
In a mini-oral presentation at this year’s European Society of Medical Oncology (ESMO) 2020 Virtual Annual Meeting, Dr. Grivas presented pre-specified subgroup analyses of the JAVELIN Bladder 100 trial. To briefly recap, this study enrolled patients with advanced urothelial carcinoma, defined as those with unresectable locally advanced disease or metastatic disease. To be eligible, patients had to have received platinum based induction chemotherapy with either cisplatin or carboplatin in combination with gemcitabine and have not had disease progression after 4-6 cycles of therapy. Among these patients with responsive or stable disease on induction chemotherapy, enrolled patients were randomized to avelumab and best supportive care or best supportive care alone. The primary endpoint was overall survival.
Pre-specified subgroups assessed in this presentation included best response to first-line induction chemotherapy (complete response/partial response vs stable disease) and location of disease (visceral vs non-visceral). PD-L1 status was also assayed.
In total, 700 patients were enrolled and randomized in a 1:1 fashion. Median follow-up exceeded 19 months. The authors identified a survival benefit for the addition of avelumab to best supportive care in all identified subgroups. In particular, the benefit was comparable in those with complete/partial response (hazard ratio 0.69, 95% confidence interval 0.53 to 0.89) and those with stable disease (hazard ratio 0.70, 95% confidence interval 0.46 to 1.05). Additionally, stratification according to the presence of visceral disease (hazard ratio 0.82, 95% confidence interval 0.62 to 1.09) and those with non-visceral disease (hazard ratio 0.54, 95% confidence interval 0.38 to 0.76) both appeared to derive benefit.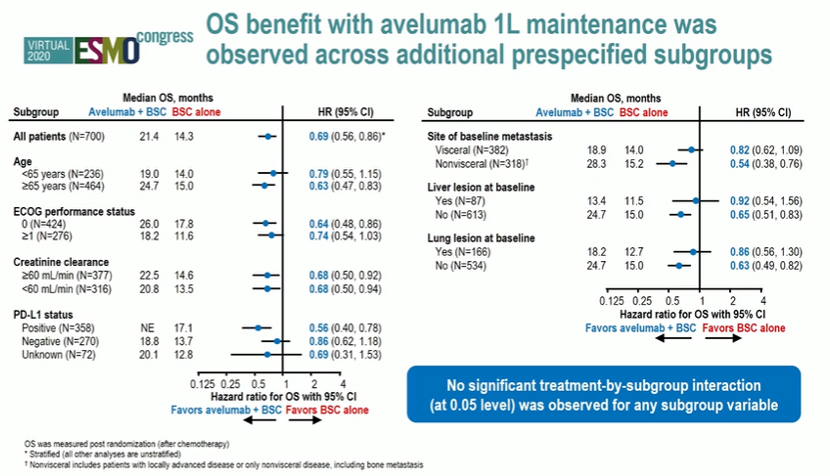
Similarly, the benefit of adding avelumab did not appear to differ based on patient age, performance status, creatinine clearance, PD-L1 status, or presence of liver or lung lesions.
Presented by: Petros Grivas, MD, Ph.D. Associate Professor and the Clinical Director of the Genitourinary Cancers Program at the University of Washington, and an Associate Member of the Clinical Research Division at the Fred Hutchinson Cancer Research Center
Written by: Christopher J.D. Wallis, Urologic Oncology Fellow, Vanderbilt University Medical Center, Twitter: @WallisCJD on Twitter at the European Society for Medical Oncology Virtual Congress, ESMO Virtual Congress 2020 #ESMO20, 18 Sept - 21 Sept 2020
Related Content:
ASCO 2020 JAVELIN Bladder 100 Phase III Results: Maintenance Avelumab + Best Supportive Case vs BSC Alone After Platinum-Based First-Line Chemotherapy in Advanced Urothelial Carcinoma
LBA1 Discussion: ASCO 2020: Checkpoint Inhibition in Metastatic Urothelial Carcinoma: Timing is Everything
JAVELIN Bladder 100: Results of First-line Maintenance Therapy Plus Best Supportive Care Demonstrates Significant Prolonged OS in Advanced Urothelial Cancer - Cora Sternberg
JAVELIN Bladder 100: Avelumab for Previously Untreated Locally Advanced or Metastatic Urothelial Carcinoma - Thomas Powles

