The recently published European Association of Urology (EAU) guidelines for upper tract urothelial carcinoma,1 clearly lay out the indications for kidney-sparing surgery, which includes low-risk disease (all must be present): (i) low-grade cytology, (ii) non-invasive on CT-urography, and (iii) low-grade ureteroscopic biopsy. Contraindications based on the guidelines including high-risk features, such as (i) multifocal disease, (ii) tumor size >2 cm, and (iii) muscle-invasive bladder cancer on previous radical cystectomy.
There are several options for how to perform kidney-sparing surgery for upper tract urothelial carcinoma. Percutaneous access has been used in the past, but there is a risk for seeding and thus ureteroscopy has become the most commonly used modality for treatment. Dr. Shariat uses digital ureteroscopy with the addition of narrow-band imaging, which improves detection rates by 23%. Laser vaporization of the tumor (laser settings 10 Hz/1J) is an effective management strategy and Dr. Shariat emphasizes it is important to perform a no-touch technique when treating these lesions. Newer techniques include confocal laser endomicroscopy, which provides an in vivo optical biopsy of the upper tract, using a 0.85 mm probe in a standard ureteroscope. Optical coherence tomography uses 1300 nm near-infrared light, with the ability to work through a flexible or semi-rigid ureteroscope. This provides strong anatomical visualization of the tissue layers and can be used to assess stage and depth of tumor invasion into different anatomical tissue layers.
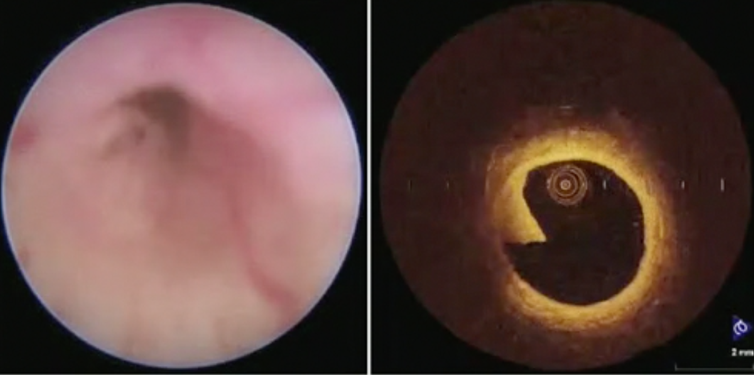
Dr. Shariat favors a re-ureteroscopy in 6-8 weeks to re-evaluate the previously treated region, as well as have the opportunity to treat any residual/recurrent tumors.
In a systematic review of oncologic outcomes for kidney-sparing surgery versus radical nephroureterectomy, Seisen et al. found no significant difference between segmental ureterectomy and radical nephroureterectomy in terms of cancer specific survival (CSS) or any other oncologic outcomes.2 Only patients with low-grade and noninvasive tumours experienced similar CSS after ureteroscopy or percutaneous treatment when compared with radical nephroureterectomy, despite an increased risk of local recurrence following endoscopic management of upper tract urothelial carcinoma.
In an effort to improve the delivery of chemotherapeutics to the upper tract and potentially decrease recurrence risk, new approaches are needed one of which is the utilization of Mitogel to improve the dwell time of mitomycin C in the upper urinary tract. MitoGel, JelmytoTM (formerly UGN-101), is a combination of mitomycin C with RTGel, a reverse-thermal hydrogel composed of a combination of polymers that allows it to exist as a liquid at cold temperatures but solidify to a gel state at body temperature.

The recently published OLYMPUS trial was a phase 3, open-label, single-arm trial designed to assess the efficacy, safety, and tolerability of UGN-101 in patients with low-grade, noninvasive upper tract urothelial cancer.3 Enrolled patients received six once-weekly instillations of UGN-101 as an induction course, administered via retrograde instillation with ureteral catheterization. Four to six weeks following initial treatment, patients received their primary disease evaluation including ureteroscopy, selective upper tract cytology, and for-cause biopsy where indicated. Complete response was defined as a negative endoscopic evaluation and the absence of histologic or cytologic evidence of disease. Among 110 patients screening, 74 were enrolled and 71 patients received treatment. Among the 71 patients who received at least one dose, 42 patients (59%, 95% CI 47-71%) had a complete response at the time of primary disease evaluation. Of the remainder, 8 (11%) had a partial response, 12 (17%) had no response, 6 (8%) had newly diagnosed high-grade disease, and 3 (4%) had an indeterminate response. Of the 42 patients with complete response, 41 entered follow-up. Of these, 29 (71%) received at least one dose of maintenance therapy and 6 (15%) were continuing on maintenance therapy at the time of data cut-off. With regard to toxicity, 67 patients (94%) experienced adverse events, and 26 (37%) patients experienced severe adverse events. Among adverse events of particular interest, renal functional impairment was noted in 14 patients (20%). There was also a significant burden of urinary tract morbidity: 68% had an adverse event related to the urinary system including 11 (23%) who did not require surgical intervention, 24 (50%) who required transient stent placement, 11 (23%) who required long-term stent placement, and 2 (4%) who required nephroureterectomy due to the need for permanent drainage as a result of ureteral stenosis.
Dr. Shariat concluded with the following take-home messages:
- There is over-treatment in ~20-30% of cases of upper tract urothelial cancer for patients undergoing radical nephroureterectomy
- Kidney-sparing management is safe in low-risk upper tract urothelial cancer
- Risk-stratification is essential and patients should have a re-ureteroscopy ~6 weeks after their treatment
- Compliance is essential as patients utilizing kidney-sparing modalities require strict long-term follow-up
Presented by: Shahrokh F. Shariat, MD, Department Chair and Professor of Urology, University of Vienna, Vienna, Austria
Written by: Zachary Klaassen, MD, MSc – Assistant Professor of Urology, Georgia Cancer Center, Augusta University/Medical College of Georgia Twitter: @zklaassen_md at the 12th European Multidisciplinary Congress on Urological Cancers (EMUC) (#EMUC20 ), November 13th - 14th, 2020
References:
- Roupret M, Babjuk M, Capoun O, et al. European Association of Urology Guidelines on Upper Tract Urothelial Carcinoma: 2020 Update. Eur Urol 2020 Jun 24:S0302-2838(20)30427-9.
- Seisen T, Peyronnet B, Dominguez-Escrig JL, et al. Oncological outcomes of kidney-sparing surgery versus radical nephroureterectomy for upper tract urothelial carcinoma: A Systematic review by the EAU Non-muscle invasive bladder cancer guidelines panel. Eur Urol 2016 Dec;70(6):1052-1068.
- Kleinmann N, Matin SF, Pierorazio PM, et al. Primary chemoablation of low-grade upper tract urothelial carcinoma using UGN-101, a mitomycin-containing reverse thermal gel (OLYMPUS): An open-label, single-arm, phase 3 trial. Lancet Oncol 2020 Jun;21(6):776-785.


