Munich, Germany (UroToday.com) Radiation to the primary tumor in the setting of metastatic castration sensitive prostate cancer (mCSPC) is controversial. A large retrospective population-based study (SEER) found that five-year overall survival might be improved if local therapy is administered (radical prostatectomy or brachytherapy) compared with no local surgery at all.1 Another retrospective study using a different American database (NCDB) saw improvement in overall survival when mCSPC were treated with prostate radiotherapy (RT).2
These results were replicated using a European database (Munich Cancer Registry), which found that undergoing RP was independently associated with decreased cancer-specific mortality3. In a small non-randomized feasibility study, 23 patients with mCSPC received cytoreductive radical prostatectomy, and compared with the control group, had improved time to castration-resistant disease and cancer-specific survival rates (95.6% vs 84.2%, p=0.043), but no difference in overall survival4. This study seeks to answer that question using one of the arms of STAMPEDE.
In this arm of STAMPEDE, men with newly diagnosed metastatic prostate cancer were randomly assigned to either ADT +/- docetaxel or ADT +/- docetaxel + prostate radiotherapy in a 1:1 ratio. Two different RT regimens were allowed. Patients were stratified based on age, nodal status, performance status, and docetaxel use. 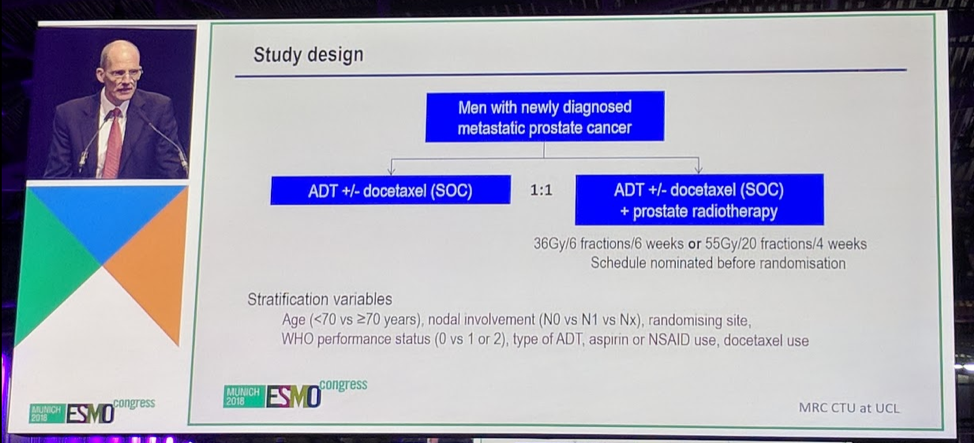
The investigators believed that local radiation in the metastatic setting would be more beneficial to patients with low volume disease than high volume disease and thus pre-specified those cohorts as well. This study accrued rapidly, with around 1000 patients in each arm. Baseline characteristics were well balanced.
In terms of failure free survival (FFS), patients receiving radiation had improved FFS over standard of care, with a hazard ratio of 0.07, p<0.00005. At 3 years, FFS for patients receiving radiation was 32% compared with 23% of patients receiving standard of care. However, there was no overall survival benefit in unselected patients. 
Subgroup analysis of overall survival by radiation regimen did not demonstrate any difference either. However, subgroup analysis by disease burden did show a difference as the investigators had hypothesized. Patients with low burden disease had a hazard ratio of 0.68, with a 3-year overall survival of 73% in the standard of care arm, compared with 81% of patients receiving SOC + radiation. 
Radiation was well tolerated and there were no significant differences in symptomatic local events in SOC or SOC + radiation. Interestingly, the point estimates of benefit for radiation were almost identical to that of HORRAD5, an almost identical, smaller trial of radiation for patients with metastatic prostate cancer. In HORRAD, there was no significant difference in overall survival but trended towards benefit with radiation. Here, with a larger sample size, we see the benefit exists for low volume patients. 
In summary, this large study demonstrates that treating the prostate with radiation in patients with metastatic CSPC has an overall survival benefit for patients with low volume disease. These results are practice changing. Of note, this study was done prior to patients being on abiraterone. However, a contemporary trial is examining whether or not this benefit continues in the abiraterone/LATITUDE era – PEACE 1 (NCT01957436) is a multi-center phase III study which will compare 4 different arms: ADT + docetaxel, ADT + docetaxel + abiraterone acetate + prednisone, ADT + docetaxel + radiation, and ADT + docetaxel + abiraterone acetate + prednisone + radiation.
Additional questions brought up by the discussant include:
- Is there a role for surgery in patients with mCSPC?
- Two studies (G RAMMP [NCT02454543] and TROMBONE [ISRCTN15704862]) are comparing whether or not surgery has a role in this population of patients.
- What is the role of metastasis directed therapy in oligometastatic disease?
- STOMP6 and POPSTAR7 show that XRT in this setting is worthy of further study and there are two ongoing clinical trials in this setting, CORE (NCT02759783) and PCS IX (NCT02685397)
- Should radiation therapy be offered in other tumor types with low volume metastatic disease?
- Much work still needs to be done
Presented by: Chris C. Parker, MD, Institute of Cancer Research, Sutton, GB
Written by: Jason Zhu, MD. Fellow, Division of Hematology and Oncology, Duke University, Twitter: @TheRealJasonZhu at the 2018 European Society for Medical Oncology Congress (#ESMO18), October 19-23, 2018, Munich Germany
References:
- Culp SH, Schellhammer PF, Williams MB. Might men diagnosed with metastatic prostate cancer benefit from definitive treatment of the primary tumor? A SEER-based study. European Urology 2014;65:1058-66.
- Rusthoven CG, Jones BL, Flaig TW, et al. Improved Survival With Prostate Radiation in Addition to Androgen Deprivation Therapy for Men With Newly Diagnosed Metastatic Prostate Cancer. Journal of Clinical Oncology 2016;34:2835-42.
- Gratzke C, Engel J, Stief CG. Role of radical prostatectomy in metastatic prostate cancer: data from the Munich Cancer Registry. European Urology 2014;66:602-3.
- Heidenreich A, Pfister D, Porres D. Cytoreductive Radical Prostatectomy in Patients with Prostate Cancer and Low Volume Skeletal Metastases: Results of a Feasibility and Case-Control Study. The Journal of Urology 2015;193:832-8.
- Boevé L, Hulshof M, Vis A, et al. PD10-10 A PROSPECTIVE, RANDOMIZED CONTROLLED TRIAL EVALUATING OVERALL SURVIVAL IN PATIENTS WITH PRIMARY BONE METASTATIC PROSTATE CANCER (MPCA) RECEIVING EITHER ANDROGEN DEPRIVATION THERAPY (ADT) OR ADT COMBINED WITH CONCURRENT RADIATION THERAPY TO THE PROSTATE, FINAL DATA FROM THE HORRAD TRIAL. The Journal of Urology 2018;199:e231-e2.HORRADBoevé L, Hulshof M, Vis A, et al. PD10-10 A PROSPECTIVE, RANDOMIZED CONTROLLED TRIAL EVALUATING OVERALL SURVIVAL IN PATIENTS WITH PRIMARY BONE METASTATIC PROSTATE CANCER (MPCA) RECEIVING EITHER ANDROGEN DEPRIVATION THERAPY (ADT) OR ADT COMBINED WITH CONCURRENT RADIATION THERAPY TO THE PROSTATE, FINAL DATA FROM THE HORRAD TRIAL. The Journal of Urology 2018;199:e231-e2.
- Ost P, Reynders D, Decaestecker K, et al. Surveillance or Metastasis-Directed Therapy for Oligometastatic Prostate Cancer Recurrence: A Prospective, Randomized, Multicenter Phase II Trial. Journal of Clinical Oncology 2017;36:446-53.
- Ost P. It Ain’t Over Till the Fat Lady Sings: The POPSTAR Trial. European Urology 2018;74:463-4.


