(UroToday.com) The 23rd Annual Meeting of the Society of Urologic Oncology held in San Diego, CA between November 30th and December 2nd, 2022 was host to a session on the real life impact of CxBladder tests for the diagnosis and surveillance of bladder cancer.
Dr. John Sfakianos began his presentation by noting that current guidelines for surveillance provide little concrete guidance for when surveillance may be stopped. The AUA currently provides the following guidance by risk category:
- Low risk: After five years, shared decision making between patients and providers
- Intermediate risk: No definitive stopping point
- High risk: No definitive stopping point
Conversely, the EAU provides the following guidance:
- Low-risk tumors: After five years of follow-up, discontinuation of cystoscopy or its replacement with less invasive methods can be considered
- In patients with high risk, or very high-risk disease treated conservatively, recurrences after ten years of tumor-free survival are not unusual. As such, life-long follow-up is recommended.
Although cystoscopies have low, non-zero morbidity, they remain psychologically cumbersome for patients. Currently, urinary biomarkers are used as adjunct (or companion) tests to improve detection at the time of flexible cystoscopy or as replacement tests to reduce the number of flexible cystoscopies. Everyone’s goal, however, is to have a noninvasive monitoring method for patients with non-muscle invasive bladder cancer.
The following questions for surveillance of patients with non-muscle invasive bladder cancer remain:
- Can we use non-invasive methods to adjust surveillance schedules?
- Can we use non-invasive methods to stop cystoscopies when risk is very low?
- Can we use non-invasive methods to personalize scheduling?
- Do we have to adjust for never recurrence versus multiple recurrences?
In order to answer these questions, we need to better understand the natural history of patients with bladder cancer. Dr. Sfakianos presented, as of yet, unpublished data from a cohort of 386 patients with non-muscle invasive bladder cancer, from Karolinska, Sweden, who were followed for a median of 10.8 years (IQR: 4.2 to 20.2 years). Of these 386 patients, 284 (73.6%) LG or HG Ta disease and 55 patients (14.2%) died of bladder cancer.

Long-term follow-up of this cohort showed:
- There was a clear separation of the cancer-specific survival curves amongst patients with LG Ta, HG Ta and HG T1 disease, as demonstrated below:

- Recurrence and progression rates beyond five years in patients with HG T1 disease are essentially a never occurrence and occur only rarely in patients with LG Ta disease.

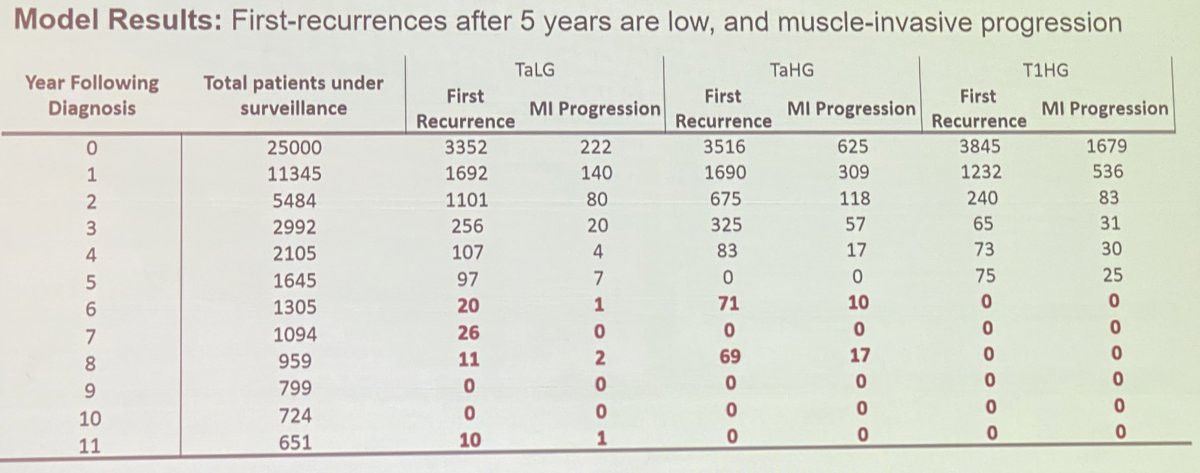
As such, these results can inform how we tailor our surveillance strategies in these patients, as a function of survival time from initial diagnosis. Dr. Sfakianos next addressed how he uses Cxbladder in his practice, by risk category:
- Low-risk patients: For patients without recurrence by three years, he currently uses Cxbladder, a urinalysis, and urine cytology instead of cystoscopy
- Intermediate-risk patients: For patients without recurrence by five years, he currently uses Cxbladder, a urinalysis and urine cytology every 6 months instead of a cystoscopy
- High-risk patients: For patients without recurrence by five years, he currently uses Cxbladder, a urinalysis, and urine cytology every 6 months instead of a cystoscopy
Dr. Sfakianos next went on to present a case study highlighting the utility of Cxbladder from his own practice This was an 83-year-old male who initially presented with hematuria in 2017. The patient was found to have CIS of the bladder. He was treated with induction BCG and maintenance for 1 year which was completed in 2019. The patient had no evidence of disease recurrence and had undergone surveillance cystoscopies and urine tests every six months. The patient was doing well on surveillance until February 2020, when he was found to have a negative cystoscopic examination, but urine cytology showed evidence of malignant cells. Faced with this clinical dilemma, Dr. Sfakianos ordered Cxbladder Monitor test, which came back as 2.7 (below the pre-defined cut off of 3.5).

Despite the negative test result, Dr. Sfakianos took the “safe” option of taking the patient back to the operating room for random biopsies and an upper tract evaluation, with all the work up negative for presence of gross malignancy. He subsequently has had positive cytology results, but with negative cystoscopies and negative Cxbladder Monitor test results during his surveillance. His last evaluation as of May 2022 still showed no evidence of disease.
While peer-reviewed studies1 have demonstrated that Cxbladder Monitor has an excellent negative predictive value in the range of 97 to 98.5%, Dr. Sfakianos’ group performed an internal “real world” analysis, that validated the findings from these prior studies. In this series of 161 patients with non-muscle invasive bladder cancer and 439 Cxbladder Monitor tests, all patients underwent a cystoscopy plus cytology with subsequent TURBT in case of a positive cystoscopy. This analysis demonstrated that overall, for all patients, the negative predictive value was 90.0%. For LG Ta and HG T1 patients, the corresponding negative predictive value (NPV)s were 80.2% and 97.0%, respectively.
Dr. Sfakianos concluded his talk by emphasizing that understanding the risk recurrence for the various NMIBC risk groups and having a test such as Cxbladder Monitor with a high NPV may allow us to improve/personalize noninvasive surveillance testing for our patients.
Presented by: John Sfakianos, MD, Assistant Professor of Urology and Urologic Oncology, Icahn School of Medicine at Mount Sinai, New York, NY
Written by: Rashid Sayyid, MD, MSc – Society of Urologic Oncology (SUO) Clinical Fellow at The University of Toronto, @rksayyid on Twitter during the 23rd Annual Meeting of the Society of Urologic Oncology (SUO), Nov 30 – Dec 2, 2022. San Diego, CA
References:


