Dr. Grimm began by highlighting that BCG is the accepted first-line approach to reduce recurrence and progression among patients with high-grade non-muscle invasive bladder cancer (NMIBC). However, BCG is associated with significant toxicity, and, as such, many efforts have been undertaken to reduce the toxicity of this treatment regime. The NIMBUS trial investigated whether a reduced frequency of BCG instillation during the induction and maintenance phases would result in similar clinical efficacy to standard BCG treatment protocols with lower adverse events, inconvenience, and cost.
The investigators enrolled patients with BCG-naïve high grade non-muscle invasive bladder cancer, whether primary or recurrent disease, solitary or multiple tumors, and with or without concurrent carcinoma in situ (CIS). All patients underwent repeat Transurethral resection of bladder tumor (TURBT) confirming the absence of high-grade papillary disease.
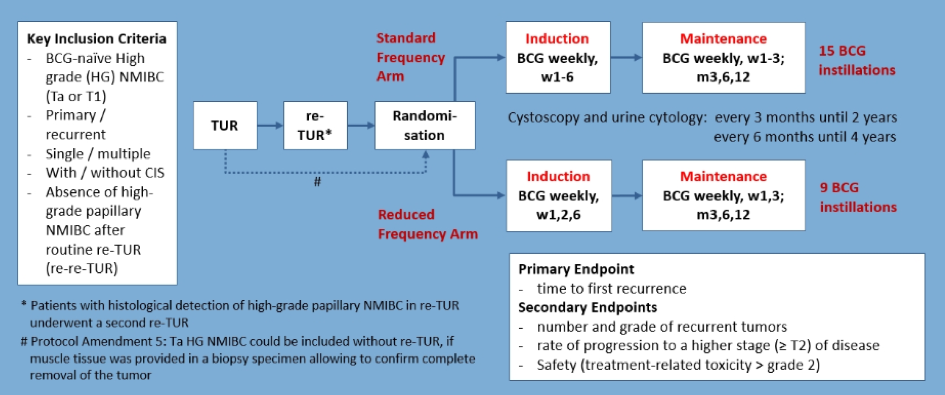
Patients were then randomized to two BCG instillation schedules. The standard BCG protocol was administered with 6 weekly induction treatments followed by 3 weekly maintenance treatments at 3, 6, and 12 months. In the intervention arm, patients received BCG at 1, 2, and 6 weeks followed by maintenance at weeks 1 and 3 at 3, 6, and 12 months. As a result, over 12 months, patients in the standard arm received 15 BCG instillations while those in the reduced frequency arm received 9 BCG instillations.
The primary outcome of interest was the time to first recurrence while key secondary outcomes included the number and grade of recurrent tumors, the rate of progression to muscle-invasive disease, and safety (including treatment-related adverse events > grade 2).
The authors powered this study with a non-inferiority design on the basis of therapeutic equivalence defined as the lower bound of the 95% confidence interval being higher than the hazard ratio of 0.75 for recurrence. This resulted in a sample size of 1000 total patients to give a power of 80%. Recruitment was delayed on account of BCG shortage which modified statistical assumptions. As a result, a total of 824 patients were then targeted. However, at the time of data cut-off (July 1, 2019), the authors report on data from 345 randomized patients.
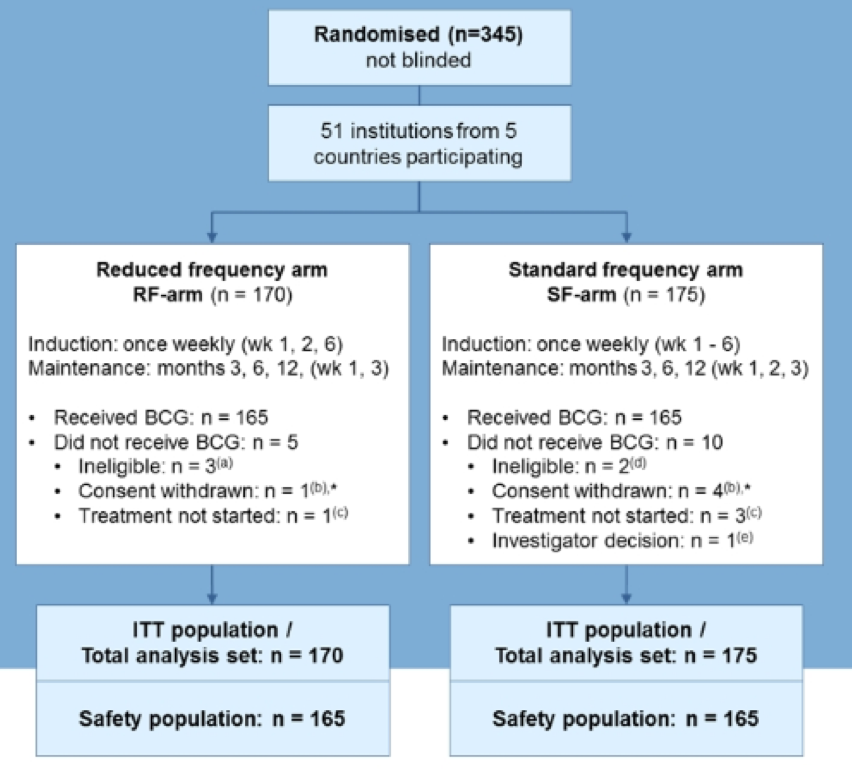
Baseline characteristics were well balanced between the two groups, including center of randomization, Ta vs T1 disease, concomitant CIS, single vs multiple tumors, and BCG strain (Medac, Connaught, or Tice).
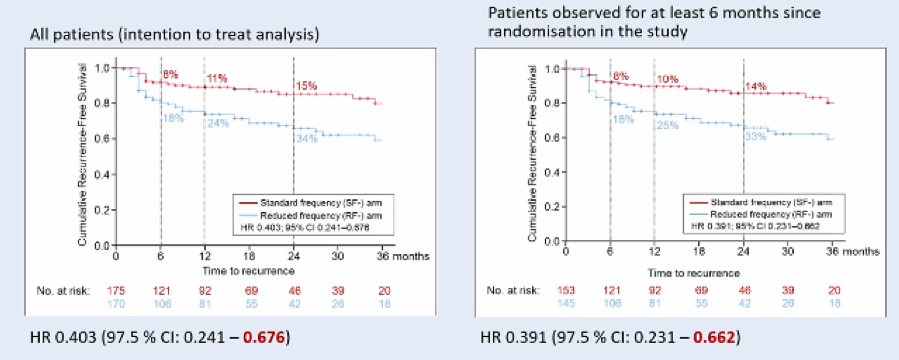
With a median follow-up of 12 months, in the overall population, 67 patients (19.4%) of patients had recurrence and 7 (2%) had progression to muscle-invasive disease. Rates of recurrence were somewhat higher in patients in the reduced frequency arm (27% vs 12%) while progression to muscle-invasive disease was somewhat lower in these patients (0.6% vs 3.4%).
Assessing time to recurrence utilizing the Kaplan Meier method, the authors found a significantly lower recurrence-free survival among patients who received the reduced frequency of BCG.
Dr. Grimm emphasized that these data demonstrate relatively low rates of recurrence among patients receiving the standard BCG treatment schedule, following complete TURBT.
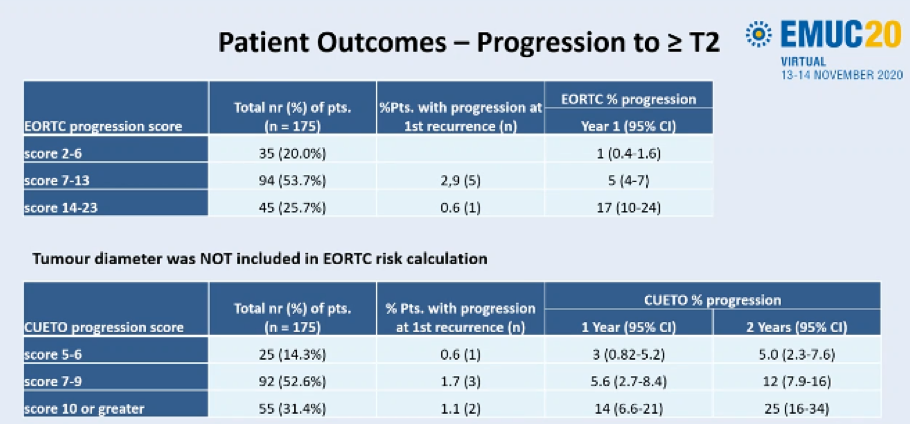
In post hoc analyses, the authors compared recurrence risks observed among patients receiving standard schedule BCG in this cohort to those predicted by commonly utilized nomograms including the European Organization for Research and Treatment of Cancer (EORTC) score and CUETO score. In each case (apart from Club Urológico Español de Tratamiento Oncológico (CUETO) low-risk patients, score 0-4), recurrence rates were lower in the NIMBUS cohort than predicted which Dr. Grimm suggested that this was due to the routine use of repeat TURBT in this study cohort.
Dr. Grimm then presented data on rates of progression to muscle-invasive disease. Again utilizing predicted rates based on EORTC progression score and CUETO progression score, Dr. Grimm highlighted that rates in this cohort were lower than expected.
Dr. Grimm closed by highlighting that a reduced frequency schedule of BCG treatment in patients with high-grade NMIBC is inferior to the standard schedule and should not be used, in spite of reduced toxicity.
Presented by: Marc-Oliver Grimm, MD, Professor, Otto-Schott-Institut der Friedrich-Schiller-Universität Jena, Jena, Germany
Written by: Christopher J.D. Wallis, Urologic Oncology Fellow, Vanderbilt University Medical Center Contact: @WallisCJD on Twitter at the 12th European Multidisciplinary Congress on Urological Cancers (EMUC) (#EMUC20 ), November 13th - 14th, 2020
Related Content:
ASCO GU 2020: Treatment of High-Grade Non-Muscle Invasive Bladder Carcinoma: Results of the Phase III Clinical Trial (NIMBUS)
EMUC 2020: Novel Options for the Treatment of High-Risk Non-muscle Invasive Bladder Cancer


