Up until recently, there had been no treatment changes in the last 30 years for second-line therapy for metastatic urothelial carcinoma (MUC). There had been no 2nd line FDA approved treatments for this entity for a significant period. However, in the last year aside from the discovery of the important role of immune checkpoint inhibitors, there have been several additional novel agents that have demonstrated promising results. One of those novel agents is Enfortumab vedotin (EV), which is an antibody-drug conjugate that delivers a microtubule-disrupting agent to tumors expressing Nectin-4 (which is mostly overexpressed in urothelial cancers). It was found to be expressed in 93% of MUC samples.1 The EV-101 phase 1 study was a dose escalation and expansion study, with EV administered on days 1,8, and 15 of a 28-day cycle. This drug showed promising results and phase 2/3 studies are ongoing.
Another novel agent is Erdafitinib in FGFR altered MUC. It is an experimental small molecule inhibitor of FGFR. A phase 2 study evaluating multiple doses and schedules of this drug demonstrated an impressive objective response rate (ORR) of 40%. There are studies evaluating the effect of other FGFR inhibitors (Rogaratibin), and active agents in the RTK/RAS pathway, and PI3K/AKT/mTOR pathway.
Dr. Bellmunt moved on to discuss the role of immune checkpoint inhibitors in MUC. There are currently 7 US FDA, and 5 EAU approved treatments for use in urothelial carcinoma (Figure 1). The immune checkpoint inhibitors used in the platinum-refractory setting are shown in figure 2, and those used as the first line in cisplatin-ineligible patients are shown in figure 3. The significantly fast approval of this agents by the FDA and EAU is quite impressive. However, the FDA had recently issued an alert (May 2018) that monotherapy with pembrolizumab or atezolizumab given in a trial to patients with low PD-L1 status had decreased their survival compared to patients who received cisplatin or carboplatin-based chemotherapy. Therefore, the recommendation had changed, and in the ongoing trials, these treatments should be offered only to patients with high PD-L1 status. For the low PD-L1 status patients, carboplatin based-chemotherapy should continue to be given.
Figure 1 – Approved Immune Checkpoint Inhibitors in Urothelial Carcinoma:
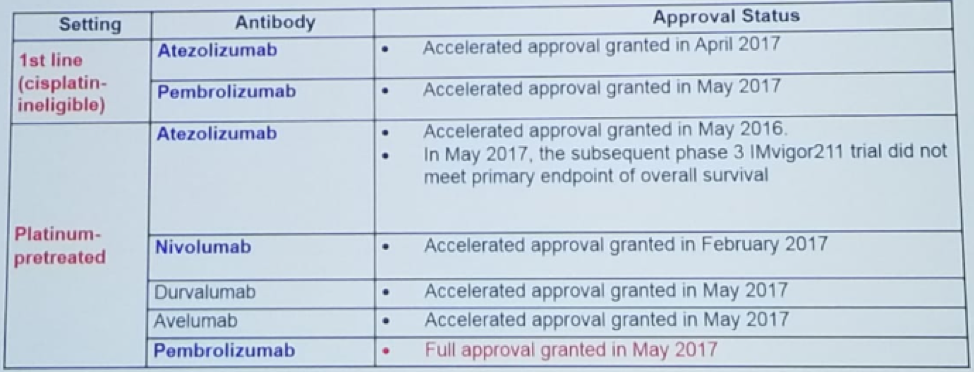
Figure 2 – Immune Checkpoint Inhibitors in the Platinum-refractory Setting:
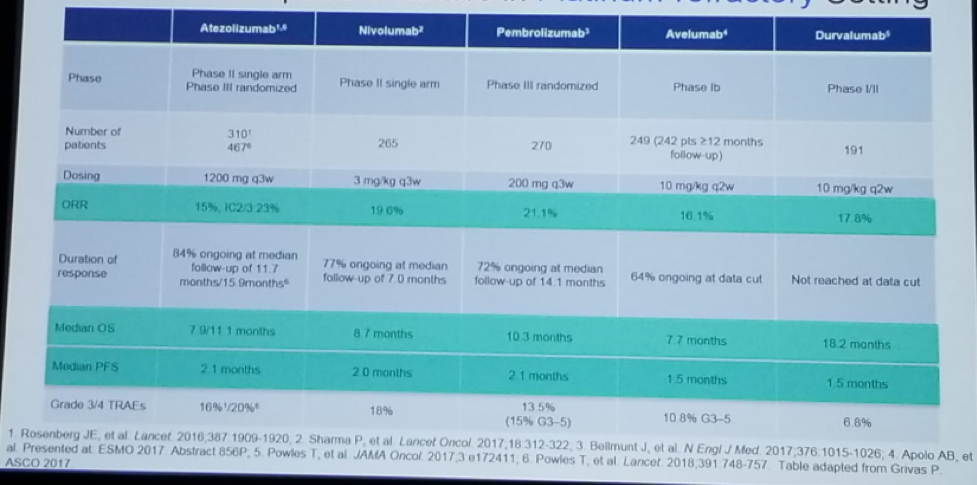
Figure 3 – Immune Checkpoint Inhibitors as the First Line in Cisplatin-ineligible Patients:
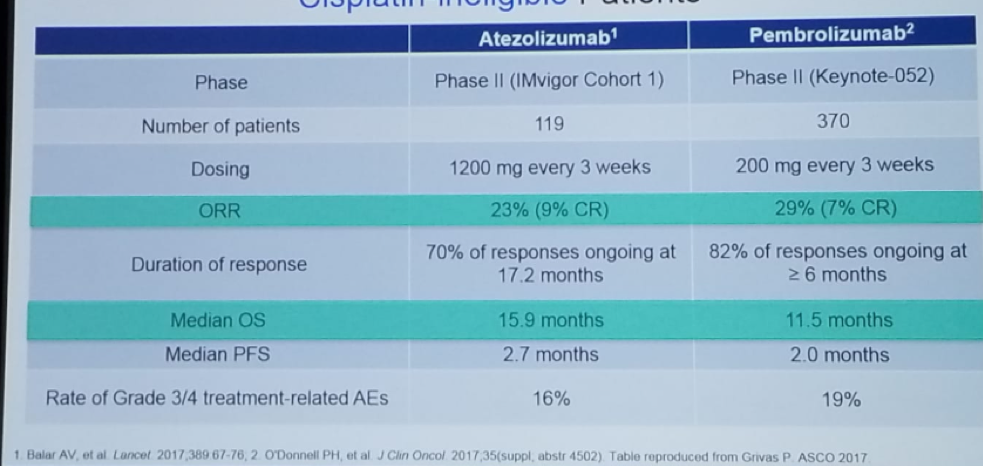
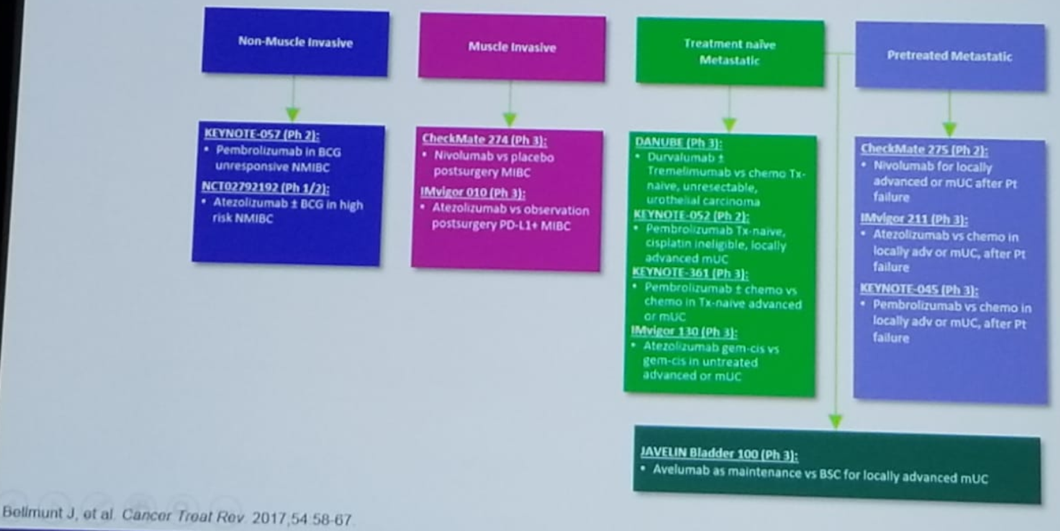
There have been two second-line phase 3 trials in MUC: The Keynote-045 trial, comparing pembrolizumab to the standard of care, and IMvigor211 trial, comparing atezolizumab to the standard of care. These trials demonstrated improved overall survival for the immune checkpoint inhibitors in the intention to treat analyses. Figure 4 nicely demonstrates the evolution of systemic therapy for urothelial cancer as of 2019. Figure 5 demonstrates all the ongoing trials for all stages of urothelial carcinoma: including non-muscle invasive, muscle-invasive, treatment-naïve metastatic patients, and pretreated metastatic patients.
Figure 4- The Evolution of Systemic Therapy for Urothelial Carcinoma as of 2019:
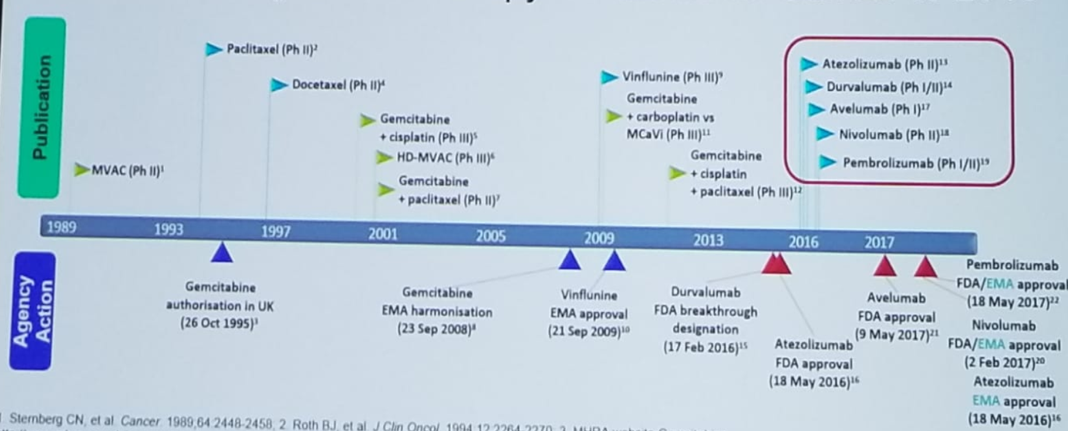
Figure 5 – Ongoing trials in bladder cancer – what does the future hold?
An important trial to mention is the PURE-01 (NCT02736266) trial of neoadjuvant pembrolizumab before radical cystectomy for muscle-invasive bladder cancer.2 This trial showed a complete pathologic response to pembrolizumab in 39.5% of patients. Another trial in the same setting is the ABACUS phase 2 single-arm trial of neoadjuvant atezolizumab before radical cystectomy.3 This trial demonstrated 29% pathological complete response in all patients together, 40% complete response in PD-L1 positive patients only, and 16% complete response in PD-L1 negative patients only. There are currently other risk-adapted neoadjuvant studies in development.
Lastly, for the setting of non-muscle invasive BCG refractory bladder cancer, the KEYNOTE-057 single arm phase 2 trial was developed (NCT02625961). 4 In this study, pembrolizumab demonstrated a promising complete response rate of 38.8%.
The last topic discussed by Dr. Bellmunt was the need for biomarkers in all various bladder cancer disease settings. One such suggested biomarker is the tumor mutational burden, which correlates with response to immune checkpoint inhibitor blockade in multiple solid tumors.
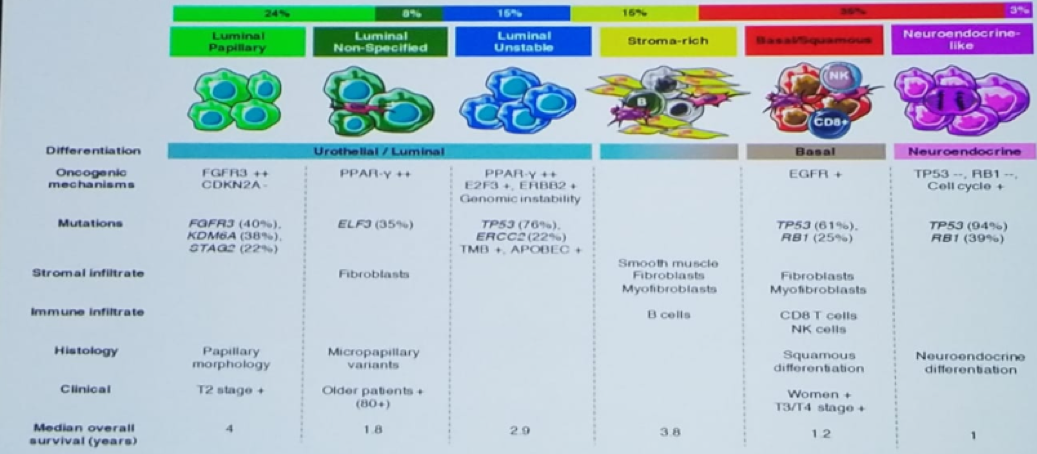
Dr. Bellmunt concluded his talk describing the new consensus molecular classification of muscle-invasive bladder cancer, which now demonstrates six distinct subtypes of bladder cancer (basal/Squamous, Luminal Papillary, Luminal Unstable, Stroma-rich, Luminal Non-Specified, and Neuroendocrine-like), as seen in figure 6. These would help direct proper treatment for the right subtype of bladder cancer. According to Dr. Bellmunt, in the future we will see that the combination of various immunotherapy agents will be the key to improving outcomes. An enormous challenge in the field is to find which combination works best in which tumor, and which patient within that tumor type. There is still a need to better understand the biology behind immunotherapy and discover effective biomarkers that would aid diagnosis and treatment decisions.
Figure 6 – The consensus molecular classification of muscle-invasive bladder cancer:
References:
- Petrylak et al. JCO 2017
- Necchi A et al. ASCO 2018 abstract 4507
- Powles T. et al. ASCO 2018 abstract 4506
- De Wit R. et al. Annals of Oncology 2018
Written by: Hanan Goldberg, MD, Urologic Oncology Fellow (SUO), University of Toronto, Princess Margaret Cancer Centre, Twitter: @GoldbergHanan at the CUOS – Canadian Uro-Oncology Summit 2019, #CUOS19 January 10-12, 2019 Westin Harbour Castle, Toronto, Ontario, Canada

