Biomarker analyses that can better characterize the disease and identify novel therapeutic opportunities could further improve outcomes with Avelumab or other immune checkpoint inhibitors in patients with UC. PD-L1 protein expression, tumor mutational burden (TMB), and gene expression signatures have been identified as candidate biomarkers, but there is variable data showing their predictive utility2.
The JAVELIN 100 bladder trial is the first randomized phase 3 trial comparing immune checkpoint inhibitor against the best standard of care and therefore provides a unique opportunity to evaluate the predictive utility of several emerging biomarkers in UC.
The study design of the JAVELIN 100 bladder trial is shown in figure 1. The methodology details on the hypotheses regarding the predictive utility of tumor biomarkers are shown in figure 2. In summary, tumor tissue was collected from either the primary or metastatic tumor prior to first-line chemotherapy. The tissue was subjected to biomarker analysis by either immunohistochemistry or next-generation sequencing. Ad hoc exploratory analyses of associations between tumor biomarkers and overall survival (OS) were conducted.
Figure 1 – JAVELIN 100 bladder trial design:
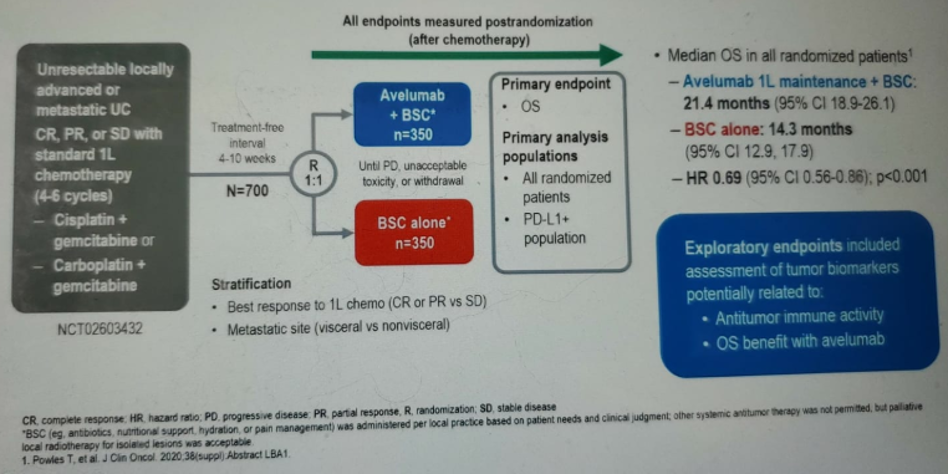
Figure 2- Methodology used to create hypotheses on the predictive utility of tumor biomarkers:

Next, Dr. Sridhar showed the OS benefit in subgroups defined by PD-L1 expression on both tumor cells and immune cells (Figure 3).
Figure 3 – Overall survival benefit in subgroups defined by PD-L1 expression on tumor cells and immune cells:

Next, Dr. Sridar showed the OS in subgroups defined by TMB and PD-L1 status (Figure 4). This showed an advantage for the Avelumab +BSC when TMB was above the median, but not below. A similar beneficial effect was seen in high PD-L1 status, but not in low PD-L1 status.
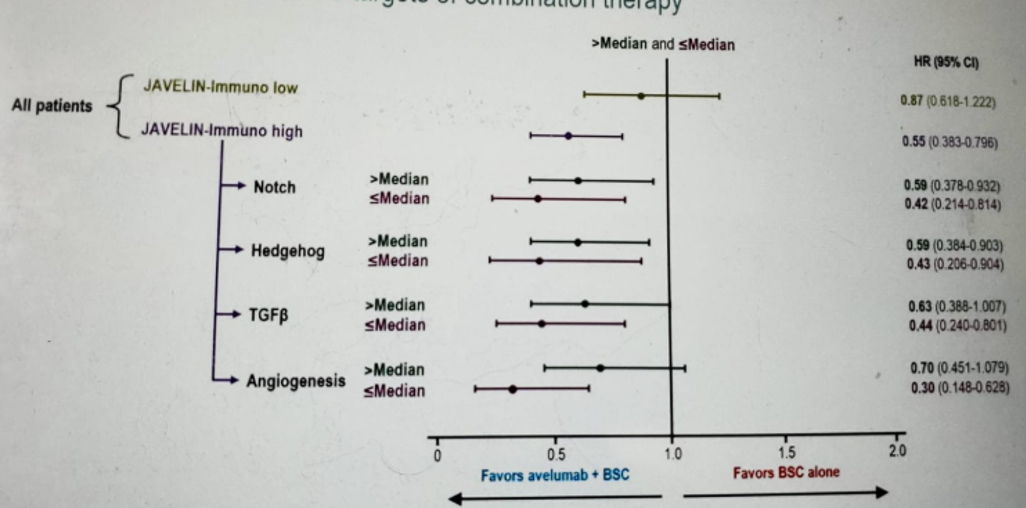
Lastly, Dr. Sridhar showed the impact of mutation profiles on OS benefit with Avelumab (Figure 5). Tumor gene expression data can identify genes that may be associated with OS benefit from Avelumab. Figure 6 shows the exploration of the relationships between established immune active gene signatures and outcomes. There are also signaling pathways that may reduce the OS benefit in patients with elevated antitumor immunity (figure 7).
Figure 4 – Overall survival benefit in subgroups defined by tumor mutational burden and PD-L1 status:

Figure 5 – Impact of mutation profiles on overall survival benefit in patients treated with overall survival:
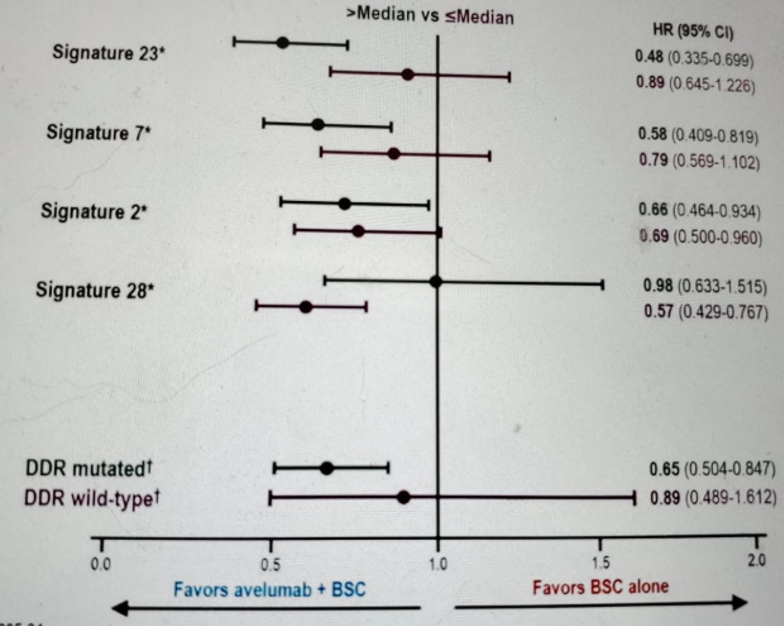
Figure 6 – The relationship between established immune active gene signatures and outcomes:
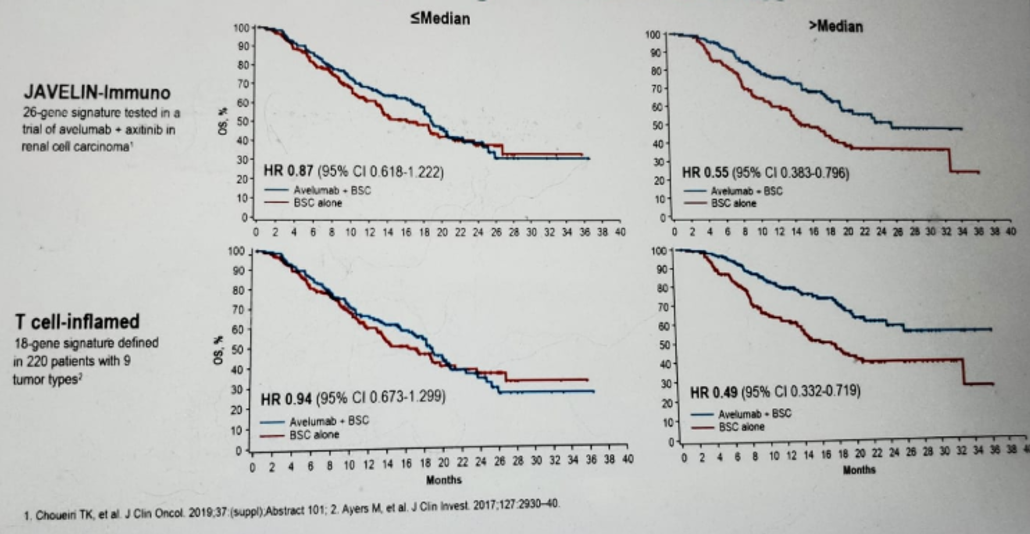
Figure 7 – Signaling pathways with a possible reduction of overall survival in patients with elevated antitumor immunity:
Summarizing her talk, Dr. Sridhar concluded that the positive results of the JAVELIN 100 bladder trial enabled exploratory analysis of predictive biomarkers that may identify patients who would obtain an OS benefit from Avelumab first-line maintenance therapy + BSC vs. BSC alone. The results demonstrated that none of the biomarkers assessed, either alone or in combinations, optimally predicted OS benefit with Avelumab. The are multiple other biomarkers that were identified as potentially predictive, which could be explored further in the future. Lastly, the JAVELIN 100 bladder trial led to the approval of first-line maintenance Avelumab in patients with advanced UC, independent of their PD-L1 status. Combinations of biomarkers could help to further characterize patients with advanced UC who may benefit from Avelumab first-line maintenance therapy.
References:
1. Powles T, Park SH, Voog E, et al. Maintenance avelumab + best supportive care (BSC) versus BSC alone after platinum-based first-line (1L) chemotherapy in advanced urothelial carcinoma (UC): JAVELIN Bladder 100 phase III interim analysis. Journal of Clinical Oncology 2020; 38(18_suppl): LBA1-LBA.
2. Galsky MD, Mortazavi A, Milowsky MI, et al. Randomized Double-Blind Phase II Study of Maintenance Pembrolizumab Versus Placebo After First-Line Chemotherapy in Patients With Metastatic Urothelial Cancer. Journal of clinical oncology: official journal of the American Society of Clinical Oncology 2020; 38(16): 1797-806.
Presemted by: Srikala (Kala) Sridhar, MD, MSc, FRCPC, Vice Chair, Medical Advisory Board, Princess Margaret Hospital, Associate Professor Department of Medicine, Division of Medical Oncology at the University of Toronto, Toronto, Ontario
Written by: Hanan Goldberg, MD, MSc., Assistant Professor of Urology, SUNY Upstate Medical University, Syracuse, NY, USA @GoldbergHanan at the European Society for Medical Oncology Virtual Congress, ESMO Virtual Congress 2020 #ESMO20, 18 Sept - 21 Sept 2020
Related Content:
ESMO Virtual Congress 2020: Subgroup Analyses from JAVELIN Bladder 100: Avelumab first-line (1L) maintenance + Best Supportive Care (BSC) vs BSC Alone with 1L Chemotherapy for Advanced Urothelial Carcinoma
ESMO Virtual Congress 2020: Patient-Reported Outcomes from JAVELIN Bladder 100: Avelumab First-Line Maintenance Plus Best Supportive Care vs Best Supportive Care Alone For Advanced Urothelial Carcinoma
JAVELIN Bladder 100: Avelumab for Previously Untreated Locally Advanced or Metastatic Urothelial Carcinoma - Thomas Powles

