As an early initiative of the GURC Best Practice group, the authors present a descriptive analysis of provincial reimbursement policies for mCRPC therapies. Using the Canadian Agency for Drugs and Technologies in Health (CADTH) pan-Canadian Oncology Drug Review (pCODR), provincial funding summaries, provincial cancer care guidelines and formularies, pharmaceutical manufacturers, and the GURC network of clinicians, provincial reimbursement policies were reviewed for approved mCRPC therapies where available. Since Yukon, Northwest Territories and Nunavut do not participate in pCODR and limited information was available through supplemental sources, they were excluded from this study.
To summarize pictographically the interprovincial variation in treatment access, the authors showed a heat map that depicts key differentiating factors related to access as a composite score (Figure 1).
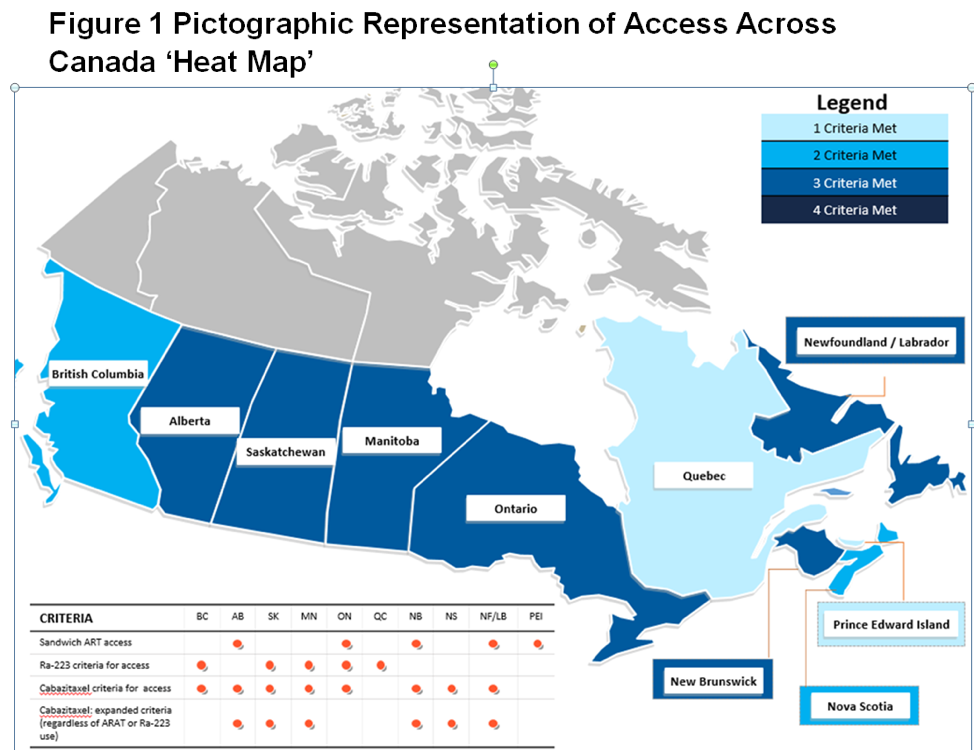
As the results show, interprovincial disparities were observed in access to androgen receptor-axis-targeted (ARAT) therapies, radium-223, and cabazitaxel. (Table 1, 2, 3).
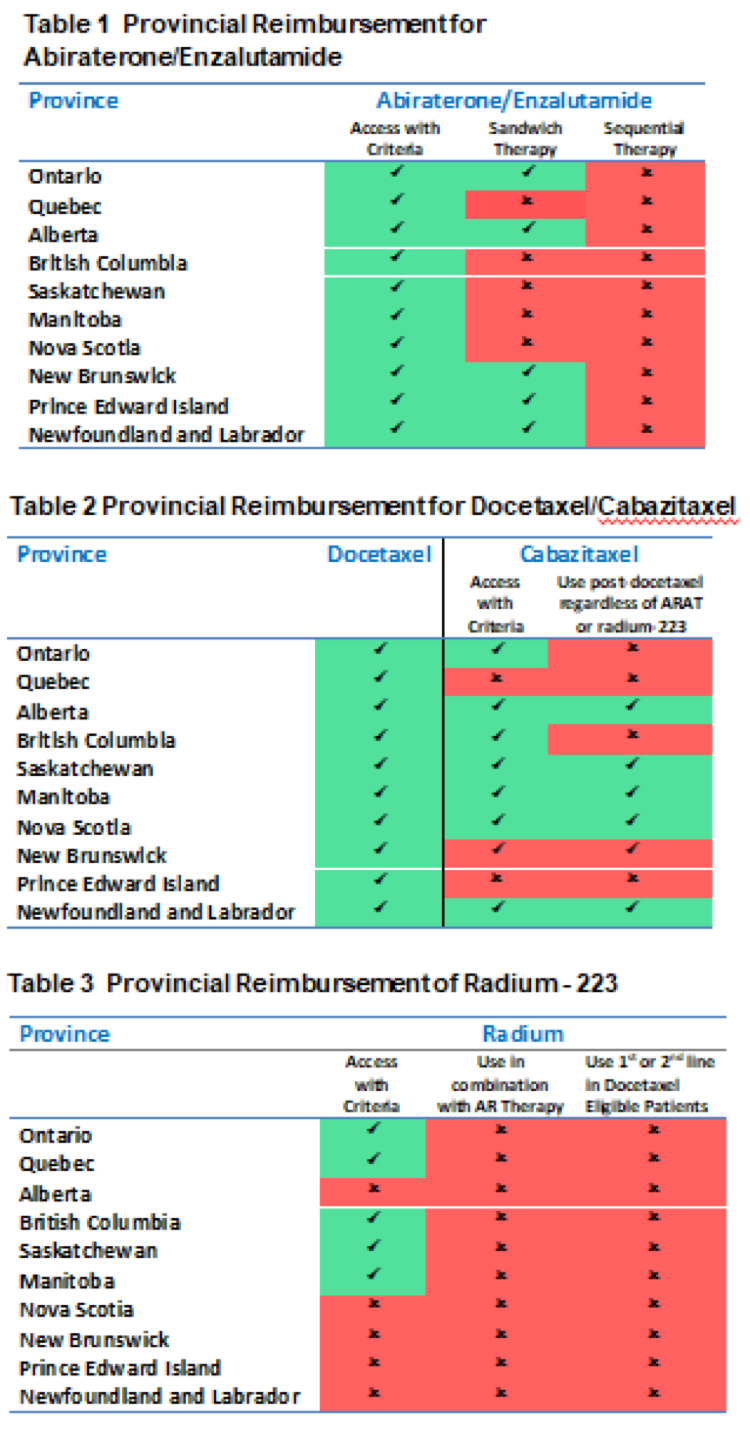
However, Docetaxel and ARAT therapy were shown to be readily available across all the provinces. However, ‘Sandwich’ therapy, where one ARAT is used pre-chemotherapy and a second is used after progression on chemotherapy is funded in only 5 provinces. Sequential ARAT, where the use of 1 ARAT therapy is followed by another ARAT therapy is not allowed in any of the provinces studied (Figure 1). Lastly, Ra-223 is funded in 5 provinces to varying degrees and not available in 5 provinces, leaving approximately 20% of Canadians without access to Ra-223 (Figure 1).
Interestingly, several provinces have instituted restrictions on the types of physicians who can prescribe mCRPC therapies. In Alberta, prescribing is limited to approved designated physicians. In Manitoba, prescribers must be affiliated with the provincial cancer center. Prince Edward Island only designated prescribers may prescribe mCRPC treatments.
In conclusion, while all provinces have access to docetaxel and ARATs, access to various treatments, such as sandwiching sequential ARATs with docetaxel is funded only in some of the provinces. Ra-223 and cabazitaxel access vary considerably across Canada. These inequalities in access to life-prolonging therapies could potentially lead to disparities in survival and quality of life among mCRPC patients.
Presented by: Dixon T.S. Woon, MD, Princess Margaret Hospital, University of Toronto, Toronto, ON, Canada
Co-Authors: Thenappan Chandrasekar1, Lorne Aaron2, Naveen S. Basappa3, Kim N. Chi4, Henry J. Conter5, Brita Danielson3, Sebastien J. Hotte6, Shawn Malone7, Fred Saad8, Bobby Shayegan6, Laura Park-Wyllie9, Robert Hamilton1.
Author Information:
1. Princess Margaret Hospital, University of Toronto, Toronto, ON, Canada
2. Service d-Urologie and Centre de la Prostate, Hôpital Charles LeMoyne, Longueuil, QC, Canada
3. Cross Cancer Institute, University of Alberta, Edmonton, AB, Canada
4. BC Cancer Agency, University of British Columbia, Vancouver, BC, Canada
5. William Osler Health System, University of Western Ontario, Brampton, ON, Canada
6. Juravinski Cancer Centre, McMaster University, Hamilton, ON, Canada
7. The Ottawa Hospital, University of Ottawa, Ottawa, ON, Canada
8. Centre Hospitalier de l’Université de Montréal, University of Montreal, Montreal, QC, Canada
9. Medical Affairs, Janssen Inc., Toronto, ON, Canada
Written By: Hanan Goldberg, MD, Urologic Oncology Fellow (SUO), University of Toronto, Princess Margaret Cancer Centre, @GoldbergHanan at the 73rd Canadian Urological Association Annual Meeting - June 23 - 26, 2018 - Halifax, Nova Scotia

