(UroToday.com) Dr. Laurence Albiges provided a discussion of two clinical studies, one from the COSMIC-021 trial examining combination cabozantinib and atezolizumab in untreated metastatic clear cell renal cell carcinoma, and the other an RNA expression biomarker-driven trial of tyrosine kinase inhibitor versus nivolumab monotherapy versus nivolumab and ipilimumab.
She began by summarizing the COSMIC-021 data, which showed ORR rates of 53 and 58% respectively in response to cabozantinib 40 mg of 60 mg in combination with atezolizumab.
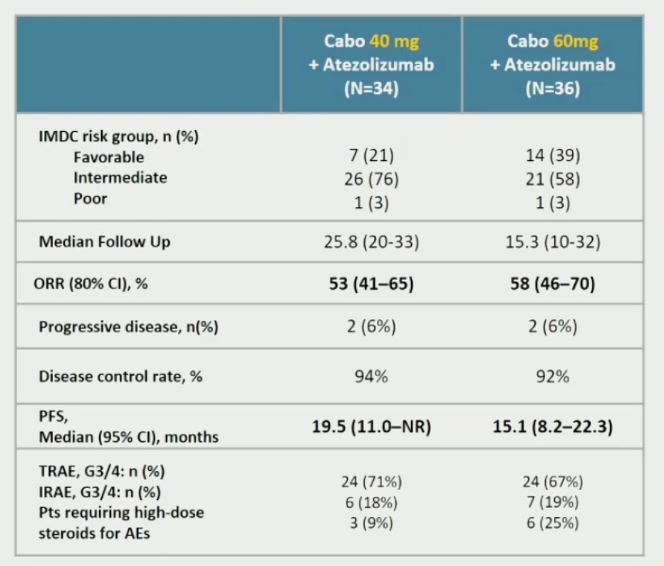
Dr. Albiges then posed many questions surrounding this combination therapy.
- Is cabozantinib is the right partner for treatment with atezolizumab? Though not a correct head-to-head comparison, this early phase data compares favorably with published data for bevacizumab and atezolizumab, the latter which was associated with an objective response rate of 37% and median progression free survival of 11.2 months.
- Is cabozantinib is the right tyrosine kinase inhibitor to use? The answer may be yes as cabozantinib has a broad spectrum of TKI activity and has clear immunomodulatory effects such as upregulating antigen presentation, downregulating immunosuppressive cell populations, and increasing infiltration of CD8+ T cells.
- Is atezolizumab is the right combination partner for cabozantinib? This is not clear, as with the caveats of comparing outcomes from two separate trials cabozantinib and atezolizumab had comparable response rates to cabozantinib and nivolumab.
- What is the right dose of cabozantinib? This is unclear based on the current data.
- Finally, she then compared this regimen in the context of other first line treatment option in mccRCC. While the response rates and progression-free survival times are comparable, Dr. Albiges was intrigued by the lower rates of progressive disease in COSMIC-021 relative to other regimens.

Finally, Dr. Albiges reviewed the biomarker data from this trial suggesting that PD-L1+ tumors with high CD8+ Tcell numbers were more likely to respond to therapy.
She concluded by saying that COSMIC-021 shows the combination of cabozantinib is active with atezolizumab in first-line untreated mccRCC, with molecular subtypes that are enriched for responders. Further work is required to understand whether cabozantinib is the optimal TKI for use with immunotherapy combinations, and safety is likely to be one consideration when choosing treatment options.
Dr. Albiges then discussed the BIONIKK phase 2 biomarker-driven trial building on prior transcriptomic signatures to assess if certain populations could be identified that are enriched for response to various therapies. She postulated that perhaps a more interesting arm for the immune-low population could have been a combination TKI and immune checkpoint blockade but was otherwise complimentary of the study design.
She concluded with the following slide:

Presented by: Laurence Albiges, MD, PhD, Vice Chair, Department of Cancer Medicine, Gustave Roussy Institute, Villejuif, France
Written by: Alok Tewari, MD, PhD, Medical Oncologist at the Dana-Farber Cancer Institute, at the 2020 European Society for Medical Oncology Virtual Congress (#ESMO20), September 19th-September 21st, 2020.

