High risk tumors in prostate cancer have at least seven different definitions, beginning from the first definition created by D’Amico in 1998 and published in JAMA, to the most recent definition described in the EAU guidelines in 2018. In recent years there has been an increasing use of radical treatment in the setting of locally advanced and very high-risk prostate cancer, as can be seen in figure 1.
Figure 1 – Increasing use of radical treatment for high risk prostate cancer:
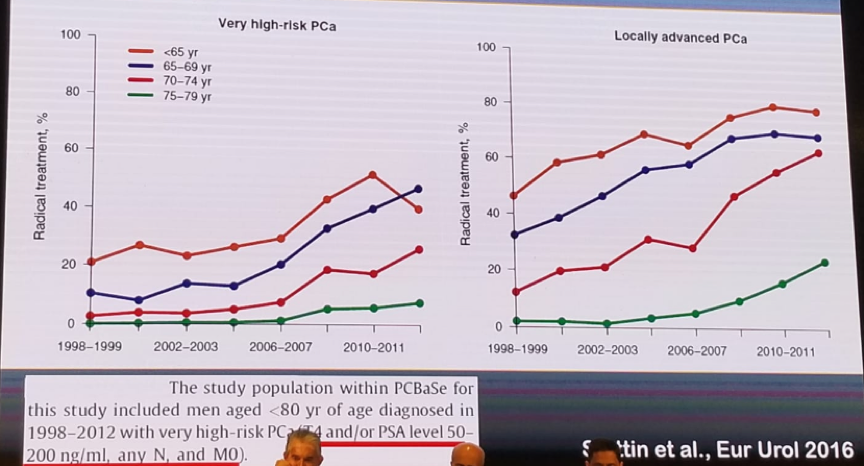
There has also been an increase in the usage of mpMRI prior to surgery to try and predict extracapsular extension. The reported sensitivity and specificity of mpMRI in this setting has been shown to be 92.9% and 66.9%, respectively.1 In addition, there has been an increase in the usage of fusion biopsies, demonstrating that the best results are obtained when both systematic and fusion biopsies are performed concomitantly.2
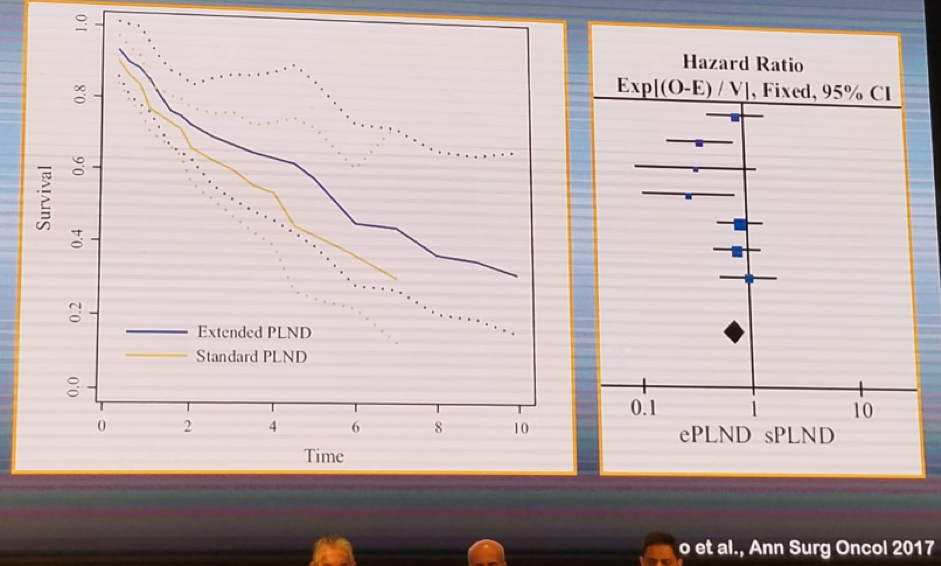
When treating high risk-prostate cancer, several additional controversial issues need to be addressed. One of these controversaries include whether standard or extended pelvic lymph node dissection should be performed. There is some data showing a survival benefit for patients undergoing an extended pelvic lymph node dissection, as shown in figure 2.
Figure 2 – Survival outcomes in standard vs. extended pelvic lymph node dissection in radical prostatectomy:
The number of involved lymph nodes has a profound effect on the survival outcomes of patients undergoing radical prostatectomy. Data from Dr. Wirth’s institution in Dresden, Germany, clearly showed a long-term survival advantage for patients undergoing radical prostatectomy with node negative disease, compared to patients with node-positive disease (Figure 3).
Figure 3 – Comparison of long-term mortality in patients undergoing radical prostatectomy with node negative and node-positive disease:

Concordance exists between the functional outcomes of these patients to the tumor risk category, with higher risk disease having lower functional scores.3
The next topic discussed was whether adjuvant or salvage radiotherapy should be given to these high-risk patients, following surgery. Data published in 2012 has shown that early adjuvant treatment confers an advantage in biochemical progression free survival, as shown in figure 4.
Figure 4 – Postoperative radiotherapy for high risk disease:

Another topic discussed was the comparison between radical prostatectomy and radiotherapy. Data from the national prostate cancer registry from Sweden, demonstrated a benefit for radical prostatectomy in grade group 1-3 disease.4 A similar comparison between radical prostatectomy, radiotherapy and hormonal therapy from the CaPSURE database has also demonstrated an advantage to surgery over radiotherapy and hormonal treatment.5 On a side note, radiotherapy has been shown to increase the risk of secondary cancers following treatment for prostate cancer with an odds ratio of 2.08 (1.48-2.91), p=0.0001.6
In a systematic review and meta-analysis analyzing the effect of neoadjuvant hormonal therapy prior to radical prostatectomy, no significant evidence had been presented to show an advantage of neoadjuvant hormonal treatment.7 Therefore, there is currently no role for neoadjuvant hormonal therapy prior to surgery. However, in a randomized prospective trial, flutamide given to patients with pT3-4N0 disease after radical prostatectomy, has been shown to improve recurrence free survival but not overall survival, as seen in Figure 5.
Figure 5 – Randomized controlled trial comparing placebo to Flutamide after radical prostatectomy in patients with pT3-T4 N0 disease:

When radiotherapy is given to high-risk patients, long term hormonal therapy (for 3 years) has been shown to increase survival, with a hazards ratio of 0.6 (95% C.I. 0.45-0.8), p=0.0004.8 However, there has been a recent publication in the European Urology journal, demonstrating that for high-risk patients, hormonal therapy for 18 months instead of 36 months confers better biochemical failure and similar disease-free survival rates, as shown in figure 6.
Figure 6 – Comparison of 18 months vs. 36 months of hormonal therapy with radiotherapy for high-risk disease prostate cancer:

The last topic discussed was the role of surgery in oligo-metastatic disease. There have been some retrospective analyses, including a population-based SEER study, demonstrating a benefit for oligo-metastatic patients undergoing cytoreductive radical prostatectomy. 9,10 Furthermore, studies assessing the effect of radiotherapy to the prostate in oligometastatic patients have also shown a benefit to this cytoreductive treatment in both biochemical free survival and overall survival.11 However, in a systematic review of these studies, published in 2018, the authors concluded that although local treatment in metastatic prostate cancer appears to be feasible, its oncological effect remains unclear due to high susceptibility of available studies to significant selection bias.12
Dr. Wirth concluded his talk emphasizing some key points. First, radical prostatectomy or radiotherapy with hormonal therapy confer promising results for high-risk prostate cancer patients. Second, adjuvant or early salvage radiotherapy after surgery improves disease control and offers a second chance of cure. Third, radiotherapy might confer an increased risk of secondary malignancies, while radical prostatectomy does not. Fourth, the role of cytoreductive local treatment in oligo-metastatic disease is unclear and there is a desperate need for randomized controlled trials in this setting, before clear answers can be given.
Presented by: Manfred Wirth, Dresden, Germany
References:
- Matsuoka et al. AJR 2017
- Borkwertz, Wirth et al. AUA 2018
- Jasson et al. Eur Urol 2018
- Wiklund et al. BMJ 2014
- Cooperberg et al. Cancer 2010
- Nam et al. Lancet Oncol 2014
- Shelley et al. Cancer Treat Rev 2009
- Bolla et al. Lancet Oncol 2010
- Gratzke et al. Eur Urol 2014
- Culp et al. Eur Urol 2014
- Cho et al. Plos One 2016
- Tilki D et al. Int. J. of Urol 2018


