Subsequently, further follow-up of SPARTAN, PROSPER, and ARAMIS has demonstrated a significant improvement in overall survival (OS) as well. However, there are a number of adverse events (AEs) of interest commonly associated with ARI therapy, such as fatigue, falls, fractures, rash, mental impairment, and hypertension, as well as interactions between ARIs and concomitantly administered drugs, which can impact patient daily life. In a poster presentation at the 2021 American Society of Clinical Oncology Genitourinary Cancers annual meeting (ASCO GU), Dr. Smith and colleagues present long-term data on the safety of darolutamide from both the double-blind and open-label portions of the ARAMIS trial.
To briefly summarize the methodology of the ARAMIS trial, the authors accrued 1509 patients with nmCRPC who were subsequently randomized 2:1 to darolutamide 600 mg twice daily (n=955) or PBO (n=554). All patients continued ADT. The trial was designed and powered to assess the primary outcome of metastasis-free survival.
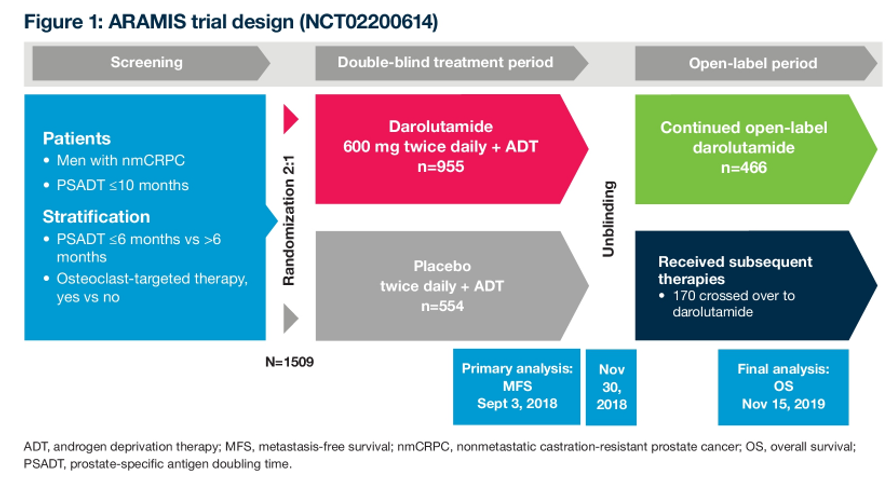
In addition to OS, other secondary endpoints included times to pain progression, first cytotoxic chemotherapy, first symptomatic skeletal event, and safety. The OS analysis was planned to occur after approximately 240 deaths, with the hierarchical assessment. Following data cut-off for the primary analysis, patients in the placebo arm who had not yet progressed could cross over to darolutamide, and patients in the darolutamide arm could continue in the open-label portion. In this analysis, the data cut-off was November 15, 2019.
At the time of final analysis, the median treatment duration for patients randomized to darolutamide was 18.5 months during the double-blinded period and 25.8 months for the combined double-blinded and open-label period. At the final cut-off date, 48.8% of patients in the darolutamide group were still receiving treatment.
The increase in the incidence of any-grade AEs (85.7% vs 89.8%) and serious AEs (26.1% vs 32.1%) from the end of the double-blind period and the end of the overall observation period (double-blind and open-label) was small, with only minor numerical changes for ARI-associated AEs observed.
When the incidences of adverse events were corrected for the duration of exposure, there were minimal differences between the double-blind and overall observation period, e.g., the fracture rate was 3.4 vs 4.0 per 100 patient-years, respectively. Fatigue was the only ARI-associated AE of interest that exhibited an incidence greater than 10%, among patients receiving darolutamide in the overall observation period. The incidence of permanent discontinuation of darolutamide due to AEs increased slightly from 8.9% during the double-blind period to 10.5% during the overall observation period; the incidence of discontinuation of placebo due to AEs during the double-blind period was 8.7%.
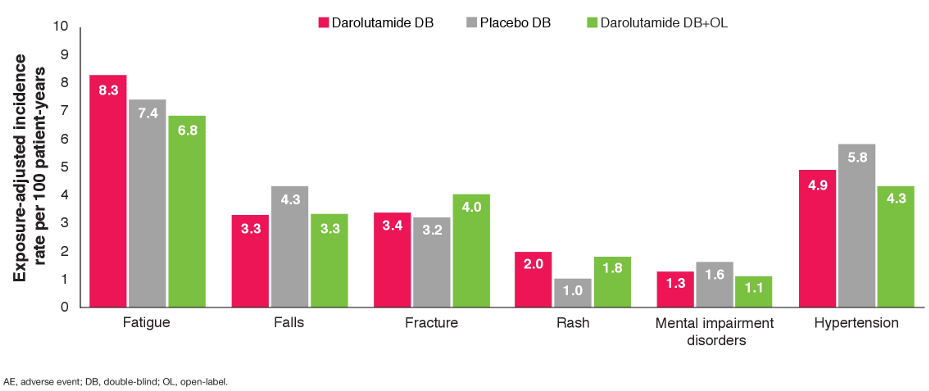
The authors conclude that, with longer durations of follow-up, darolutamide remained well tolerated with no new safety signals.
Presented by: Matthew R. Smith, MD, Ph.D., Professor of Medicine, Harvard Medical School, Assistant in Medicine, Hematology/Oncology, Massachusetts General Hospital, Boston, Massachusetts, USA
Co-Authors: Karim Fizazi, Teuvo L. J. Tammela, Felipe Melo Cruz, Luke T. Nordquist, Diana Sofia Aleman Polanco, Urban Emmenegger, Glauco Costa Silveira, Raoul S. Concepcion, Adriano Paula, Carlos Augusto de Mendonça Beato, Neil Fleshner, Martin Eduardo Richardet, Iris Kuss, Marie-Aude Le Berre, Gustavo Borghesi, Toni Sarapohja, Neal D. Shore
Written by: Christopher J.D. Wallis, Urologic Oncology Fellow, Vanderbilt University Medical Center Twitter @WallisCJD during the 2021 ASCO Genitourinary Cancers Symposium (ASCO GU), February 11th to 13th, 2021
Related Content:
Overall Survival Data from the Global, Phase 3 ARAMIS Trial in Men with Non Metastatic Castration-Resistant Prostate Cancer (nmCRPC) - Karim Fizazi


