(UroToday.com) The eighth session of the 2022 International Kidney Cancer Symposium (IKCS): North America meeting focused on regulatory considerations for new trials in advanced renal cell carcinoma (RCC). In this context, Laura Wood provided a perspective based on 30 years as a clinical trials nurse.
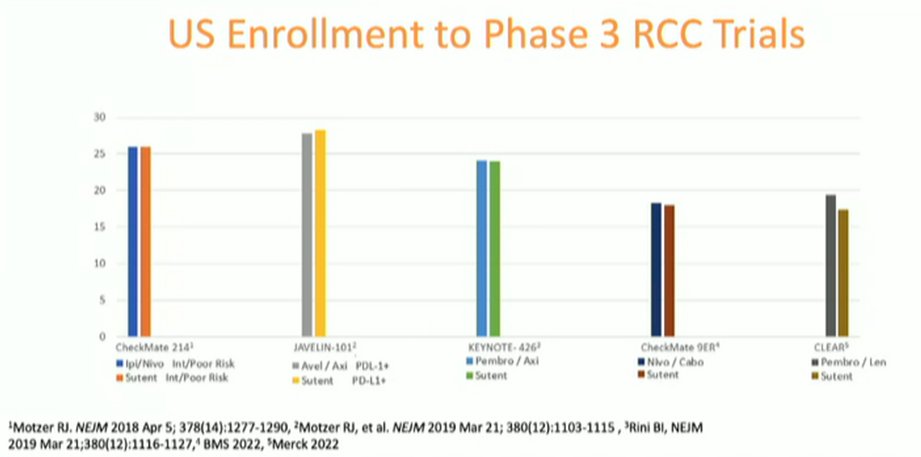
Ms. Wood began by noting that the 2021 Oncology Center of Excellence Report highlighted that the proportion of patients enrolled in phase 3 RCTs supporting approval of combination immunotherapy regimes in advanced kidney cancer was “very small”, with less than 30% of the enrolled population in any given trial.
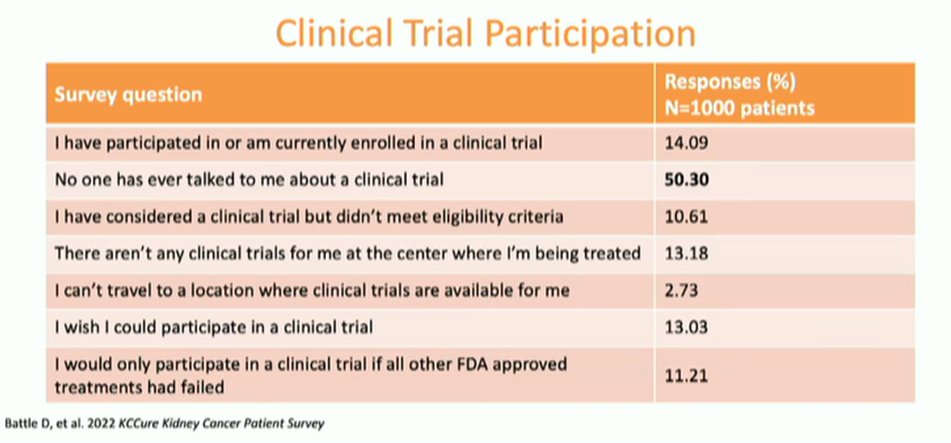
Considering why patients may not be being enrolled on trials, Ms. Wood discussed a number of patient or caregiver barriers to clinical trial enrollment, including awareness, education, access, financial considerations, interest, or the perception of the risks and benefits. She highlighted a relatively recent patient survey conducted by KCCure which noted that the majority of patients with kidney cancer had never had a clinical trial discussed with them.
Ms. Wood further discussed financial considerations regarding clinical trial participation. A large portion of this comes from uncertainty – it is often unclear the extent of investigational care costs including the co-pay for “approved” agents that may be part of the protocol as well as those related to additional lab and radiological testing that is required in the context of the trial but may also be considered as standard of care. Further, trials may be associated with increased routine-care costs and non-medical costs on account of increased visits and increased durations of visits. Additionally, clinical trial participation may necessitate larger commitments in terms of caregiver support and dependent care costs. Recently, ASCO has released a policy statement addressing financial barriers to patient participation in clinical trials which emphasized the importance of improving the policy environment for coverage of clinical trials, facilitating transparency for trial-related out-of-pocket costs, refuting the specter of inducement, and improving available data on costs of cancer clinical trials.
From an institutional perspective, Ms. Wood noted that a lack of awareness may represent a barrier to trial enrollment. However, there is an acknowledgment that trial enrollment may be associated with “patient leakage” with patients leaving a given health system’s network of care. This may have financial implications for the network and induce them not to recommend trials for their patients.
As we look to minimize barriers to clinical trial enrollment, she highlighted the importance of extending trials to rural communities but reducing the burden of access and availability as well as non-medical costs. Additionally, we must consider modifying stringent eligibility criteria that may exclude patients who would be otherwise eligible.
In the context of trial enrollment, she noted the importance of shared decision making. This includes an explanation of what clinical trials are and why one would consider participating. However, potential downsides including more frequent visits, tests, co-pays, and out-of-pocket expenses should be clearly enunciated. Putting this together, she noted that it is important to assess a patient’s interest, feasibility, and eligibility in clinical trials.

Ms. Wood emphasized the importance of both patient and caregiver education regarding what clinical trials entail, why one may want to participate, and how this may be made feasible. Acknowledging the barriers to having these discussions in detail in the clinic context, she highlighted a number of resources that may be useful including the KCA, the Lazarex Foundation, and the National Cancer Institute.
In closing, Ms. Wood noted that patients and caregivers want to know about clinical trials. Thus, it is important that we include clinical trials in shared decision-making and provides information and resource that may improve access and feasibility to participate.
Presented by: Laura S. Wood, RN, MSN, OCN, Ret.; Cleveland Clinic Taussig Cancer Center


