(UroToday.com) The European Society of Medical Oncology (ESMO) 2021 annual meeting’s non-prostate cancer mini-oral session included a presentation by Dr. Nieves Martinez Chanza discussing results of avelumab as neoadjuvant therapy in platinum eligible and ineligible patients with muscle-invasive bladder cancer (MIBC). Cisplatin-based neoadjuvant chemotherapy followed by radical cystectomy is considered as standard of care for non-metastatic muscle-invasive bladder cancer patients based on a modest survival benefit correlated with pathological complete response. However, up to 50% of patients are ineligible for cisplatin-based chemotherapy. Avelumab is a monoclonal antibody directed against PD-L1, indicated in locally advanced or metastatic urothelial cancer. Neoadjuvant studies demonstrate promising antitumor activity of immune checkpoint inhibitors, with single-agent <ypT2N0 rates of 39-56% and in combination with chemotherapy <ypT2N0 rates of 56-69%. Dr. Martinez Chanza and colleagues hypothesized that neoadjuvant avelumab would increase the rates of pathologic response and improve clinical outcomes when combined with standard chemotherapy. At the ESMO 2021 congress, they reported preliminary data from the AURA trial assessing preoperative avelumab with two cisplatin-based regimens in the cisplatin eligible cohort.
The AURA trial is a prospective, multicenter, randomized, phase II trial for patients with cT2-4aN0-2M0 bladder carcinoma. Cisplatin-eligible patients received cisplatin-gemcitabine plus avelumab or dose-dense MVAC plus avelumab (1:1). The study schema for AURA is as follows:
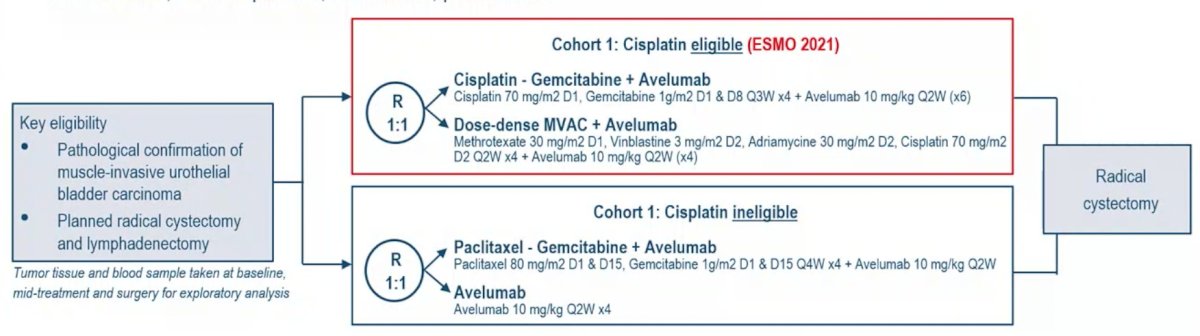
The primary endpoint was pathological complete response (ypT0/isN0) with the objective, in each arm, to show pathological complete response rate > 25% (90% power reached in case of pathological complete response rate > 45%). Twostep design was used with planned interim analysis after 28 evaluable patients per arm. Secondary endpoints were pathologic downstaging rate (<ypT2N0) and safety.
At the interim analysis data cut-off, 56 cisplatin-eligible patients were evaluable. For the cisplatin-gemcitabine + avelumab arm (n=28), the median age was 69 years (range: 41- 81), 64% were male, 64% were cT2N0, and 7% were cT4N0. For the dose-dense MVAC + avelumab arm (n=28), the median age was 62 years (range: 51-77), 79% were male, 61% were cT2N0, and 7% were cT4N0. Six patients did not undergo surgery but were included in intention to treat analysis. In the cisplatin-gemcitabine + avelumab arm, 54% of patients met the primary endpoint of pathologic complete response, compared to 61% in the dose-dense MVAC + avelumab arm:

The most common grade 3/4 adverse events were thrombocytopenia (29%), acute kidney injury (18%), neutropenia (14%), and anemia (13%). No patients required steroids for immune-related adverse events, and no treatment-related deaths were reported. The IDMC recommended stopping accrual in this cohort because the endpoint was reached.
Dr. Martinez Chanza concluded her presentation of the AURA trial with the following concluding statements:
- Neoadjuvant avelumab in combination with a standard cisplatin-based regimen demonstrated a high pathologic response rate: DD-MVAC: 64%; cisplatin-gemcitabine: 57%
- Cisplatin-based neoadjuvant chemotherapy plus avelumab is associated with manageable adverse events which did not compromise surgical resection
- Further results in the cisplatin-ineligible cohort, survival data, and translational research projects are ongoing
Presented by: Nieves Martinez Chanza, Medical Oncology, Jules Bordet Institute, Brussels, Belgium
Written by: Zachary Klaassen, MD, MSc – Urologic Oncologist, Assistant Professor of Urology, Georgia Cancer Center, Augusta University/Medical College of Georgia, @zklaassen_md on Twitter during the 2021 European Society for Medical Oncology (ESMO) Annual Congress 2021, Thursday, Sep 16, 2021 – Tuesday, Sep 21, 2021.


