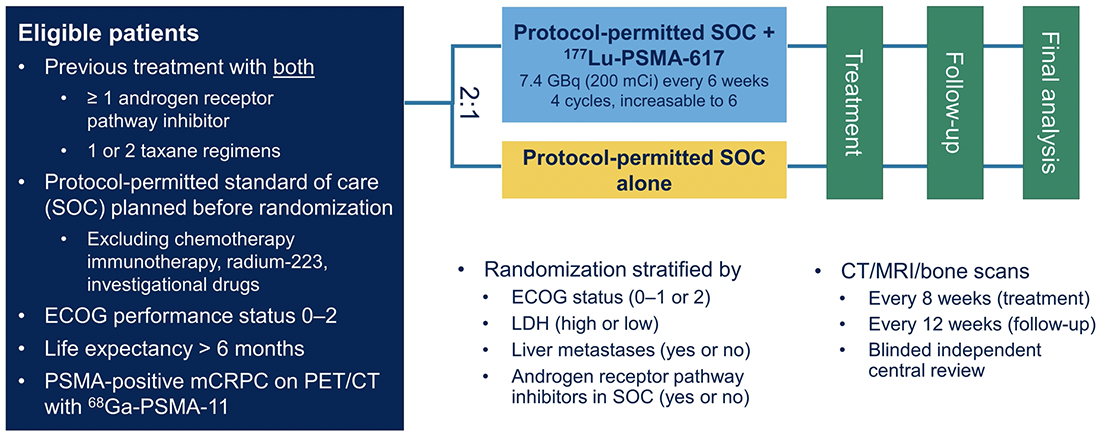
The authors assessed two alternate primary endpoints: radiographic progression-free survival (rPFS) using PCWG3 criteria by independent central review (ICR) and overall survival (OS). As alternate primary endpoints, the study would be deemed positive if either or both were positive. To inform their sample size calculations, the study was designed on the premise of a median rPFS of 4 months without 177Lu-PSMA-617 and 6 months with 177Lu-PSMA-617 (hazard ratio 0.67). Thus, for rPFS, this design allows an 84% power for this hazard ratio with 364 events in 557 patients. Accordingly, rPFS was allocated a one-sided alpha of 0.004. Similarly, with respect to overall survival, the authors assumed a median OS of 10 months without 177Lu-PSMA-617 and 13.7 months with 177Lu-PSMA-617 (hazard ratio 0.7306). Thus, 508 deaths in 814 patients would allow a 90% power to detect a difference with the HR of 0.7306 with allocation of a one-sided alpha of 0.021.
In addition to these two primary endpoints, they also assessed key secondary endpoints of objective response rate (ORR; RECIST v1.1), disease control rate (DCR), and time to first symptomatic skeletal event (SSE) as well as other secondary endpoints including safety and tolerability, biomarkers including PSA, and health-related quality of life and pain
Among 1179 screened patients, 869 met PSMA criteria and the VISION trial enrolled 831 patients between 4 June 2018 and 23 October 2019. In keeping with the 2:1 randomization schema, 551 patients were allocated to 177Lu-PSMA-617 + SOC and 280 were allocated to SOC only.
Dr. Morris emphasized that early in the study accrual, there was a very high rate of drop-out from the SOC alone arm (56%). Accrual was temporarily suspended as a result.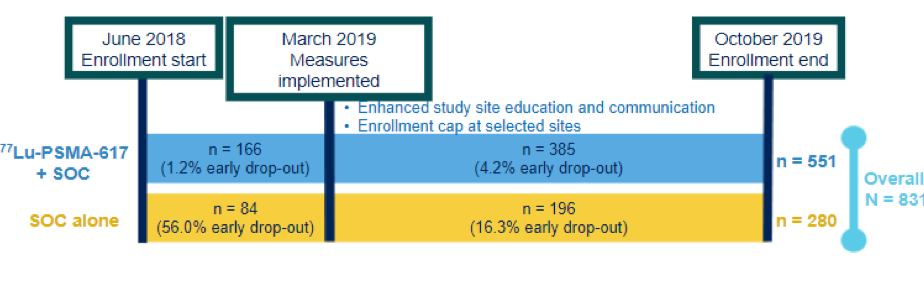
The trial group then reinforced the importance of trial conduct and subsequent drop-out from the SOC arm was lower moving forward. As a result, on the basis of discussion with the FDA, the rPFS analysis focused on patients accrued after March 2019.
As expected in the randomization design, demographic characteristics and baseline features were well balanced between the two treatment groups. Notably, approximately 7.5% of patients were Black.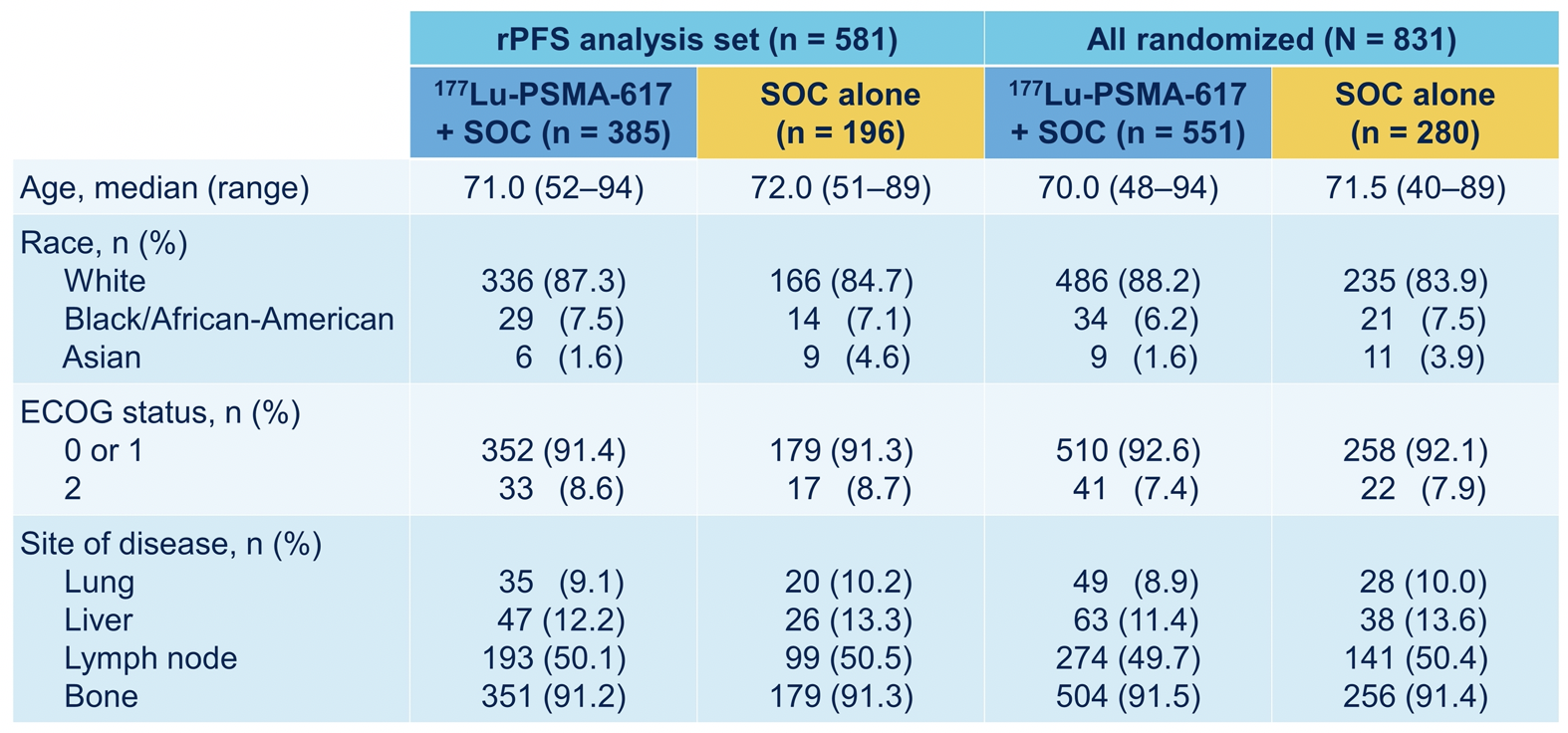
Further, pre-randomization treatment was well balanced. In both the rPFS analysis set and among all randomized patients, between 45-50% of patients had received more than one androgen receptor pathway inhibitor and 41-48% of patients had received more than one taxane regime.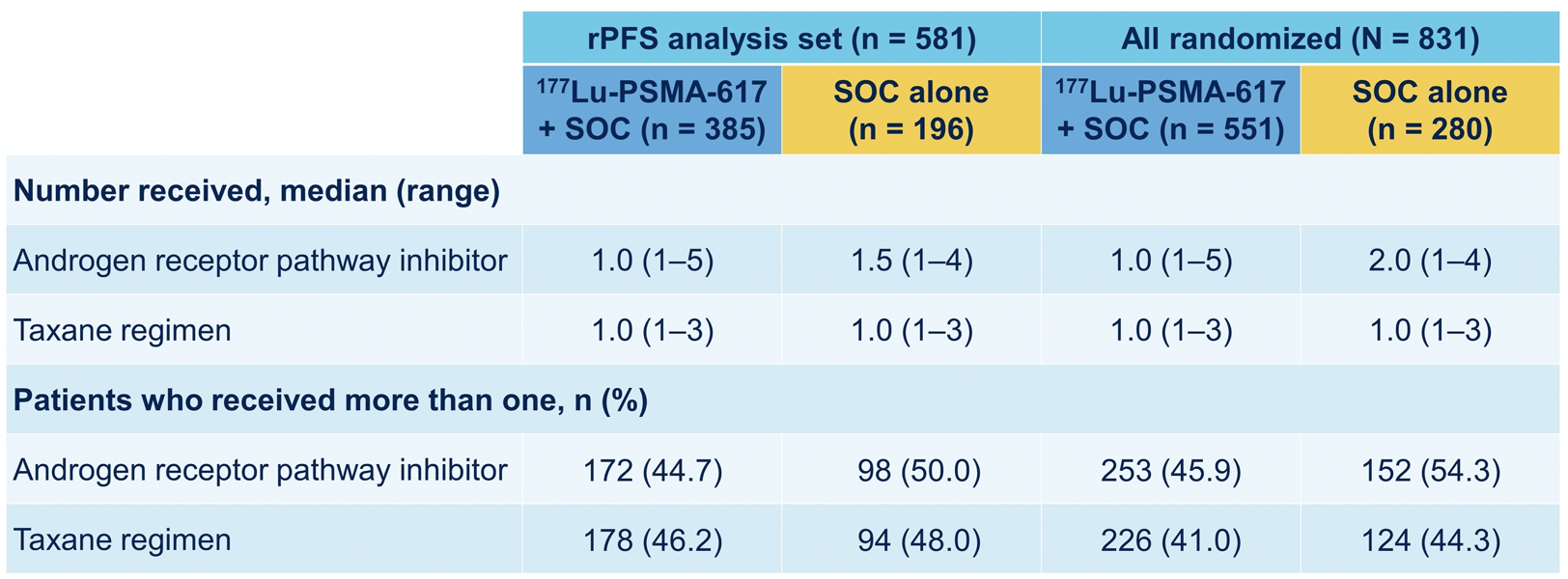
Dr. Morris then presented the primary results of the VISION trial assessing overall survival. Over a median study follow-up of 20.9 months (as of a data cut-off of 27 January 2021), treatment with 177Lu-PSMA-617 + SOC significantly improved overall survival by a median of 4.0 months (median OS, 15.3 vs 11.3 months; HR, 0.62 [95% CI: 0.52, 0.74]; p < 0.001, one-sided), compared to SOC alone, in the overall cohort of all randomized patients (n=831).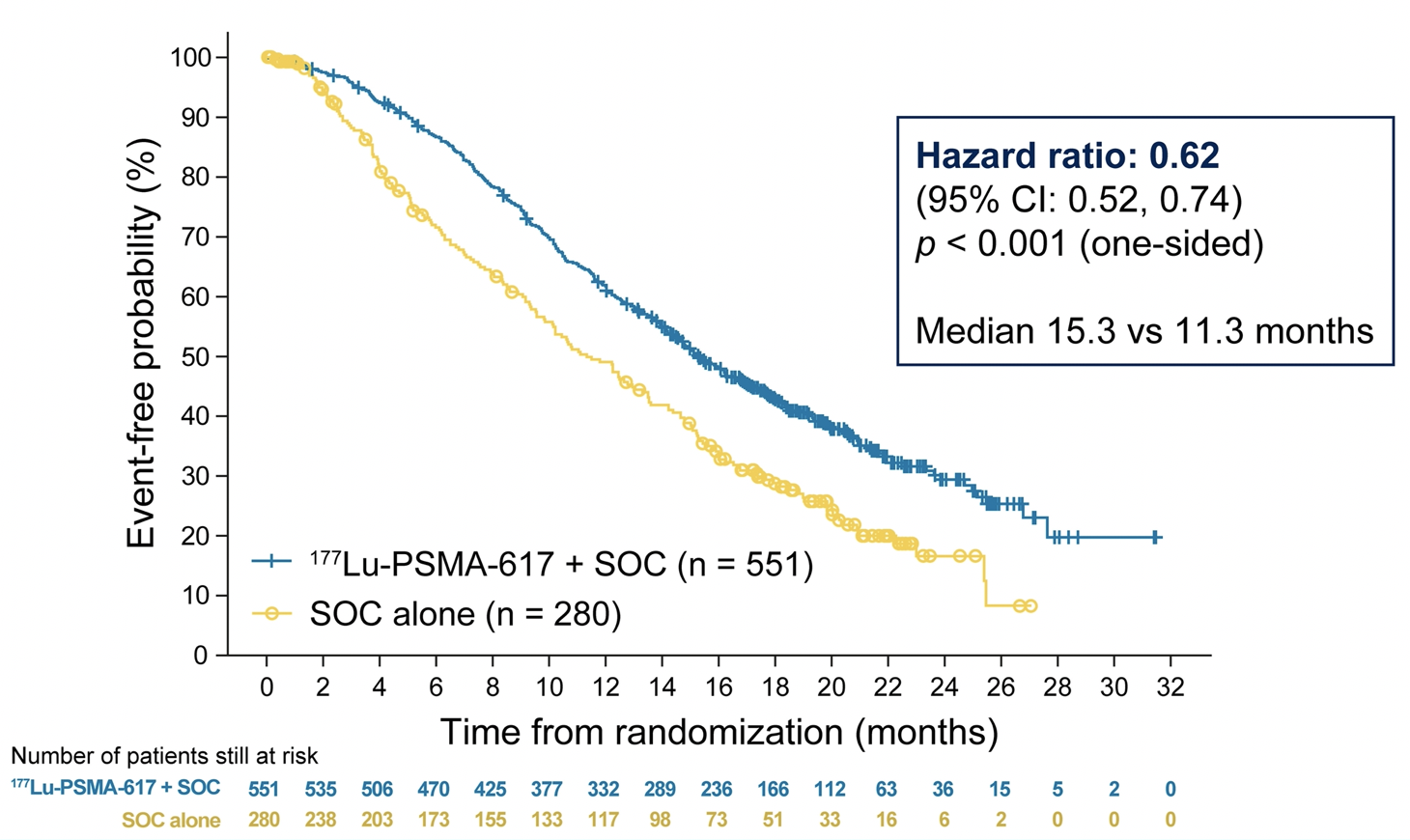
The authors further assessed overall survival in the rPFS analysis subset (n=581), showing that treatment with 177Lu-PSMA-617 + SOC significantly improved overall survival by a median of 4.2 months (median OS, 14.6 vs 10.4 months; HR, 0.63 [95% CI: 0.51, 0.7]), compared to SOC alone. The authors further performed a number of subgroup analyses showing a relatively consistent benefit of 177Lu-PSMA-617 + SOC across categories defined by the SOC used, disease characteristics, and demographic characteristics though many of these subgroup analyses were very small and are difficult to interpret. He noted that patients with liver metastasis consistently do poorly, and this was observed in VISION as well.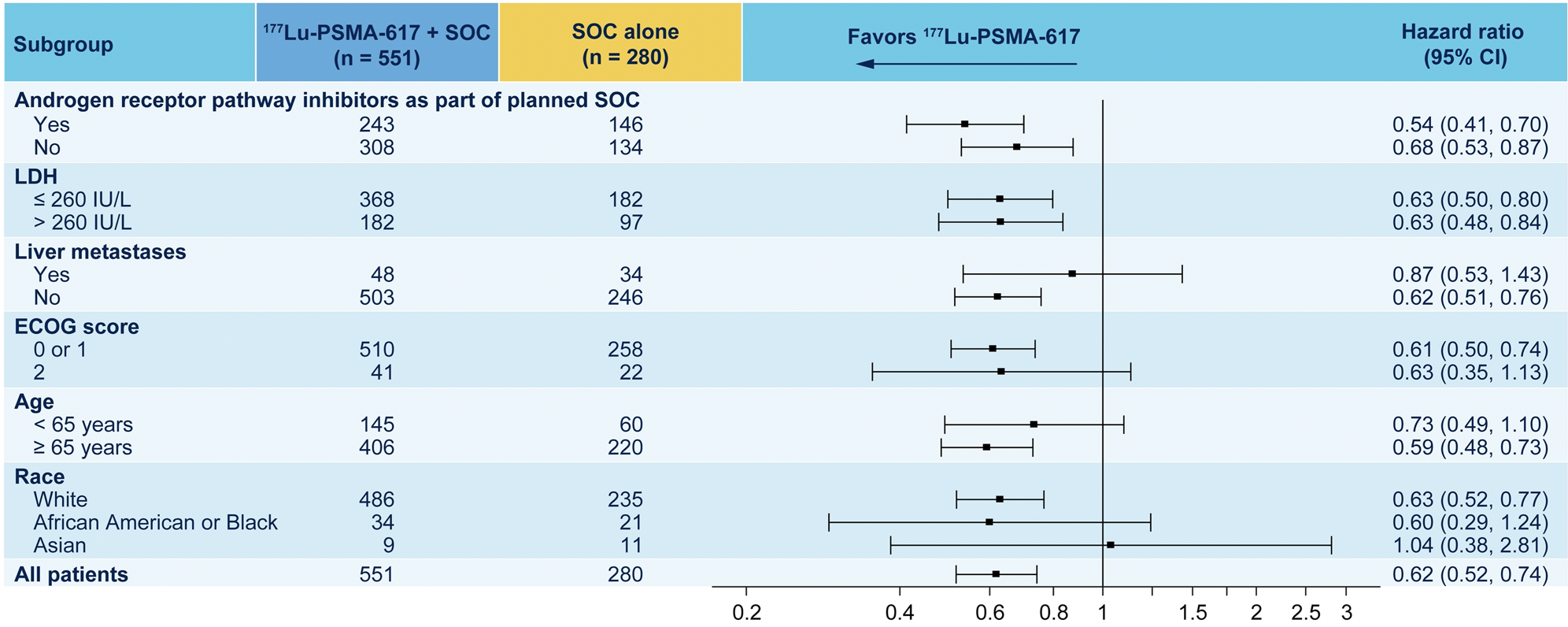
Dr. Morris then provided the results of the second, alternate primary endpoint showing that treatment with 177Lu-PSMA-617 + SOC significantly improved rPFS by a median 5.3 months (median rPFS, 8.7 vs 3.4 months; HR, 0.40 [99.2% CI: 0.29, 0.57]; p < 0.001, one-sided).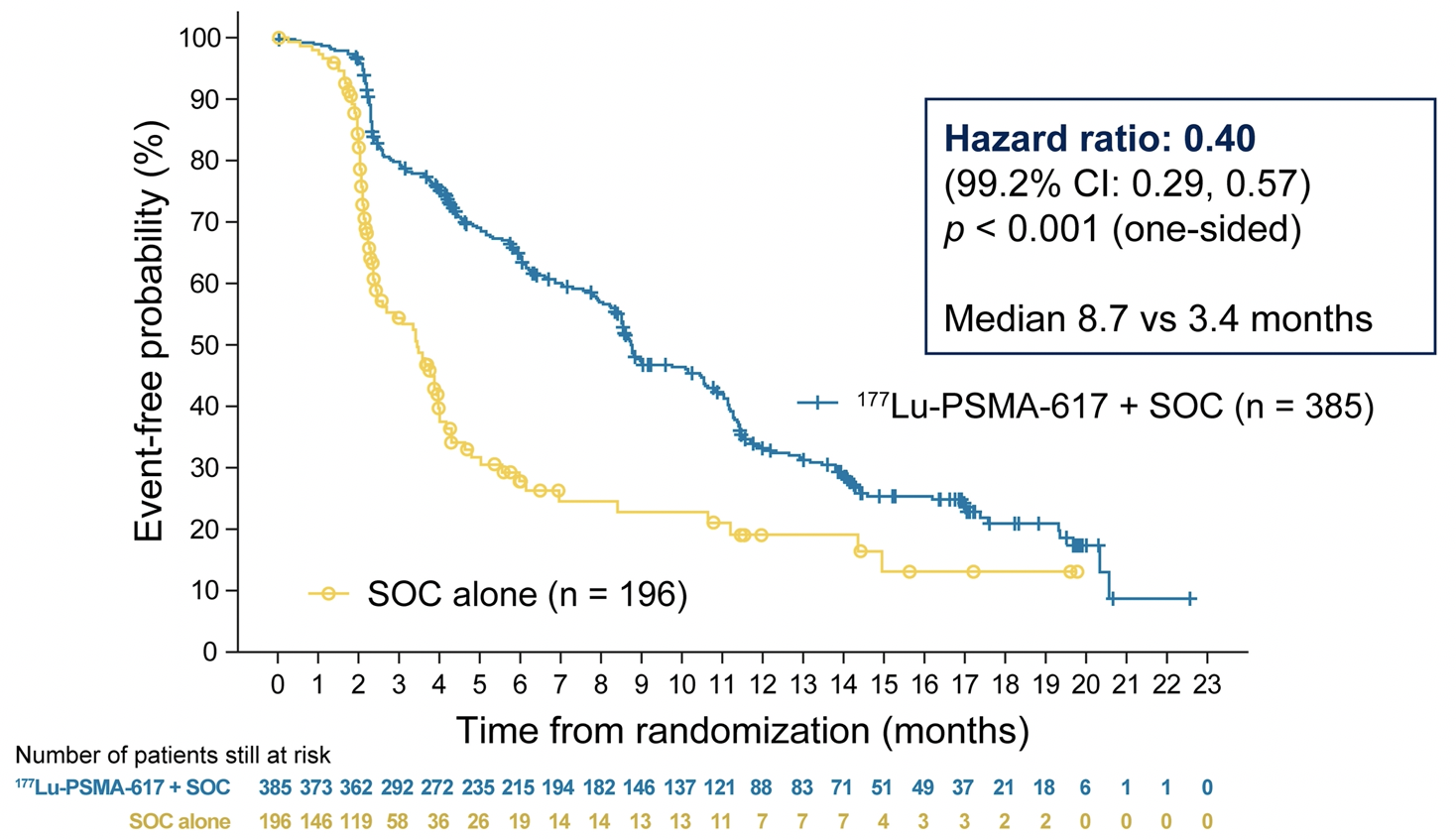
As they had done looking at overall survival, the authors then considered the effect of 177Lu-PSMA-617 + SOC in the whole randomized cohort (n=831) showing that rPFS was significantly improved by a median 5.2 months (median rPFS, 8.8 vs 3.6 months; HR, 0.42 [99.2% CI: 0.32, 0.58]). Again similar to overall survival, subgroup analyses demonstrated a generally consistent effect, though conclusions were limited in many subgroups due to small numbers.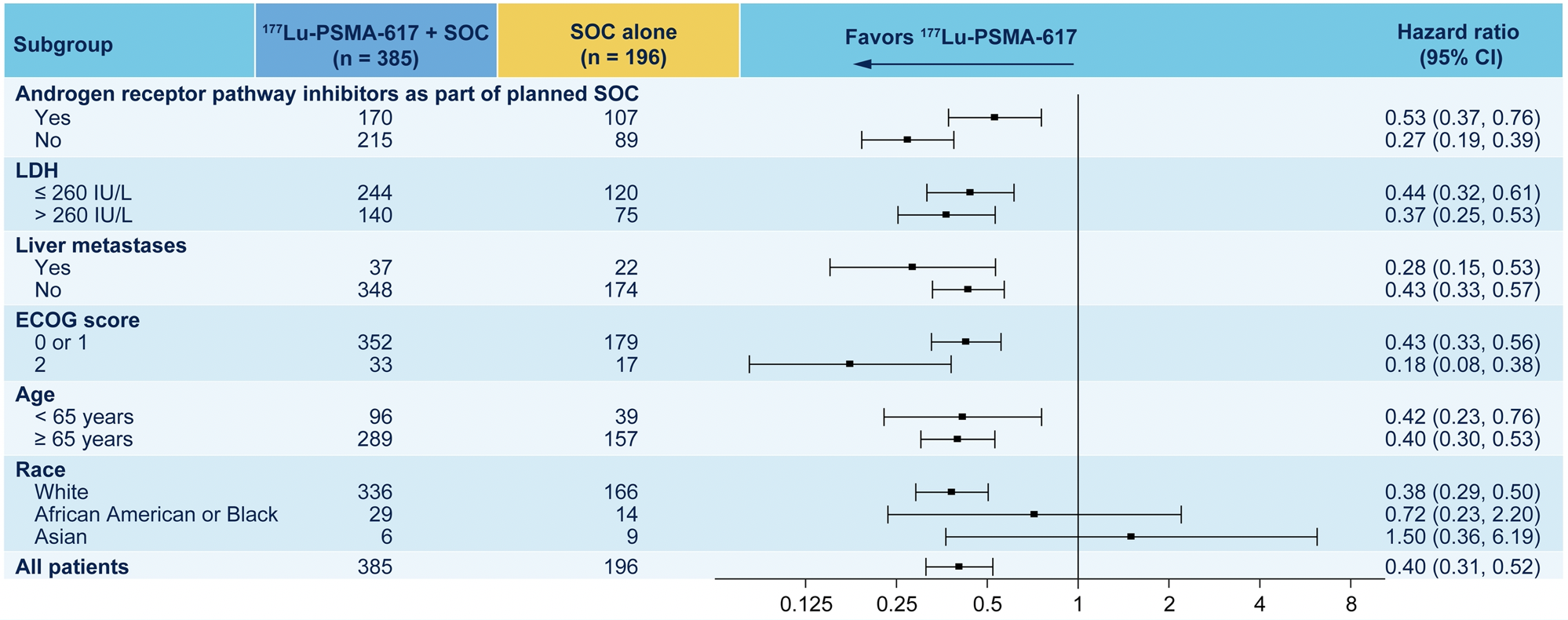
In addition to these primary endpoints, the addition of 177Lu-PSMA-617 to SOC statistically significantly improved all key secondary endpoints, including ICR-determined ORR (29.8% vs 1.7%), ICR-determined DCR (89.0% vs 66.7%) and time to first SSE (median time, 11.5 vs 6.8 months; HR, 0.50). Further, PSA responses (whether defined as a 50% decrease or an 80% decrease) were significantly more common among those treated with 177Lu-PSMA-617 + SOC.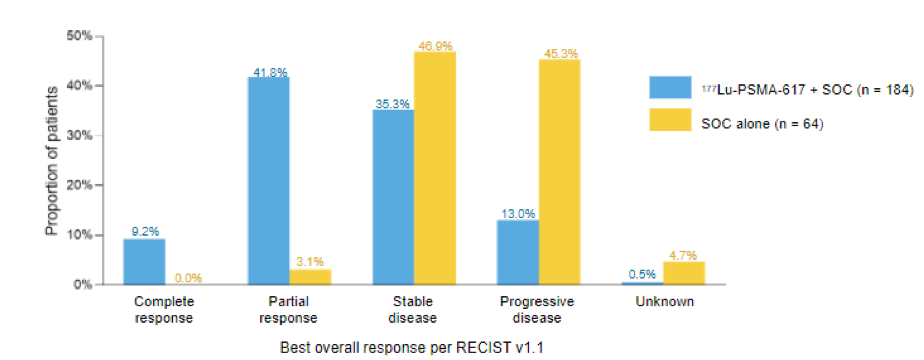
Dr. Morris emphasized that patients in the 177Lu-PSMA-617 + SOC arm had much longer durations of therapy: among those in the 177Lu-PSMA-617 + SOC arm, median duration of standard of care was 7.56 months (range 0.3-31.3 months) compared with 2.07 months (range 0.0-26.0 months) in the SOC alone arm.
Further, he emphasized that post-protocol treatment could not explain the observed survival benefits seen in the 177Lu-PSMA-617 arm as there was a somewhat higher use of chemotherapy and radiotherapy among patients randomized to the SOC alone arm. Only about 20% of patients received chemotherapy following protocol-directed therapy. Further, the use of radiopharmaceuticals was uncommon in both arms following protocol treatments.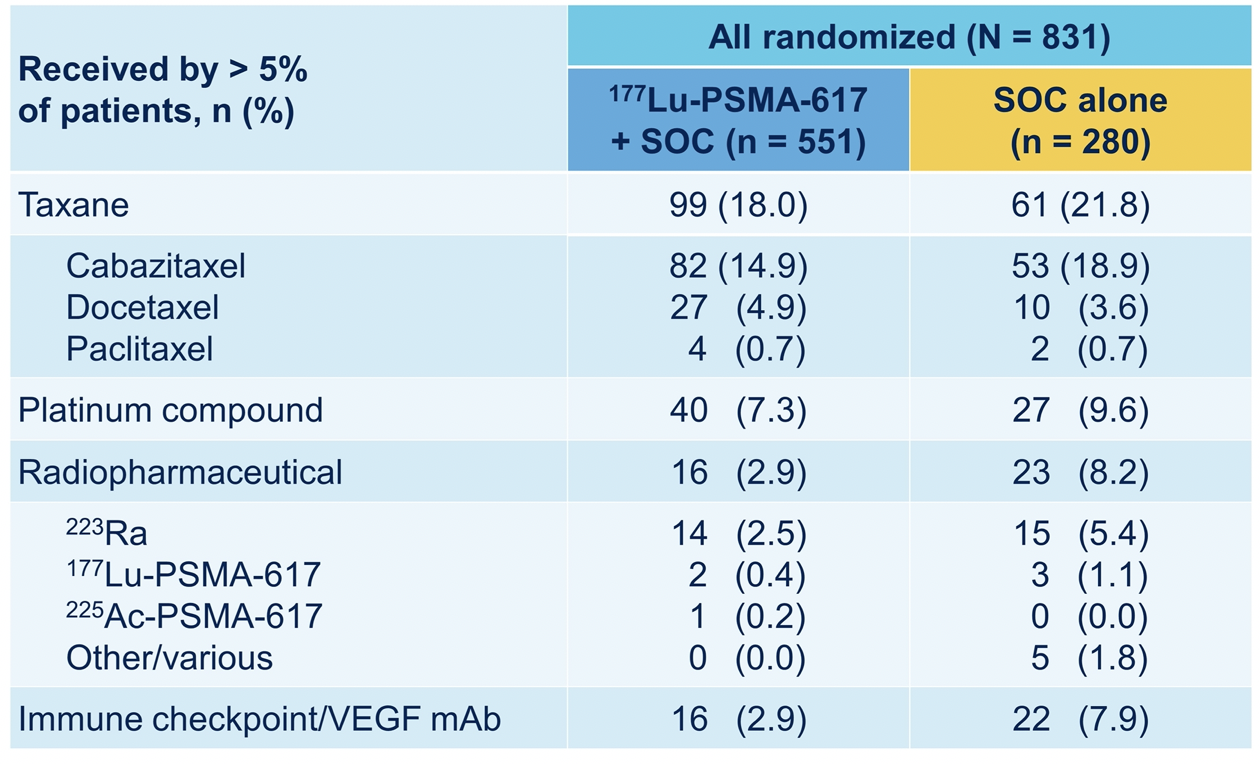
While a higher rate of high-grade (grade 3-5) treatment-emergent adverse events was observed with 177Lu-PSMA-617 (28.4% vs 3.9%), overall therapy was well tolerated. However, it bears note that there were 5 deaths attributable to 177Lu-PSMA-617 treatment. In terms of specific adverse events, treatment with 177Lu-PSMA-617 + SOC was associated with increased rates of bone marrow suppression, xerostomia, and nausea and vomiting. He concluded that these data are consistent with prior reports, and don’t represent a new safety signal.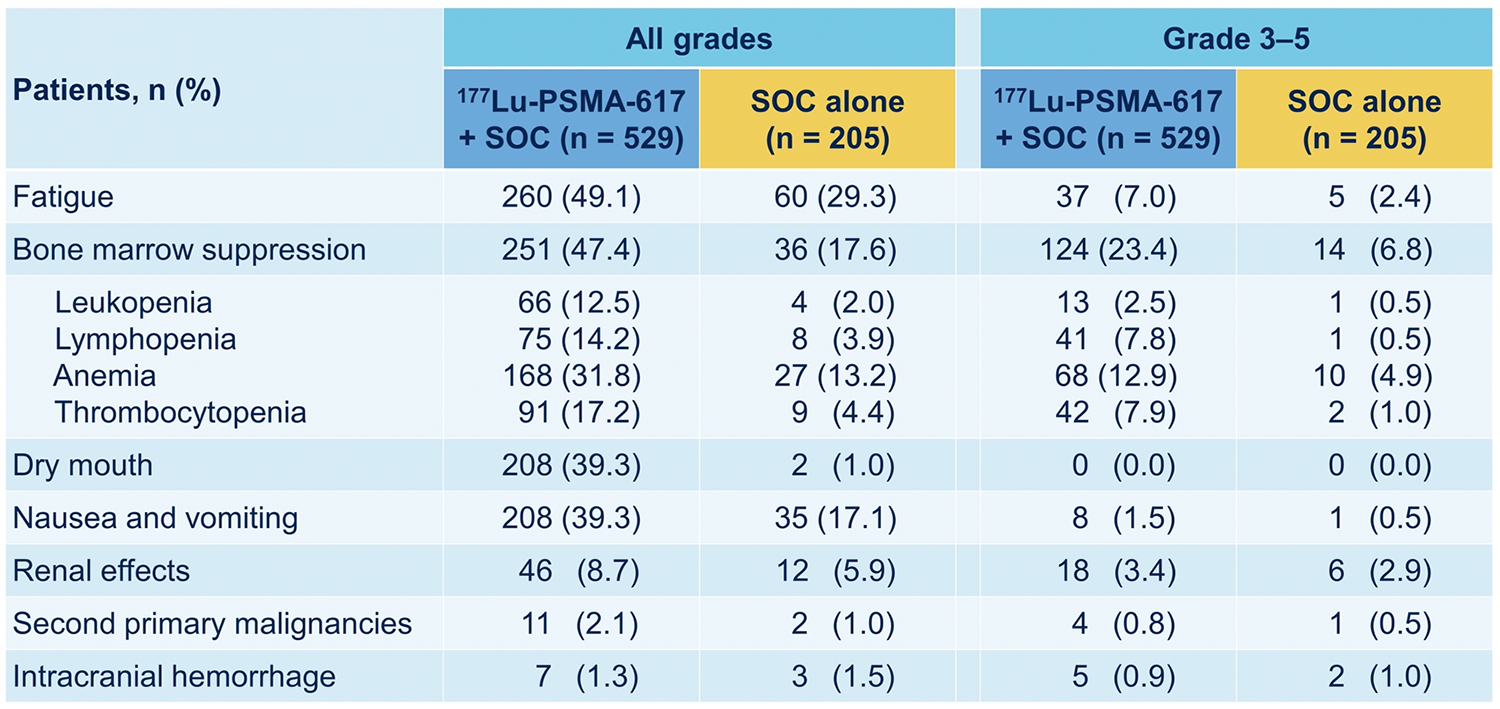
Dr. Morris then concluded that the VISION trial demonstrates that 177Lu-PSMA-617 can be safely added to SOC treatment and improves rPFS and prolongs OS compared with SOC alone in men with advanced-stage PSMA-positive mCRPC, supporting its adoption as a standard of care.
Dr. Morris then highlighted further directions for Lu-PSMA based theranostic treatment. First, he emphasized ongoing trials assessing the use of this treatment approach earlier in the disease process.
Further, he highlighted the importance of ongoing biomarker analyses from VISION, including the role of imaging to define the suitability for treatment. These biomarker analyses will also help to inform an understanding of selected populations who may most benefit, considering, in particular, those with DNA repair deficiency and specific ethnicities. Additionally, combination approaches including other accepted systemic therapies for prostate cancer as well as other treatment modalities. Further, Dr. Morris emphasized the importance of ongoing work to consider the sequencing of therapy and the use of Lu-PSMA earlier in the disease trajectory. Finally, there is significant ongoing work assessing new targets, new targeting agents, and new ligands.
Dr. Ghassan El-Haddad emphasized the importance of collaboration between nuclear medicine clinicians and urologists and medical oncologists, highlighting the importance of their involvement in multi-disciplinary clinics and tumor boards. Strengthening this collaboration was critical to reduce drop-out within the VISION trial.
Dr. Hope then raised the question of the importance of PSMA-PET/CT for inclusion eligibility. Dr. El-Haddad emphasized that the concept of theranostics relies on interplay of identified radiographic abnormalities and targeted therapeutic intervention. He highlighted the role of approval bodies to guide the importance of PSMA-PET/CT in this space. Dr. El-Haddad further emphasized the utility of PSMA-PET/CT to guide dosimetry and other considerations, beyond initial inclusion. Dr. Morris emphasized the importance that expense will guide in this field: we need to consider whether diagnostic PSMA-PET/CT is informative with predicting response to Lu-PSMA. In the absence of a predictive ability, he suggested that the cost of PSMA-PET may not be warranted.
Considering future directions, Dr. Morris suggested that he expected earlier use of Lu-PSMA may result in greater absolute improvements in benefit, though more dramatic issues with long-term toxicity issues.
Presented by: Michael Morris, MD, Medical Oncologist Clinical Director, Genitourinary Medical Oncology Service & Prostate Cancer Section Head, Division of Solid Tumor Oncology, Memorial Sloan Kettering Cancer Center, New York, New York, USA.


