In a poster presentation at this year’s European Society of Medical Oncology (ESMO) 2020 Virtual Annual Meeting, Dr. Fizazi presented updated data from the ARAMIS trial assessing the tolerability and treatment response to darolutamide in men with high-risk nmCRPC.
To briefly review, in the ARAMIS trial, 1509 patients with nmCRPC defined as high-risk for metastases based on a PSA doubling time ≤10 months were randomized in a 2:1 fashion to darolutamide 600mg PO BID or placebo while continuing standard ADT. In this analysis, the authors assess the association between decline in PSA from baseline and metastasis-free survival.
First, assessing toxicity, the authors found that 97.2% of patients randomized to darolutamide received the planned treatment course. Dose modifications were similarly frequently among those receiving darolutamide (15.2%) and placebo (9.7%). Rates of treatment discontinuation due to adverse events were no higher among patients randomized to darolutamide (8.9%) than those randomized to placebo (8.7%). These analyses support the findings of the primary report of this study.
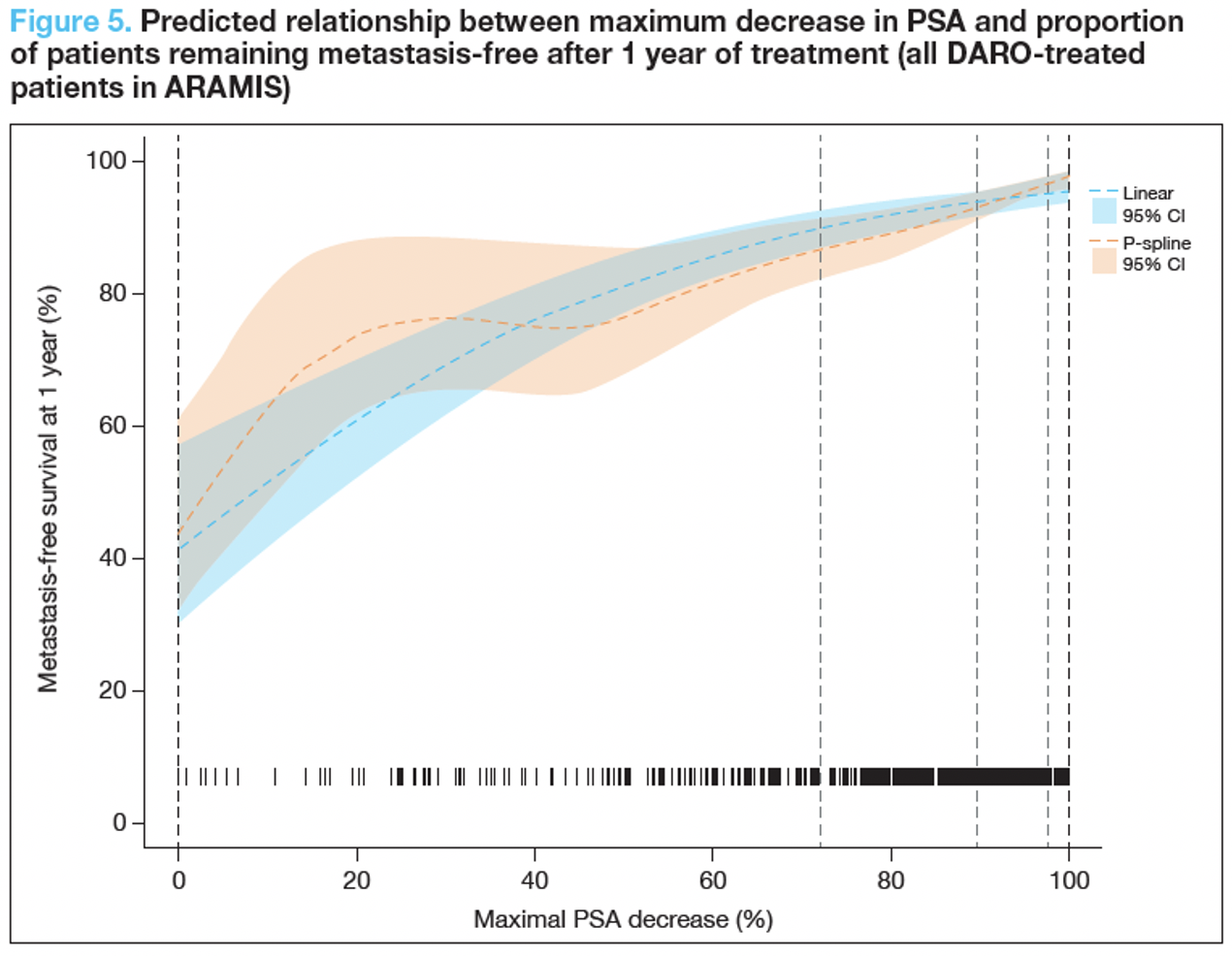
At baseline, patients randomized to darolutamide (PSA median 9.0ng/mL, range 0.3-858.3) and to placebo (PSA median 9.7ng/mL, range 1.5-885.2) had similar PSA levels. Following the initiation of therapy, the median PSA decline among those randomized to darolutamide was 91.7% and to placebo was 31.9%. The authors demonstrated that pharmacodynamic modeling demonstrated a significant positive association between maximal PSA decrease from baseline and prolonged metastasis-free survival.1
Presented by: Karim Fizazi, MD, Ph.D., medical oncologist, Head of the Department of Cancer Medicine at the Institut Gustave Roussy, Villejuif, France, and Professor in Oncology at the University of Paris
Reference:
1. Fizazi, K., Shore, N., Tammela T. L., et al. Darolutamide in Nonmetastatic, Castration-Resistant Prostate Cancer. N Engl J Med. 2019;380:1235-46.
Written by: Christopher J.D. Wallis, Urologic Oncology Fellow, Vanderbilt University Medical Center (Twitter: @WallisCJD) at the European Society for Medical Oncology Virtual Congress, ESMO Virtual Congress 2020 #ESMO20, 18 Sept - 21 Sept 2020
Related Content:


