(UroToday.com) In the second Mini Oral session of the European Society for Medical Oncology (ESMO) Annual Congress focusing on non-prostate genitourinary cancers, Dr. Meylan presented key ancillary analyses of the phase II BIONIKK trial (NCT02960906) aimed at understanding immune markers predictive of the efficacy of immune checkpoint inhibition as first-line therapy in metastatic clear cell renal cell carcinoma (mccRCC). The primary analysis of this trial suggested that ccRCC molecular grouping could enhance response rate to nivolumab, nivolumab + ipilimumab, and VEGFR-TKI in these patients. Building on this, the authors used their comprehensive translational research program to further characterize biomarkers of efficacy for treatment with nivolumab or nivolumab + ipilimumab.
To do so, they used archived paraffin-embedded (FFPE) tumor tissue samples which were collected for all patients within the BIONIKK trial. All of these samples were centrally reviewed by an expert pathologist. Using immunohistochemistry, they quantified main immune populations including T cells (CD3), B cells (CD20), tertiary lymphoid structures (TLS), and markers of immune activation (PD-1/ki67).
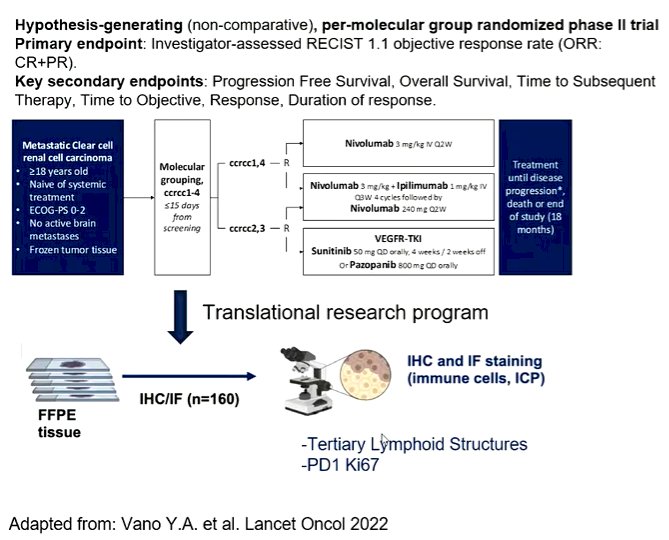
The authors considered three key outcomes: objective response rate (ORR), progression-free survival (mPFS) evaluated by investigator (RECIST1.1) and early progression, defined by progression events occurring before 6 months.
Among the 199 patients in the BIONIKK trial, 160 had available FFPE samples, of whom 42 were treated with nivolumab, 84 were treated with nivolumab + ipilimumab, and 34 were treated with TKI.
Among nivolumab-treated patients, a greater number of tertiary lymphoid structures (TLS) (defined with a threshold >2, representing 33% of the samples) was associated with higher ORR (73% vs 15%, p<0.01), longer mPFS (not reached (IC95%: 8.0-NE months) vs 2.4 months (IC95%: 2.3-8 months), p=0.015) and less early progressions (18% vs 63%, p=0.03).

In patients who received nivolumab + ipilimumab, TLS>2 correlated with response (71% vs 40%, p=0.02) while higher densities of Ki67+PD1+ cells were associated with longer mPFS (16.3 months, IC95%: 10.9-NE, vs 8.25 months (IC95% 4.8-13.8 months, p=0.015), and lower events of early progression (16% vs 41%, p=0.04).

In a combination of TLS (>2) and Ki67/PD1 (>median), the authors were able to identify patients with both high response rates (80% vs 43% p<0.01) and lower early progression events (5% vs 36%, p=0.02).

Thus, the authors conclude that this ancillary analysis of a biomarker driven clinical trial demonstrates that the number of TLS and ki67+PD1+ density likely predict efficacy of both nivolumab and of nivolumab + ipilimumab in frontline mccRCC patients.
Presented by: Maxime Meylan, PhD, Dana-Farber Cancer Institute, Boston, MAWritten by: Christopher J.D. Wallis, University of Toronto Twitter: @WallisCJD during the 2022 European Society for Medical Oncology (ESMO) Annual Congress, 9-13 September 2022.


