(UroToday.com)The 2023 European Society of Medical Oncology (ESMO) Annual Congress held in Madrid, Spain between October 20th and 24th, 2023 was host to a neoadjuvant and adjuvant therapy in genitourinary cancers session. Dr. Fabio Schutz discussed global access consideration in the systemic treatment of genitourinary cancers.
From an epidemiologic standpoint, the incidence of kidney and bladder cancer is much higher in ‘developed’ countries such as those in North America and Europe. Conversely, we see that the cancer-associated mortality rates in these countries are comparatively lower. Is this discordance a result of the ‘developed’ countries having better registries, better access to care in these countries, or both?
This discordance is not limited to geographic regions but also is clearly influenced by economic factors, with similar trends observed (increased incidence, relatively lower mortality) observed in the higher income regions/countries as well.
The incidence of RCC is increasing across all geographic regions in a relatively consistent manner. However, there are geographically heterogeneous trends in RCC-related mortality. In the graph below, we clearly see that the blue bars (RCC incidence) exceed 0, indicating an increase in incidence globally. However, in the orange bar graph below, we see that RCC-related mortality is decreasing in North American, North European, and Western European countries. Conversely, it is increasing in Central/South American and Southern European countries.1 Dr. Schutz noted that this is likely due to disparities in access to systemic treatment options for RCC.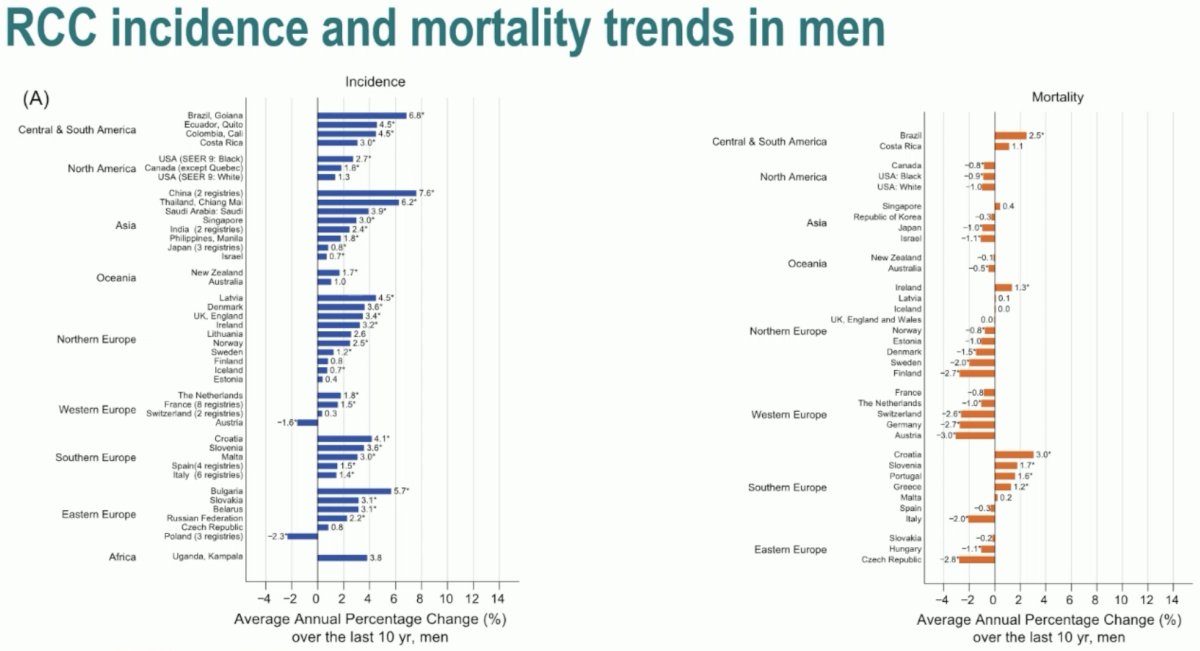
This mortality pattern is consistent with data on total health expenditure per capita, provided by the WHO. A seen below, the highest figures are observed in the North American and Northern/Western European countries. This is likely being translated into improved access to healthcare in these countries.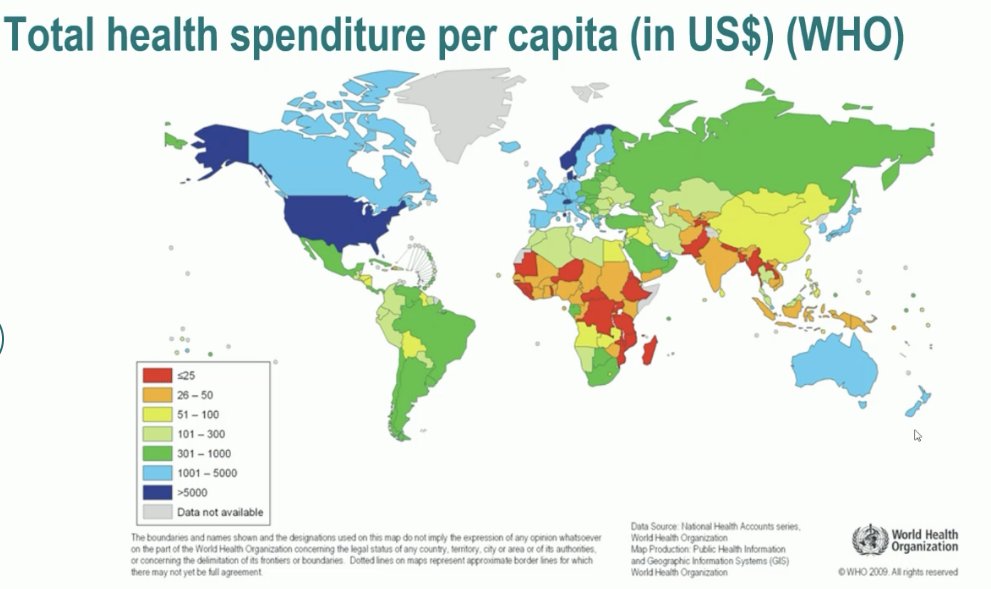
There is a known exponential relationship between total health expenditure and life expectancy, with countries spending more per capita deriving improved life expectancies. One notable outlier though is the United States, with the highest expenditure per capita of any country, but with a relatively lower life expectancy per capita expenditure compared to other countries such as Norway and The Netherlands.
There are major differences in the healthcare systems across different countries and regions. The United States mainly relies of private healthcare insurance companies, which usually offer excellent access to services and new technologies. In Europe, the UK, Sweden, and Norway are notable examples of well-established public healthcare systems. Germany, The Netherlands, and Switzerland rely on a social health insurance system, contributed for by individuals and employers, with private health insurances acting as supplementary coverage for additional services. In Eastern European countries, private health insurances are more commonly used, as public healthcare systems may be less well developed.
Another important aspect of healthcare delivery is drug approval by regulatory agencies. There are discrepancies in these approvals across regions, which impact the availability of these drugs accordingly. For example, adjuvant nivolumab, based on the results of CheckMate 274,2 is approved for all patients in the USA (FDA) and Brazil, but only for PD-L1 positive tumors in Europe by the European Medical Agency. Another example is the 1st line mCRPC combination of olaparib + abiraterone (PROpel)3, which is approved in the US for BRCA1/2 positive patients, whereas it is approved for all metastatic castration resistant prostate cancer (mCRPC) patients in the 1st line, even those without homologous recombination repair genes.
What about geographic variations in access to novel therapies in the context of clinical trials? The map below shows geographic variations in the number of clinical trials worldwide, per data from ClinicalTrials.gov. Again, we see the highest number of ongoing trials in the North American and Northern/Western European countries.
Specifically, how does the USA compare to the rest of the world with regards to clinical trials? The discrepancy is most pronounced for phase 1 trials, with ~85% of those trials worldwide being conducted in the United States. This discrepancy decreases with phase 2 and 3 trials, although remains disparately higher in the United States, with >60% of phase 3 trials worldwide conducted there.
Are clinical trials the ‘great neutralizers’ of health care disparities? How do patient outcomes compare across geographic regions when patients are treated within the context of clinical trials? Based on global data published by Fay et al. in 2018, it appears that these patients’ outcomes are similar across different regions, income levels, and life expectancies, highlighting the importance of access to clinical trials for our patients.
What about the healthcare economic burden? Dr. Schutz noted that this has not always been a priority for pharmacologic companies. Going back to the phase 1 CheckMate-003 established nivolumab dosing at 3 mg/kg and the KeyNote-001 established pembrolizumab dosing at 2 mg/kg, we witnessed somewhat increasing responses with higher doses.
However, upon pharmacodynamic assessment, there was no association between dose and PD-1 or receptor occupancy. Additionally, the objective response probability seems to be more associated with clearance than with the administered dose. Thus, Dr. Schutz argued that we could be ‘smarter’ with our dosing of these drugs using lower doses to decrease costs, particularly in economically disadvantaged countries.
Additionally, a weight-based dosing scheme, as compared to a fixed dose scheme, may help further alleviate costs of systemic treatment across the globe.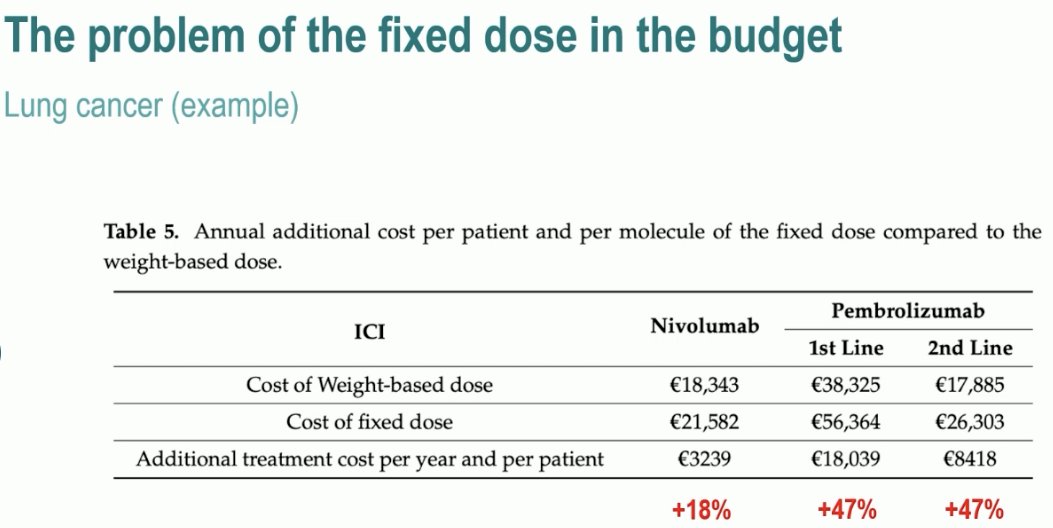
While adjuvant trials are important to potentially prolonging survival in at high-risk populations, this comes at a significant financial cost. In an analysis of CheckMate-274 (nivolumab in MIBC) and KEYNOTE-564 (pembrolizumab in RCC), the cost of averting one DFS event for nivolumab was 240,000 Euros and 890,000 for pembrolizumab within the context of these trials.5
Dr. Schutz concluded his presentation with the following take home messages:
- The incidence and mortality of most genitourinary cancers varies across different countries and regions
- "Higher incidence" in more developed countries may be biased due to higher access to healthcare system and better epidemiology/statistics
- Mortality is proportionally higher in less developed regions
- Access to care is directly associated to the development of the country / region and access to private healthcare insurance in less developed regions
- There is a high economic burden from the newer drugs, especially immune checkpoint inhibitors
- Fixed dose schedules made it even worse
- Clinical trials could be an opportunity for less developed regions
- Need for investment in infrastructure in research sites
- "Win-Win" situation accelerating drug development
Presented by: Fabio A. B. Schutz, MD, Medical Oncology, BP -Beneficência Portuguesa de São Paulo, São Paulo, Brazil
Written by: Rashid K. Sayyid, MD, MSc – Society of Urologic Oncology (SUO) Clinical Fellow at The University of Toronto, @rksayyid on Twitter during the 2023 European Society of Medical Oncology (ESMO) Annual Congress held in Madrid, Spain between October 20th and 24th, 2023
References:- Znaor A, Lortet-Tieulent, Laversanne M, et al. International variations and trends in renal cell carcinoma incidence and mortality. Eur Urol. 2015; 67(3):519-30.
- Bajorin DF, Witjes JA, Gschwend JE, et al. Adjuvant nivolumab versus placebo in muscle-invasive urothelial carcinoma. N Engl J Med. 2021 Jun 3;384(22):2102-2114.
- Clarke NW, Armstrong AJ, Thiery-Vuillemin A, et al. Abiraterone and Olaparib for Metastatic Castration-Resistant Prostate Cancer. N Engl J Med Evid, 2022; 1(9).
- Fay AP, McKay RR, Lin X, et al. Impact of Geographic Regions on Overall Survival in Patients With Metastatic Renal Cell Carcinoma: Results From an International Clinical Trials Database. J Glob Oncol. 2018; 4:1-14.
- van Waalwijk van Doorn-Khosrovani SB, Huisman A, Pisters-van Roy A, et al. Adjuvant immune checkpoint blockade revisited. Lancet Oncol. 2023; 24(7):717-719.


