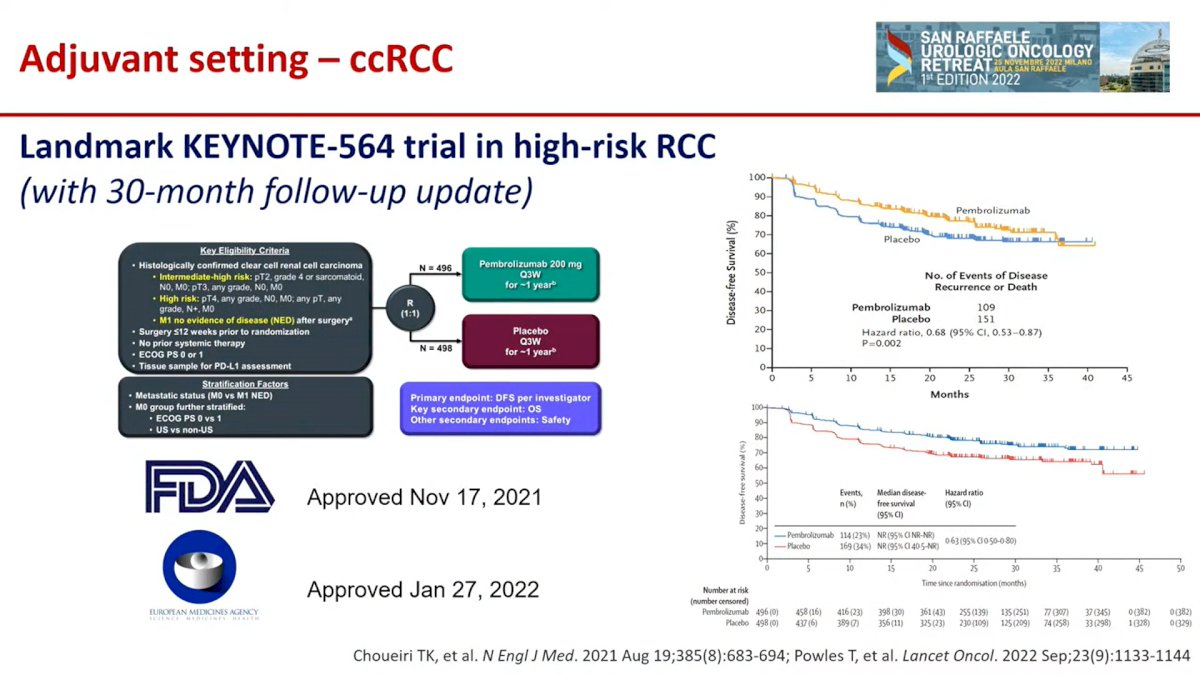
(UroToday.com) During the session dedicated to ongoing clinical trials at San Raffaele University Hospital, chaired by Prof. Andrea Necchi, more than 40 ongoing trials were shown. Dr. Marandino presented the ongoing clinical trials in Kidney cancer. She began her talk by speaking about the results of Keynote-564 which “established a new adjuvant standard of care”, said. Dr. Marandino while underlined one of the hot topics discussed by other speakers during this Meeting: geographic discrepancy to treatments. Pembrolizumab is approved in the adjuvant setting of intermediate/high risk or M1 NED clear cell renal cell carcinoma (ccRCC) by the FDA and EMA, but it is not yet reimbursed in Italy.
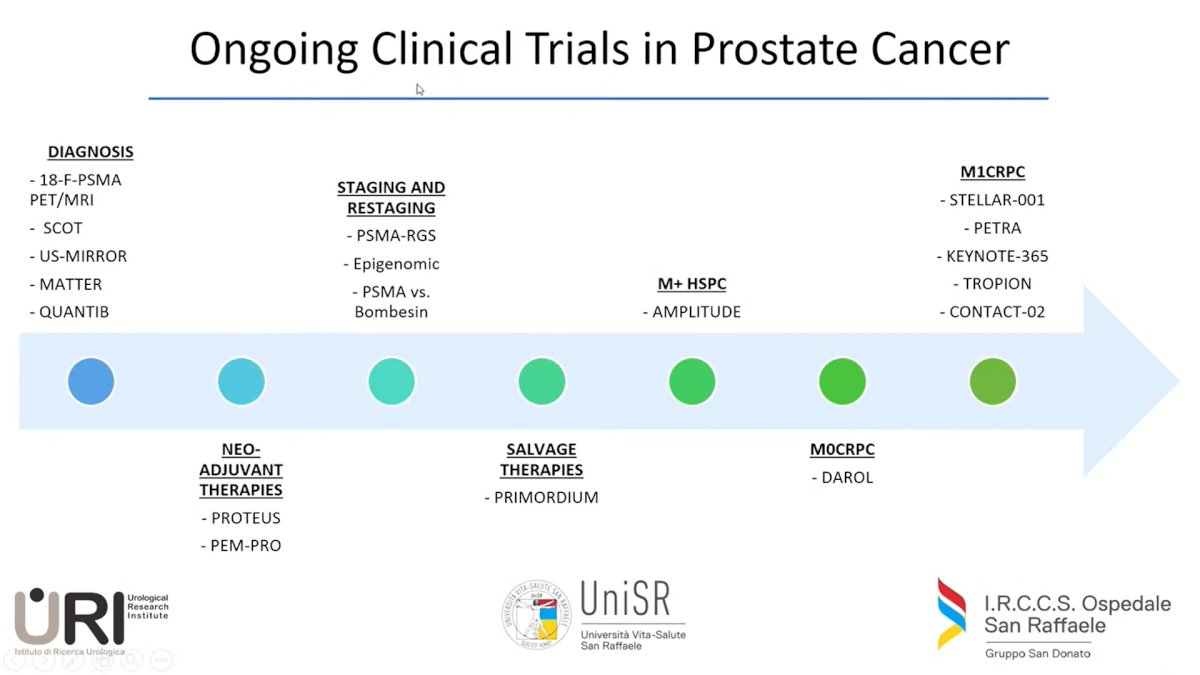
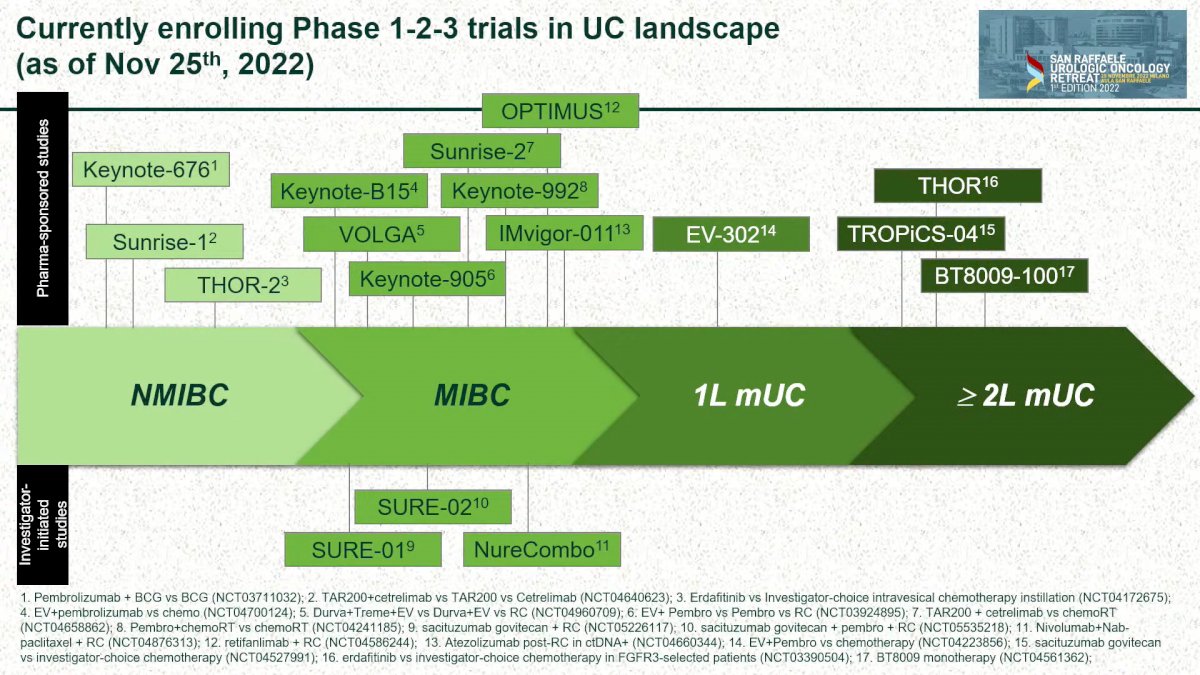
Regarding Prostate Cancer (PCa), Dr. Gandaglia showed the design of the 18 trials ongoing, ranging from the diagnosis setting to the mCRPC setting (figure 1)
Worth mentioning the initial results of a prospective single-arm study aiming to identify the optimal biopsy scheme at MRI target biopsy in men with a suspicion of PCa. In the initial results presented, the majority of csPCa were found with the first TBx core. Dr. Gandaglia showed also that according to these initial findings 12 SBx cores seem to be sufficient to diagnose up to 90% of csPCa.

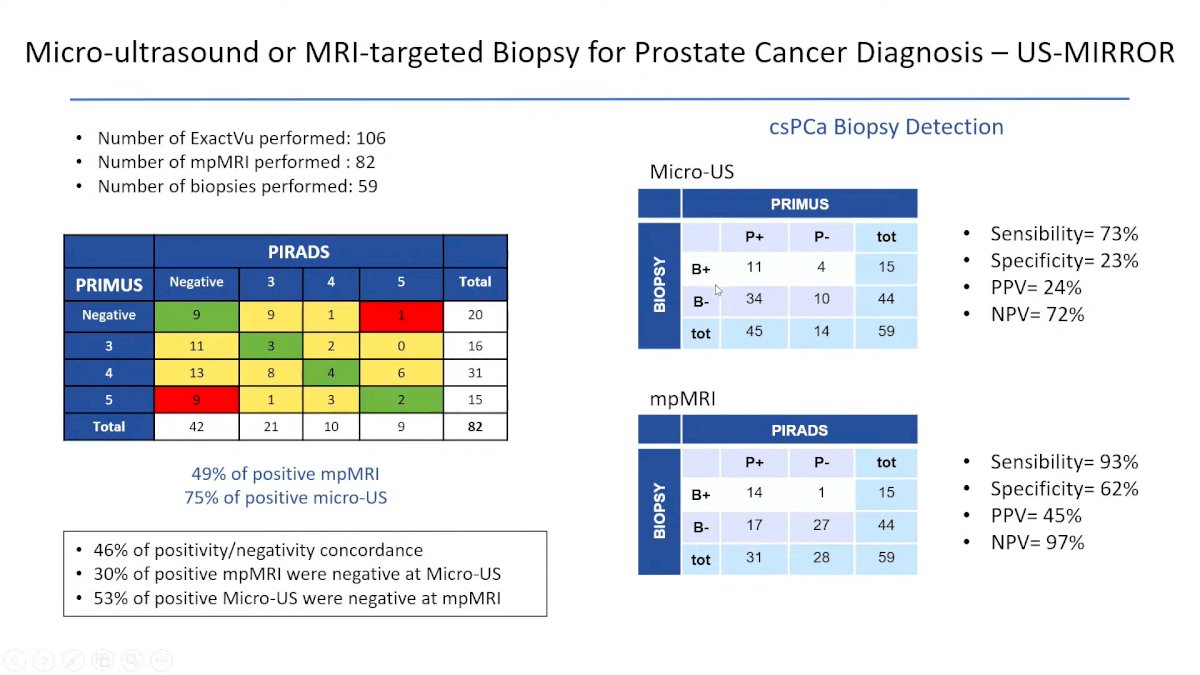
In another study, Micro ultrasound was compared to MRI targeted biopsy for PCa Diagnosis, and according to the data presented mpMRI has a higher sensibility and specificity compared to Micro-US
The 17 ongoing clinical trials in bladder cancer, were presented by Dr. Raggi.
Among all the ongoing trials, worth to mention three promising academic clinical trials ongoing in the setting of Muscle-invasive bladder cancer (MIBC). SURE-01 is a single-arm phase II study for cT2-4cN0M0 CT-naïve patients with MIBC in which 4 cycles qw3 of Sacituzimab Govitecan (SG) are administered before radical cystectomy (RC) followed by adjuvant standard of care management. The SURE-02 study has the same setting, but a different neoadjuvant and adjuvant management. In this study, SG is associated with Pembrolizumab and administered every 3 weeks per 4 cycles before cystectomy. After RC patients received Pembrolizumab as adjuvant treatment for 12 months, with progression or unacceptable toxicity. For both of the studies, the primary endpoint is a pathological complete response, while OS safety and tolerability are secondary endpoints.

The third academic study presented in this setting was the NURE-Combo trial, an open-label, single-arm, phase II study evaluating Nivolumab flat dose 360 mg + Nab-paclitaxel 125 mg/mq every 3 weeks for 4 cycles before RC as neoadjuvant treatment and adjuvant nivolumab flat dose 360 mg IV every 3 weeks up to 13 cycles in patients with cT2-4cN0M0 Urothelial Carcinoma of the bladder. The primary endpoint is a pathological complete response, while OS safety and tolerability are secondary endpoints.

For the first time ever the first interim and safety analyses of this trial were presented: of the first 13 patients,61.5% had a cT2N0 disease, and 38.5% (5) had a cT3N0 stage disease. About the efficacy data, based on the first 11 patients, 27.3% (3) of patients achieved a complete pathological response, 72.7% (8) achieved a pathological downstaging to pT<2

All the patients underwent radical cystectomy, 2 patients suspended nivolumab in the adjuvant phase due to TRAEs, and one patient suspended Nab-paclitaxel on cycle 4 during the neoadjuvant phase. 1 death occurred, after the cystectomy but was not related to the experimental drug combination.

- Giorgio Gandaglia, MD Research Fellow, Urology San Raffaele University Hospital, Milan
- Laura Marandino, MD, Medical Oncology at San Raffaele University Hospital, Milan
- Daniele Raggi, MD, Medical Oncology at San Raffaele University Hospital, Milan
Written by: Emanuele Crupi, MD, Resident in Medical Oncology at San Raffaele University Hospital, Milan, @emanuele_crupi on Twitter during the 1st San Raffaele Urologic Oncology Retreat, Friday, Nov 25, 2022


