(UroToday.com) The 2022 European Society of Medical Oncology (ESMO) annual meeting featured a kidney cancer session, including a discussant presentation by Dr. Tom Powles discussing two key abstracts including “IMmotion010: efficacy and safety from the Phase III study of atezolizumab vs placebo as adjuvant therapy in patients with renal cell carcinoma (RCC) at increased risk of recurrence after resection” presented by Dr. Axel Bex, and “Phase III RandOmized Study Comparing PErioperative Nivolumab versus Observation in Patients with RCC Undergoing Nephrectomy (PROSPER, ECOG-ACRIN EA8143), a National Clinical Trials Network trial” presented by Dr. Mohamad Allaf. Dr. Powles notes that the story to date for immune checkpoint inhibition in clear cell renal cancer has been relatively straight forward:

However, as of ESMO 2022 he notes that the tide is changing and there is now some uncertainty:
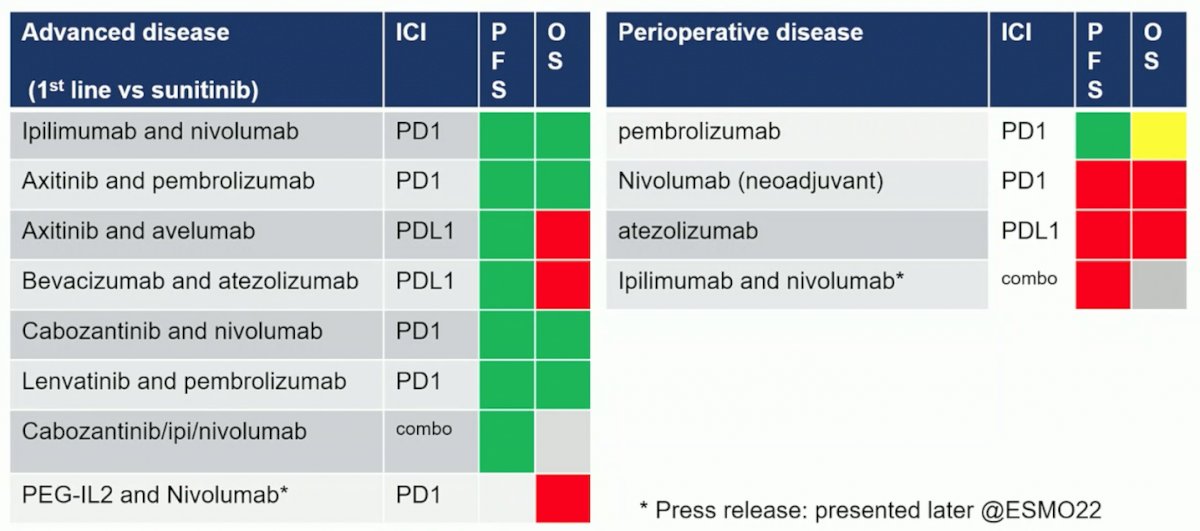
Dr. Powles notes that there are several issues that we need to address from these two perioperative studies presented at ESMO 2022:
- Is the data robust or could this all be explained by chance?
- Are the drugs the same?
- Is there an easy explanation to summarize the findings?
- Have we seen this before with adjuvant VEGF targeted therapy?
- What should we tell our patients?
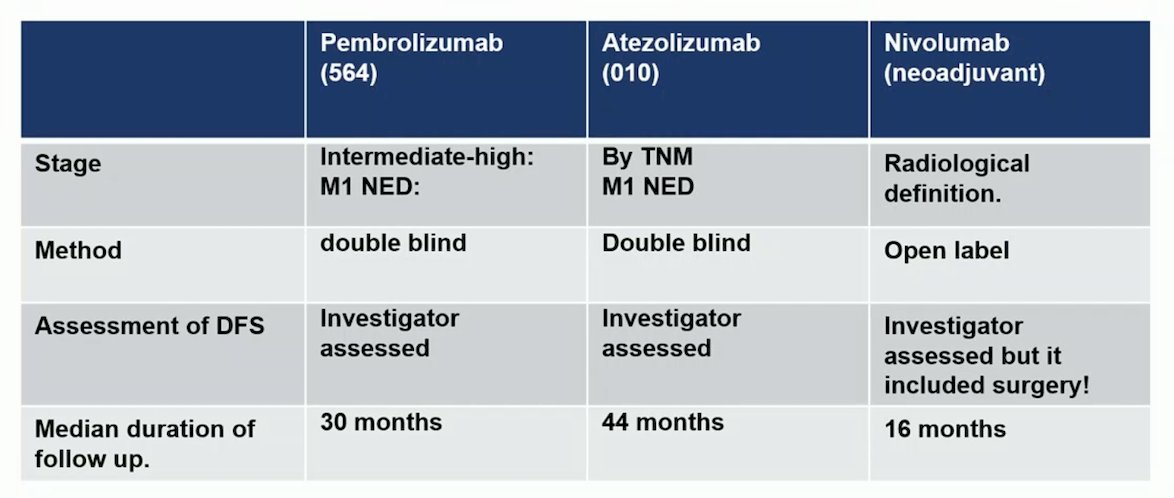
Dr. Powles highlighted that generally the pembrolizumab and atezolizumab trials were similar, but the nivolumab study was quite different, as highlighted in the following table:
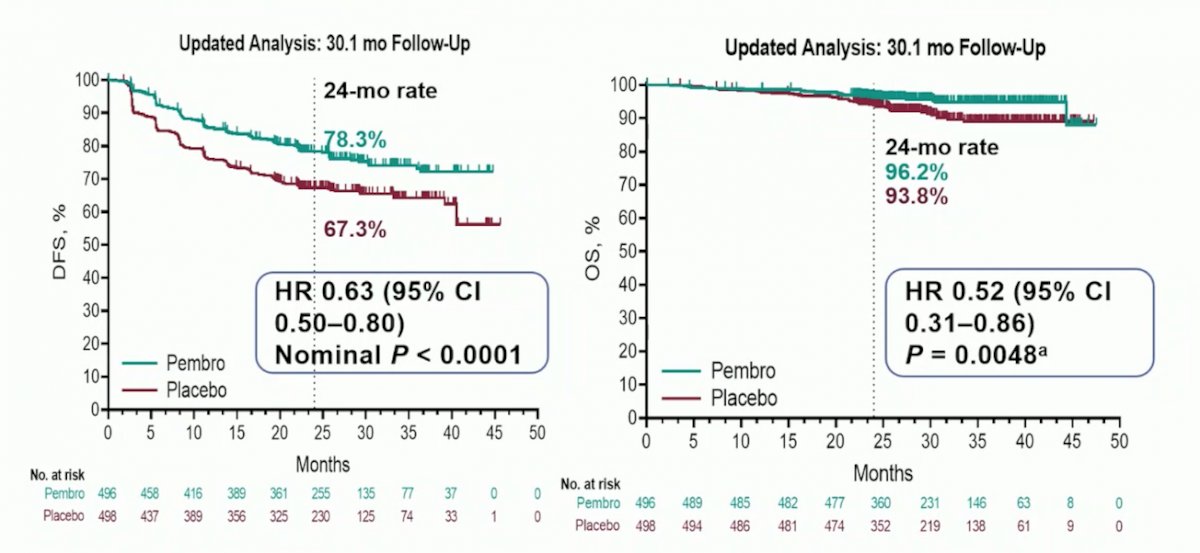
Without question, there is a signal for pembrolizumab in the adjuvant setting from the KEYNOTE-564 trial,1 which was confirmed on the update 30-month analysis:
In the IMmotion010 trial presented by Dr. Bex, the median investigator-assessed-disease free survival (DFS) was 57.2 months (95% CI 44.6, NE) for atezolizumab and 49.5 months (95% CI 47.4, NE) for placebo (HR 0.93, 95% CI 0.75, 1.15; p = 0.495). With regards to overall survival (OS), data was immature, with the median OS not reached in either arm (stratified HR atezolizumab vs placebo HR 0.97, 95% CI 0.67-1.42):

Additionally, unlike pembrolizumab, Dr. Powles notes that there was also no benefit in the M1 NED (HR 0.93, 95% CI 0.58, 1.49) nor patients with sarcomatoid differentiation (HR 0.77, 95% CI 0.44, 1.36). Interestingly, IMmotion150 showed atezolizumab had a response rate of ~25% in advanced disease, thus PD-L1 therapy may be less efficacious than PD-1 therapy:

In the phase 3 PROSPER trial, the investigational arm included nivolumab administered (480mg IV q4 weeks) with 1 dose prior to surgery followed by 9 adjuvant doses. The control arm was surgery followed by surveillance without a placebo. However, Dr. Powles notes that there are a number of factors that may have influenced the results of this trial, including (i) the study being open label, (ii) no BICR, (iii) imbalance in delays during the biopsy portion of the trial, (iv) toxicity in response to starting nivolumab preoperatively, (v) not all patients undergoing surgery, and (vi) surgical complications and other potential issues with restarting nivolumab after surgery:
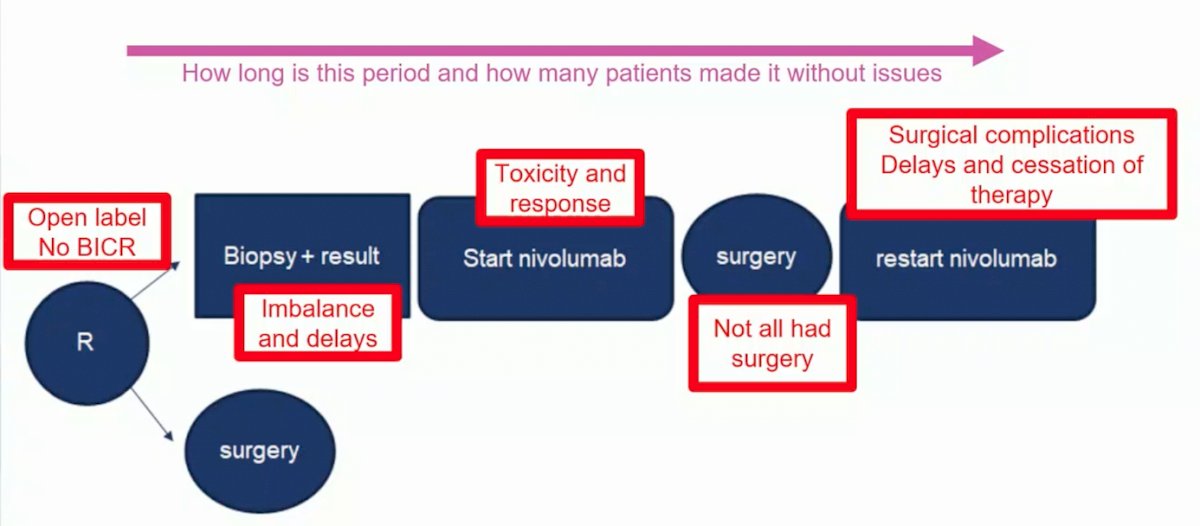
Dr. Powles notes that the interim analysis showed no difference in disease free survival, but what was the cause of the initial drop in the Kaplan-Meier curve? Furthermore, at 12 months 77% of patients were progression free or had surgery, and 53% of patients were still on study. As such, some details of this censoring are needed:
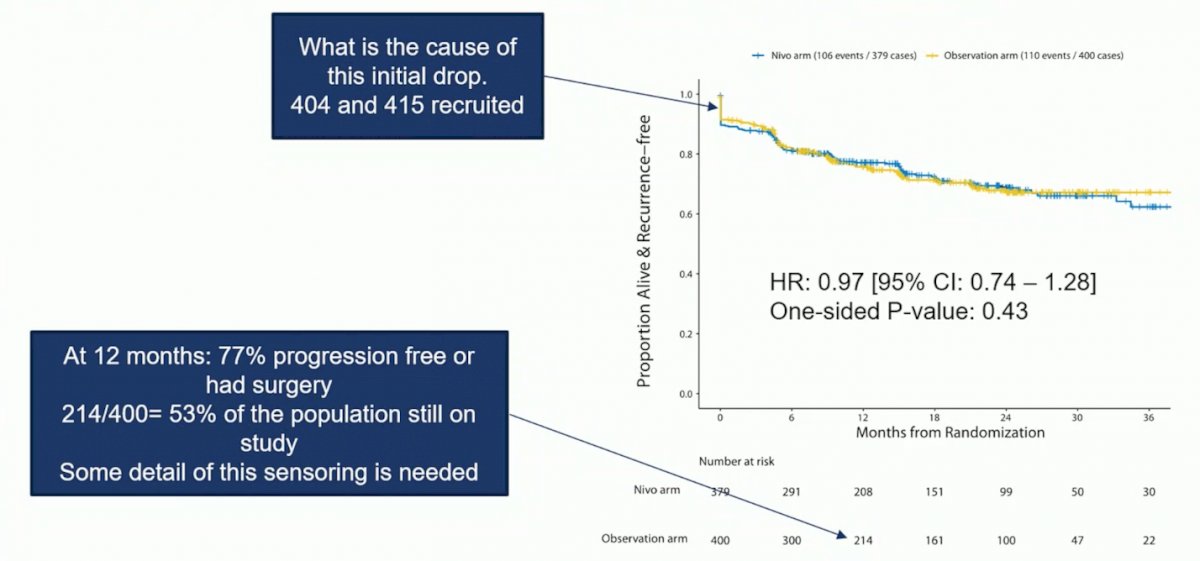
Other issues that must be considered for a neoadjuvant component of a trial is to assess the comparative control arm performance and how confident are we with the DFS/no surgery primary endpoint? Dr. Powles emphasized that the goal of these studies is OS, and even if the data are immature, we should see these outcomes even though the trial was stopped for futility. Importantly, is there a way of identifying patients who are at risk? Half the patients don’t relapse in the adjuvant studies. Early work from the bladder cancer literature assessing adjuvant atezolizumab suggests that there may be a role for ctDNA as a biomarker in this setting.
Have we seen this story before? As follows is Dr. Powles’ brief summary of adjuvant targeted studies (sunitinib, sorafenib, axitinib, pazopanib, and everolimus):
- ASSURE2 was the first trial which showed no difference in DFS or OS between placebo, sunitinib or sorafenib
- S-TRAC3 was positive for DFS but OS was very negative. However, there was a suggestion that dosing of sunitinib played a role
- This dosing issue was replicated in the pazopanib study,4 although this was a negative DFS trial
- More recently, longer periods of therapy (SORCE)5 and other agents such as axitinib6 and everolimus failed to make a clear impact on DFS, and OS remained very negative
- VEGF TKIs do not cure patients with advanced disease, and they do not have a major DFS impact in the adjuvant setting without any OS signal
To conclude his presentation discussing adjuvant immunotherapy trials for high risk RCC patients, Dr. Powles provided his final thoughts (for now!):
- The pembrolizumab trial is strongly positive for DFS, whereas the OS signal is supportive but not mature
- While PD-L1 therapy has activity, it has performed less well in clear cell RCC, which may in part explain the results of adjuvant atezolizumab
- The neoadjuvant nivolumab trial has a number of design and methodology issues, thus it is not possible to judge the comparative activity of nivolumab based on this study
- The contradictory findings of these 3 adjuvant trials won’t be down to luck alone, as this is not an exact replication of the VEGF TKI adjuvant trial story
- Thus, we really need to start selecting patients for therapy (biomarkers)
- Patients should be aware that pembrolizumab delays DFS with a chance of life changing toxicity and a chance of improving OS in the future. Patients should also be aware that immune checkpoint inhibitors have not been able to replicate the pembrolizumab data creating new uncertainty in renal cancer
Presented by: Thomas B. Powles, MBBS, MRCP, MD, Department of Genitourinary Oncology, Barts Cancer Institute, Experimental Cancer Medicine Centre, Queen Mary University of London, St Bartholomew’s Hospital, London, UK
Written by: Zachary Klaassen, MD, MSc – Urologic Oncologist, Assistant Professor of Urology, Georgia Cancer Center, Augusta University/Medical College of Georgia, @zklaassen_md on Twitter during the 2022 European Society of Medical Oncology (ESMO) Annual Hybrid Meeting, Paris, FR, Fri, Sept 9 – Tues, Sept 13, 2022.
References:
- Choueiri TK, Tomczak P, Park SH, et al. Adjuvant Pembrolizumab after Nephrectomy in Renal-Cell Carcinoma. N Engl J Med. 2021 Aug 19;385(8):683-694.
- Haas NB, Manola J, Uzzo RG, et al. Adjuvant sunitinib or sorafenib for high-risk, non-metastatic renal-cell carcinoma (ECOG-ACRIN E2805): A double-blind, placebo-controlled, randomised, phase 3 trial. Lancet 2016;387(10032):2008-2016.
- Ravaud A, Motzer RJ, Pandha HS, et al. Adjuvant Sunitinib in High-Risk Renal-Cell Carcinoma after Nephrectomy. N Engl J Med 2016;375(23):2246-2254.
- Motzer RJ, Haas NB, Donskov F, et al. Randomized phase III trial of adjuvant pazopanib versus placebo after nephrectomy in patients with locally advanced renal cell carcinoma (RCC) (PROTECT). J Clin Oncol 2017;35(35):3916-3923.
- Eisen T, Frangou E, Oza B, Ritchie AWS, et al. Adjuvant sorafenib for renal cell carcinoma at intermediate or high risk of relapse: Results from the SORCE randomized phase III Intergroup Trial. J Clin Oncol. 2020 Dec 1;38(34):4064-4075.
- Gross-Goupil M, Kwon TG, Eto M, et al. Axitinib versus placebo as an adjuvant treatment of renal cell carcinoma: results from the phase III, randomized ATLAS trial. Ann Oncol 2018 Dec 1;29(12):2371-2378.
Related Content:
ESMO 2022: Phase III RandOmized Study Comparing PErioperative Nivolumab versus Observation in Patients with RCC Undergoing Nephrectomy (PROSPER, ECOG-ACRIN EA8143), a National Clinical Trials Network Trial
ESMO 2022: IMmotion010: Efficacy and Safety from the Phase III Study of Atezolizumab vs Placebo as Adjuvant Therapy in Patients with RCC at Increased Risk of Recurrence After Resection


