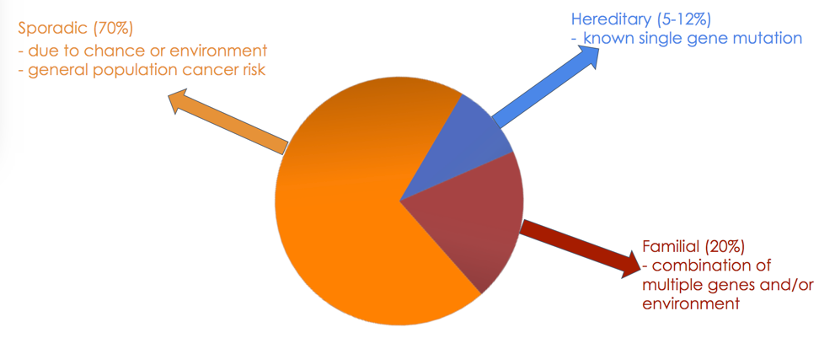
Dr. Eggener notes that why we test prostate cancer patients is because all cancer develops from genetic abnormalities, yet a relatively small percentage are hereditary (inherited via genes). However, somewhat surprisingly, prostate cancer appears to have the highest heritability among all cancers. Sporadic cancers include normal cells that have genetic abnormalities and subsequently develop cancer, whereas hereditary cancers include cells that are predisposed to causing cancer. Looking at the etiology of prostate cancer, ~70% are sporadic, 20% are familial, and 5-12% are hereditary:
Interestingly, only a small proportion of heritable prostate cancers have an identifiable germline mutation, including 12% of men with metastatic disease, and 5% of those with high-risk localized disease. Data from Pritchard and colleagues1 suggests that BRCA2 and CHEK2 are overexpressed in metastatic and localized disease.
So, who should we test and who is the appropriate patient for germline testing? According to Dr. Eggener, this includes patients that are:
- Younger age at diagnosis
- High-grade disease (Gleason score ≥8)
- Multiple generations with cancer
- Multiple family members with the same cancer (ie. prostate/breast/ovarian/pancreatic)
- Ashkenazi Jewish or African ancestry
According to the National Comprehensive Cancer Network (NCCN) guidelines, testing is recommended for patients with prostate cancer and any of the following features: (i) high-risk, very high-risk, regional, or metastatic prostate cancer, (ii) Ashkenazi Jewish ancestry, (iii) family history of high-risk germline mutations (eg. BRCA1/2 Lynch mutation, etc), and (iv) those with a compelling family history, such as a brother or father or multiple family members with prostate cancer at <60 years of age or those that died of prostate cancer. In 2017, the Philadelphia Prostate Cancer Consensus Conference was held to discuss the role of genetic testing for inherited prostate cancer risk, a comprehensive report that helps to guide decision making.2
Dr. Eggener notes that many men learn of their risk of prostate cancer from a female relative. Those with ovarian or breast cancer in their family often have genetic testing as a part of their workup. This is important as the majority of the general population does not understand that a BRCA1 or BRCA2 mutation also denotes a higher risk of prostate cancer.
There are several ways to test patients, including narrow gene panels (5-6 genes), prostate cancer-specific panels (10-15 genes), or broad panels (~80 genes) that test for many cancers. Germline testing for prostate cancer testing should include homologous recombination genes, which include BRCA1, BRCA2, ATM, PALB2, and CHEK2. When testing for Lynch syndrome, this should include MLH1, MSH2, MSH6, and PMS2. Additionally, HOXB13 is present in ~1% of men that develop prostate cancer but to date is not associated with aggressive disease. Germline testing can be obtained via blood, saliva, or buccal swabs and varies in the quality of companies and prices. It is often, but not always, covered by insurance, with a minimum price in the US of $150-$200. If the germline test is positive, there is no need to test the cancer (somatic) since all cells in the body will have the mutation.
There may be certain patients that you would consider somatic testing of cancer tissue, which requires a biopsy, preferably of a metastatic site. If a somatic biopsy is performed, this panel should include BRCA1, BRCA2, ATM, PALB2, FANCA, RAD51D, CHEK2, and CDK12 in patients with metastatic prostate cancer and men with non-metastatic prostate cancer. If the somatic testing finds mutations in BRCA1, BRCA2, ATM, PALB2, and CHEK2 mutations, you may want to consider confirmatory germline testing, according to Dr. Eggener. Tumor testing for MSI-H or dMMR can be considered in patients with regional or castration-naïve prostate cancer and this is also recommended in all patients with metastatic castration-resistant prostate cancer (mCRPC). If MSI-H or dMMR mutations are found, a referral should be made for genetic counseling to assess for Lynch syndrome and possibly for pembrolizumab treatment. Somatic testing may be repeated with progression following treatment, as tumors may have induced or required mutations over time.
Clinically, if a patient has a known germline mutation, it may be associated with a higher incidence of prostate cancer, more aggressive disease, an undetermined risk of prostate cancer and/or an elevated risk of other cancers. Testing may also result in no mutation identified, which Dr. Eggener states is by far the most common result. Furthermore, the result may be a variant of uncertain significance, whereby a genetic change was found, which may or may not increase the risk of cancer, but the result is neither positive nor negative.
If a patient has a known mutation but no diagnosis of cancer, this may certainly modify screening frequency/intensity, may lead to lifestyle modifications, risk-reducing measures, and screening for additional cancers. If you have been diagnosed with cancer and have a known germline mutation, it may impact surveillance and treatment, use of targeted therapies, and screening for additional cancers. Additionally, in men with a positive germline mutation, it is encouraged that this knowledge be shared with other family members, allowing screening for additional cancers, as well as diet and lifestyle modifications.
Looking in more detail at specific genes, alterations in BRCA2, ATM, and possibly CHEK2, are associated with not only increased incidence but also increased aggressiveness of disease. Genes associated with an increased incidence (but not aggressiveness) include BRCA1, HOXB13, MSH2, MSH6, MLH1, and PMS2. In a cohort of 1,211 men from Johns Hopkins Hospital Among on active surveillance, 289 patients experienced grade reclassification.3 There were 11 of 26 with mutations in a three-gene panel, which was associated with an adjusted hazard ration of 1.96 (95% confidence interval [CI] 1.004-3.84) for grade reclassification. Furthermore, reclassification occurred in six of 11 carriers of BRCA2 mutations and 283 of 1200 noncarriers (adjusted hazard ratio [HR] 2.74, 95% CI 1.26-5.96, p=0.01):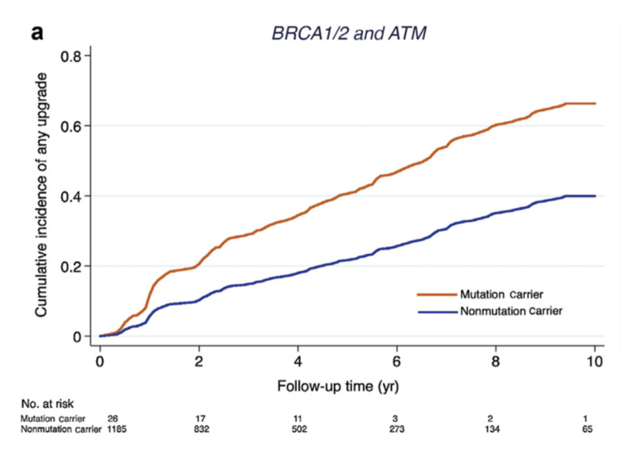
Dr. Eggener notes that there is anxiety for the patient and their family members, as family members may become upset by learning about a genetic risk. There can also be frustration or confusion with a lack of specific information, as it is often unclear as to the information or recommendations. It is often unknown whether results may impact your ability to get life insurance, disability insurance, or long-term care insurance.
Dr. Eggener concluded his presentation by discussing the genetic counseling of prostate cancer patients with the following take-home messages:
- We are in the early stages of understanding prostate cancer heritability
- It is important to ask patients about their personal and family history
- If you do testing, you will identify mutations to directly help men and their families
Presented by: Scott Eggener, MD, Bruce and Beth White Family Professor of Surgery and Radiology, Vice Chair, Section of Urology, Director, High Risk and Advanced Prostate Cancer Clinic, University of Chicago, Chicago, Illinois
Written by: Zachary Klaassen, MD, MSc, Assistant Professor of Urology, Georgia Cancer Center, Augusta University/Medical College of Georgia, Augusta, Georgia, Twitter: @zklaassen_md at the 2020 Société Internationale d'Urologie Virtual Congress (#SIU2020), October 10th - October 11th, 2020
References:
1. Pritchard, Colin C., Joaquin Mateo, Michael F. Walsh, Navonil De Sarkar, Wassim Abida, Himisha Beltran, Andrea Garofalo et al. "Inherited DNA-repair gene mutations in men with metastatic prostate cancer." N Engl J Med 375 (2016): 443-453.
2. Giri, Veda N., Karen E. Knudsen, William K. Kelly, Wassim Abida, Gerald L. Andriole, Chris H. Bangma, Justin E. Bekelman et al. "Role of genetic testing for inherited prostate cancer risk: Philadelphia Prostate Cancer Consensus Conference 2017." Journal of Clinical Oncology 36, no. 4 (2018): 414.
3. Carter, H. Ballentine, Brian Helfand, Mufaddal Mamawala, Yishuo Wu, Patricia Landis, Hongjie Yu, Kathleen Wiley et al. "Germline mutations in ATM and BRCA1/2 are associated with grade reclassification in men on active surveillance for prostate cancer." European urology 75, no. 5 (2019): 743-749.

