Barcelona, Spain (UroToday.com) Taxanes (docetaxel, cabazitaxel) and androgen receptor-targeted therapies (ARTs; abiraterone, enzalutamide) are the standard of care in metastatic castration resistant prostate cancer (mCRPC). However, patients may not respond to abiraterone after progressing on prior enzalutamide or vice versa. Cabazitaxel retains activity in patients progressing on prior docetaxel, abiraterone, or enzalutamide. Importantly, the optimal treatment sequence of these agents is unknown. The CARD study compared cabazitaxel vs. abiraterone or enzalutamide in patients with mCRPC previously treated with docetaxel and the alternative ART. At the ESMO 2019 Presidential session, Ronald De Wit, MD, PhD, presented the initial results of CARD.
Patients in CARD had mCRPC previously treated with ≥ 3 cycles of docetaxel and progressed ≤ 12 months on an alternative ART, in any order, and were randomized 1:1 to cabazitaxel (25 mg/m2 IV Q3W + prednisone + G-CSF) vs. abiraterone (1000 mg PO + prednisone) or enzalutamide (160 mg PO). Randomization was stratified by Eastern Cooperative Oncology Group (ECOG) performance status (0/1 vs 2), time to progression on prior ART (≤ 6 vs 6–12 months) and ART timing (before vs after docetaxel). Other key inclusion criteria were histologically confirmed mCRPC, PSA value of >= 2 ng/mL, serum testosterone <0.5 ng/mL, and adequate organ and bone marrow function. The primary endpoint was radiographic progression free survival (rPFS), with 196 rPFS events needed to test a hazard ratio of 0.67 for cabazitaxel (80% power, 2-sided alpha 0.05). Other secondary efficacy endpoints included pain response, time to symptomatic skeletal-related events, safety, health-related quality of life, and biomarkers.
The CARD study design:
Overall, 255 patients were randomized. The median age was 70 years and this was quite an elderly population with 31.0% ≥ 75 year of age. Furthermore, the median number of cycles was higher for cabazitaxel vs ART (7 vs 4). rPFS was significantly improved with cabazitaxel vs ART (median 8.0 vs 3.7 months; HR 0.54, 95% CI 0.40–0.73; p < 0.0001), which was also consistent across the majority of subgroups: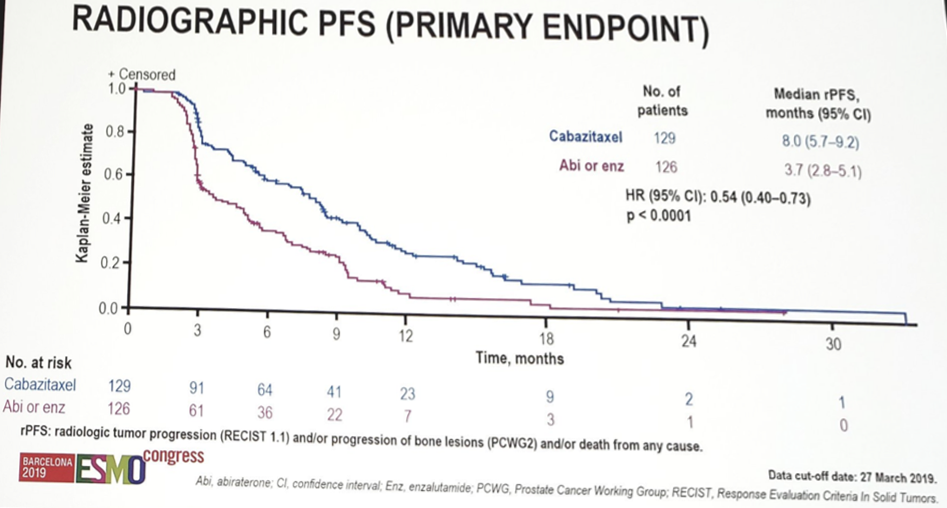
Cabazitaxel also robustly improved overall survival (OS) (median 13.6 vs 11.0 months; HR 0.64, 95% CI 0.46–0.89; p = 0.0078) despite crossover, as well as PFS (median 4.4 vs 2.7 months; p < 0.0001), confirmed PSA50 response (35.7% vs 13.5%; p = 0.0002) and tumor response (36.5% vs 11.5%; p = 0.004).
Pain response (45.0% vs 19.3%, p < 0.0001) and time to symptomatic skeletal events (median NR vs 16.7 months; HR 0.59, 95% CI 0.35–1.01; p = 0.05) were also improved with cabazitaxel. In a post-hoc analysis, cabazitaxel significantly improved rPFS regardless of prior therapy sequence: enzalutamide after docetaxel and abiraterone (median 7.4 vs 4.8 months; HR 0.57, 95% CI 0.36–0.90); abiraterone after docetaxel and abiraterone (median 8.2 vs 3.4 months; HR 0.44, 95% CI 0.29–0.67).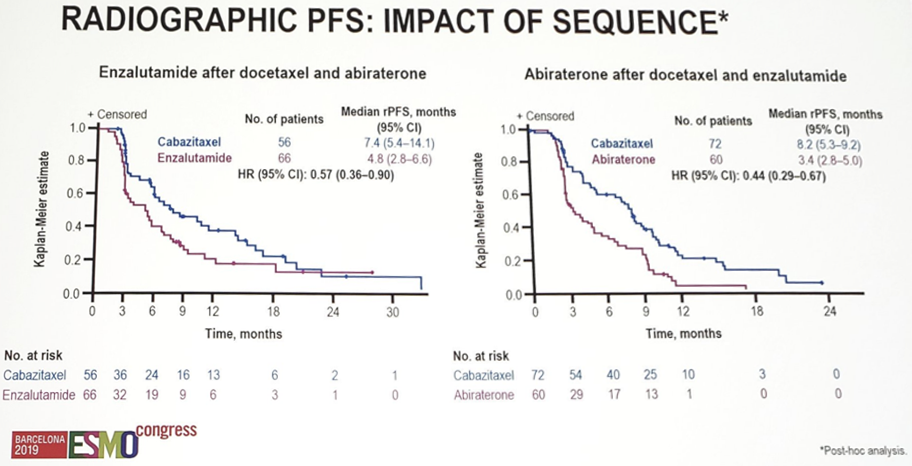
Grade ≥ 3 adverse events occurred in 56.3% on cabazitaxel vs 52.4% of patients on ART. For cabazitaxel vs ART, main grade ≥ 3 adverse events were: renal disorders (3.2% vs 8.1%), infections (7.9% vs 7.3%), musculoskeletal pain/discomfort (1.6% vs 5.6%), cardiac disorders (0.8% vs 4.8%), spinal cord/nerve root disorders (2.4% vs 4.0%), asthenia/fatigue (4.0% vs 2.4%), diarrhea, peripheral neuropathy, and febrile neutropenia (3.2% vs 0%). Adverse events led to death in 7 patients (5.6%) on cabazitaxel vs 14 patients (11.3%) on ART.
Dr. De Wit concluded his presentation of the CARD trial with the following take home messages:
- CARD met its primary objective as cabazitaxel more than doubled rPFS vs enzalutamide or abiraterone.
- Cabazitaxel reduced the risk of death by 36% vs enzalutamide or abiraterone.
- Other secondary endpoints favored cabazitaxel: PFS, PS, tumor and pain responses, as well as time to symptomatic skeletal event.
- rPFS with cabazitaxel remained superior regardless of abiraterone or enzalutamide sequence.
- These results support the use of cabazitaxel over abiraterone or enzalutamide in this setting.
Following Dr. De Wit’s presentation, the CARD trial was published in The New England Journal of Medicine.
Clinical trial identification: NCT02485691
Reference:
de Wit, Ronald et al. 2019. "Cabazitaxel versus Abiraterone or Enzalutamide in Metastatic Prostate Cancer". New England Journal of Medicine. Massachusetts Medical Society. doi:10.1056/nejmoa1911206.
Presented by: Ronald De Wit, MD, PhD, Dept of Medical Oncology Erasmus University Medical Center Daniel den Hoed Cancer Center, Rotterdam, The Netherlands
Written by: Zachary Klaassen, MD, MSc – Assistant Professor of Urology, Georgia Cancer Center, Augusta University/Medical College of Georgia, Twitter: @zklaassen_md, at the 2019 European Society for Medical Oncology annual meeting, ESMO 2019 #ESMO19, 27 Sept - 1 Oct 2019 in Barcelona, Spain
Further Related Content:
ESMO 2019: Invited Discussant (LBA13) - The CARD Trial, A Randomized, Open-Label Study of Cabazitaxel vs Abiraterone or Enzalutamide in mCRPC


