Dr. Nguyen notes that high-quality radiation therapy for high-risk disease needs long-term ADT and a brachytherapy boost. Dr. Nguyen doesn’t take the addition of ADT likely as there are several bothersome and worrisome side effects of ADT:

Work from Dr. Nguyen and his team using SEER-Medicare data suggest that depression risk is increased with longer duration ADT: <6 months ADT use (HR 1.12); 6-12 months ADT use (HR 1.26); >12 months ADT use (HR 1.37)1. The literature assessing ADT and cardiovascular death has historically been inconclusive, however, a meta-analysis of RCTs performed by Dr. Nguyen demonstrated no association: overall fixed effects model, HR 0.93 (95%CI 0.79-1.10)2. Furthermore, ADT reduces prostate-cancer-specific mortality (RR 0.69; 95%CI 0.56-0.84) and all-cause mortality (RR 0.86; 95%CI 0.80-0.93).
Despite the advantages of ADT in high-risk prostate cancer management, it is underused: 23% of high-risk patients receive no ADT. Older age and brachytherapy boost are associated with non-ADT use. Furthermore, nearly half of patients receiving ADT are planned for <2 years and nearly half of those planned for 2 years don’t get the full 2 years of treatment. So, the question is “how long is enough” for ADT when combined with radiotherapy? Data presented at GU ASCO suggests that 18 months is better than 6 months (PCSM HR 0.70, 95%CI 0.50-0.97 favoring 18 months); a 29% reduction in distant progression was the main driver of the 30% reduction in PCSM according to Dr. Nguyen. Updated data from the PCS IV trial testing 18 vs 36 months of ADT in combination with radiation therapy suggest there is no difference between these treatment durations (HR 1.02, 95%CI 0.81-1.29)3. Although biochemical recurrence was worse in the 18-month treatment arm (HR 0.714, 95%CI 0.532-0.952), interestingly there was non-compliance in the 36 month ADT arm biasing the results towards equivalence. Based on this data, some centers are adopting 18 months of ADT, although the NCCN still recommends 2-3 years of ADT for high-risk disease.
Dr. Nguyen also feels strongly that high-quality treatment should include a brachytherapy boost. A Canadian RCT of 398 patients with high-risk disease were randomized to pelvic RT to 46 Gy + 1 year of ADT + prostate EBRT 32 Gy vs pelvic RT to 46 Gy + 1 year of ADT + brachytherapy boost (LDR 115 Gy). Data presented at the 2015 American Brachytherapy Society annual meeting suggests that patients receiving a brachytherapy dose escalation have a significantly improved biochemical PFS compared to low-dose therapy; this is particularly true for patients with NCCN high-risk disease:
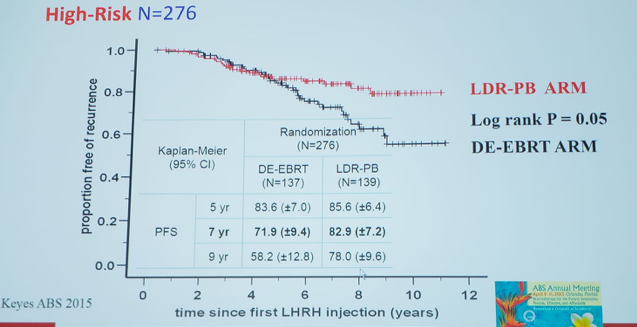
There was a significantly higher rate of Grade 3 GU toxicity in the brachytherapy arm, otherwise, there was no difference in GU Grade 4 toxicity or GI toxicity.
Historically there has been controversy about surgery vs radiation for high-risk prostate cancer. A meta-analysis by Wallis et al.4 found radiation therapy associated with an increased risk of prostate cancer-specific death (HR 2.08) and increased risk of overall death (HR 1.68). As Dr. Nguyen notes, very few of these patients in those studies had high-quality radiation therapy (long-term ADT and brachytherapy boost). Last year a JAMA retrospective study assess the clinical outcomes of patients with Gleason score 9-10 cancer after definitive treatment5. The authors found that adjusted 5-year prostate cancer-specific mortality rates were radical prostatectomy, 12% (95%CI, 8%-17%); EBRT, 13% (95%CI, 8%-19%); and EBRT + brachytherapy boost, 3% (95%CI, 1%-5%). EBRT + brachytherapy boost was associated with significantly lower prostate cancer-specific mortality than either RP or EBRT (cause-specific HRs of 0.38 [95% CI, 0.21-0.68] and 0.41 [95% CI, 0.24-0.71]). These data cautiously suggest that patients with high-risk prostate cancer may have a clinical advantage when treated with EBRT + brachytherapy.
Dr. Nguyen thinks that the future directions for high-risk radiotherapy treatment include using genomics to select patients for personalized de-intensification vs intensification therapy. In fact, clinical-genomic risk groups improve the C-index over NCCN/D’Amico groups by 20-40%6. Because of this, the genomic clinical risk stratification will be used to stratify the next US national randomized trial for high-risk prostate cancer (n=2400).
Dr. Nguyen concluded this talk on RT for the treatment of high-risk disease with the following arguments:
- High quality RT for high risk prostate cancer should include long-term ADT and brachytherapy boost
- High-quality RT and surgery plus appropriate post-op therapy appear to have similar survival outcomes
- A future trial is planned using genomics to intensify/deintensify treatments of RT-managed high-risk prostate cancer.
Written by: Zachary Klaassen, MD, MSc – Assistant Professor of Urology, Georgia Cancer Center, Augusta University/Medical College of Georgia, Twitter: @zklaassen_md, at the 16th Meeting of the European Section of Oncological Urology, #ESOU19, January 18-20, 2019, Prague, Czech Republic
References:
- Dinh KT, Reznor G, Muralidhar V, et al. Association of Androgen Deprivation Therapy With Depression in Localized Prostate Cancer. J Clin Oncol 2016 Jun 1;34(16):1905-1912.
- Nguyen PL, Je Y, Schutz FA, et al. Association of androgen deprivation therapy with cardiovascular death in patients with prostate cancer: a meta-analysis of randomized trials. JAMA 2011 Dec 7;306(21):2359-2366.
- Nabid A, Carrier N, Martin AG, et al. Duration of Androgen Deprivation Therapy in High-risk Prostate Cancer: A Randomized Phase III Trial. Eur Urol 2018 Oct;74(4):432-441.
- Wallis CJD, Saskin R, Choo R, et al. Surgery Versus Radiotherapy for Clinically-localized Prostate Cancer: A Systematic Review and Meta-analysis. Eur Urol 2016 Jul;70(1):21-30.
- Kishan AU, Cook RR, Ciezki JP, et al. Radical Prostatectomy, External Beam Radiotherapy, or External Beam Radiotherapy With Brachytherapy Boost and Disease Progression and Mortality in Patients With Gleason Score 9-10 Prostate Cancer. JAMA Mar 6;319(9):896-905.
- Spratt DE, Zhang J, Santiago-Jimenez M, et al. Development and Validation of a Novel Integrated Clinical-Genomic Risk Group Classification for Localized Prostate Cancer. J Clin Oncol 2018 Feb 20;36(6):581-590.
What is High Quality Local Treatment in High Risk Localized Prostate Cancer? Surgery

