(UroToday.com) Despite a number of therapies with proven survival benefit, the prognosis of patients with advanced urothelial carcinoma remains poor. In addition to cytotoxic chemotherapy using a platinum-based regime, the use of checkpoint inhibitors has demonstrated promise. However, long-term responses are rare.
In a mini-oral presentation at this year’s European Society of Medical Oncology (ESMO) 2020 Virtual Annual Meeting, Dr. Pavlos Msaouel presented results of a multi-cohort phase II trial evaluating sitravantinib in combination with nivolumab in patients with advanced urothelial carcinoma. Sitravantinib is a spectrum-selective tyrosine kinase inhibitor which targets a number of receptors including TAM receptors (Tryo3, Axl, and MerTK), split family receptors (VEGFR2 and c-Kit), and c-Met. Through this mechanism sitravantinib has effects but in terms of anti-tumor activity and modulation of the immune system through reduction of Type 2 tumor-associated macrophages, regulatory T cells, and myeloid-derived suppressor cells in addition to enhancing T cell-mediated anti-tumor immune responses.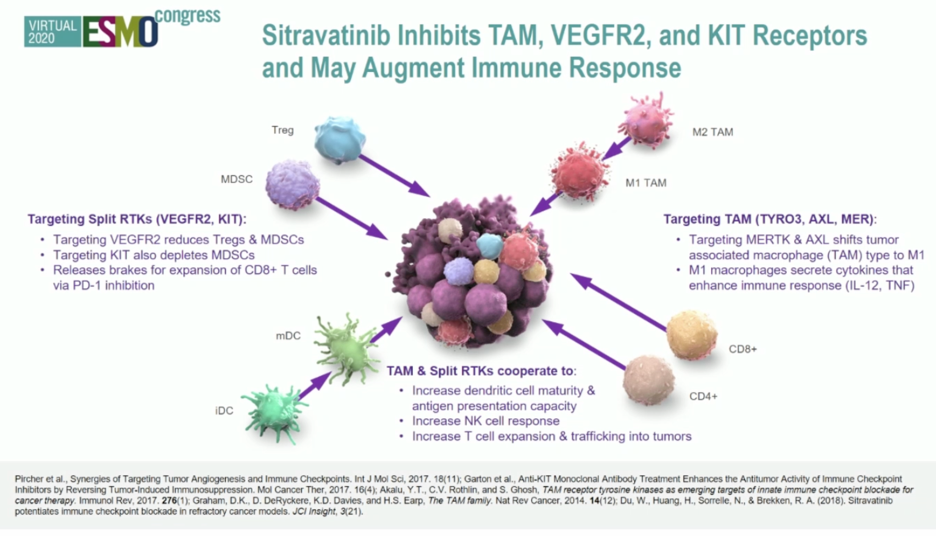
Thus, the authors hypothesized that there would be an augmentation of the benefit of checkpoint inhibitor therapy with the addition of sitravantinib.
In this presentation, the authors presented results of cohort 5 of their multi-cohort phase II trial assessing patients with advanced or metastatic urothelial carcinoma who had disease progression on or after treatment with platinum-based chemotherapy who had yet to receive checkpoint inhibitor therapy. Patients were administered sitravantinib orally in addition to nivolumab intravenously. The authors used a Predictive Probability Design to allow progression through stages of the study based on results in the initial phases.
Thus far, the authors have enrolled 40 patients of whom 38 formed the full analysis population and 30 were suitable for evaluation of the clinical activity. The authors found objective responses in 11 of 30 patients (37%) with a single confirmed complete response. Among those with complete or partial responses, median duration of response has been 5.6 months (95% confidence interval 3.7 to 7.3).
With a median follow-up of 8.7 months, median progression free survival in the full analysis population (n=38) is so far is 4.0 months (95% confidence interval 1.9 to 7.3) and overall survival is 9.2 months (95% confidence interval 5.1 to 11.6 months).
Assessment of treatment toxicity demonstrated relatively few high-grade adverse events.
In conclusion, this trial remains ongoing to assess this promising clinical approach.
Presented by: Pavlos Msaouel, MD, PhD, Department of Genitourinary Medical Oncology, Division of Cancer Medicine, MD Anderson Cancer Center, Houston, Texas
Written by: Christopher J.D. Wallis, Urologic Oncology Fellow, Vanderbilt University Medical Center, Twitter: @WallisCJD on Twitter at the European Society for Medical Oncology Virtual Congress, ESMO Virtual Congress 2020 #ESMO20, 18 Sept - 21 Sept 2020
Read the Invited Discussion on this Topic:ESMO Virtual Congress 2020: What is the Future of Immunotherapy in Urothelial Cancer?

