Dr. Schade began by highlighting the current state of prostate cancer focal therapy with several energy sources already being U.S. Federal Drug Administration (FDA) approved. However, for the most part, insurance companies consider these approaches experimental and thus, there are coverage issues for many patients. However, there are at least 130 active or pending trials of focal therapy registered on clinicaltrials.gov suggesting that data to move this approach from experimental to more accepted may be soon forthcoming.
Among treatment technologies, he began by assessing high-intensity focused ultrasound (HIFU). Worldwide, there are more than 25 years of experience with transrectal HIFU, though it was only approved by the FDA for use in the United States in 2015. In general, HIFU relies on thermal ablation with coagulative necrosis. Most patients achieve good functional outcomes (including continence and erectile function) as well as reasonable oncologic outcomes.
However, transrectal ultrasound is limited by prostate size and minimal real-time feedback. In contrast, a newer approach is transurethral HIFU which is not limited by prostate size to the same degree. While the TACT trial assessing transurethral ultrasound ablation (TULSA) transurethral HIFU provided whole gland treatment, Dr. Schade suggested that it could be used for focal treatment. Moving forward, he suggested that MR-guided transrectal HIFU would allow for better real-time feedback and targeting, in part as a result of a 1,000 element phased arrange. While Phase I studies have demonstrated promising results, larger trials are underway.
Additionally, HIFU may be used for histotripsy, non-thermal ablation in contrast to the standard thermal ablation. This uses short, extremely high amplitude pulses. Using B-mode ultrasound, histotripsy based HIFU allows for real-time feedback by visualizing hyperechoic bubbles and hypoechoic treatment cavities. While this approach is promising, Dr. Schade suggested that in human studies remain five to seven years away.
Next, Dr. Schade discussed photodynamic therapy. This approach relies on systemic perfusion of a vascular photosensitizing agent. Following light activation, the photosensitizer produces free radicals that produce tissue damage. In the Phase III TOOKAD study, the use of the photosensitizer padeliporfin demonstrated a significant reduction in progression and radical therapy for patients on active surveillance. However, the FDA voted against approval of padeliporfin in February 2020, potentially spelling the end of this approach in the United States.
However, there are numerous ongoing investigations in the realm of photodynamic therapy. The first of these relates to the use of gold silica nanoparticles. Following vascular perfusion, these nanoparticles accumulate in the tumor. Laser activation of these results in tissue damage. An initial 16 patient pilot study demonstrated the preservation of functional outcome and negative biopsies at 12 months in 14 of 16 patients.
Future approaches may also utilize biologically targeted photosensitizers, such as prostate-specific membrane antigen (PSMA) targeted agents. These have demonstrated promise in xenograft models.
Another approach to focal therapy is laser ablation. This may be accomplished via either transperineal or transrectal access. Typically this is performed under MRI guidance and monitoring. To date, there are two FDA approved laser ablation platforms (Visualase® and Tranberg®). While data to date are limited to Phase II data, positive in-field biopsy rates are relatively low (~11%) without any long term quality of life impact.
Irreversible electroporation is another approach which relies upon pulsed, low energy, direct current via transperineal probe placement. This induces permeability of the cell membrane leading to apoptosis. In a Phase II study, rates of post-ablation biopsy were nearly 40% though functional outcomes were well maintained. However, there are a number of notable considerations for this approach including the need for general anesthesia with paralysis and cardiac gating (to avoid arrhythmias) as well as the lack of feedback.
Cryotherapy is another approach for focal therapy, although Dr. Schade did not discuss this except in passing.
He closed by highlighting that focal ablation depends on prostate imaging. Current, contemporary MRI has limitations for focal therapy including imaging-invisible lesions and significant underestimation of tumor volume.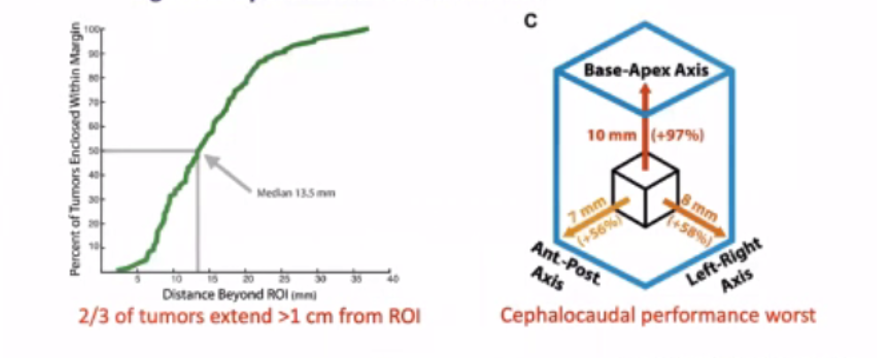
Newer imaging approaches, including PSMA PET, micro-ultrasound, and 7T MRI show promise for improved tumor detection and localization.
Presented by: George R. Schade, MD, Assistant Professor in Urology, Adjunct Assistant Professor, University of Washington Center for Industrial and Medical Ultrasound and Applied Physics Lab, University of Washington, Seattle, Washington, USA
Written by: Christopher J.D. Wallis, MD, PhD, Urologic Oncology Fellow, Vanderbilt University Medical Center, Nashville, Tennessee, USA Contact: @WallisCJD on Twitter at the Society of Urologic Oncology (SUO) - American Urologic Association (AUA) 2020 Summer Webcast Program, July 18, 2020
Related Content:
Read — SUO - AUA Summer Webcast: Focal Therapy for Prostate Cancer: SPARED Registry for Focal Therapy
Read — AUA 2020: Pivotal Trial of MRI-Guided Transurethral Ultrasound Ablation in Men with Localized Prostate Cancer: Two-Year Follow-Up

