“Patient decision aids” are structured clinical tools that facilitate shared decision-making – they present therapeutic options, including their risks and benefits, in an evidence-based fashion and help patients communicate their values. The group from Ottawa worked on creating such an aid for this challenging situation which all urologic oncologists and most urologists face regularly.
They based their model off of the International Patient Decision Aids Standards (IPDAS) and the Ottawa Decision Support Framework – these are international and regional guidelines to help physicians create an aid that doesn’t inappropriately influence the patient. They focused their efforts on cT1b-cT2 tumors, those which are most likely to encounter this decision point – larger tumors are more likely to require nephrectomy (even open nephrectomy) while smaller tumors are often offered more conservative management and partial nephrectomy is the preferred approach. The content of the decision aid was agreed upon by content and methodological experts using an iterative feedback process.
Once the content was created, a mixed methods survey was created to assess the decision aid. Both patients and urologists were recruited to evaluate the decision aid as this is a shared-decision making tool.
A structured patient decision aid presented evidence on options, including probabilities of benefits and risks. Open partial nephrectomy, laparoscopic radical nephrectomy, and observation were the three main options offered to patients. The outcomes for which probabilities were generated included: bleeding, urine leak, length of stay, renal failure, and survival. These are all items we routinely discuss with patients, but perhaps not in a standardized way – which may lend itself to bias. Simple language and pictures were used to present data at a level suitable for a wide range of patients – which is important for patient understanding.
An example of the picture aid is below:
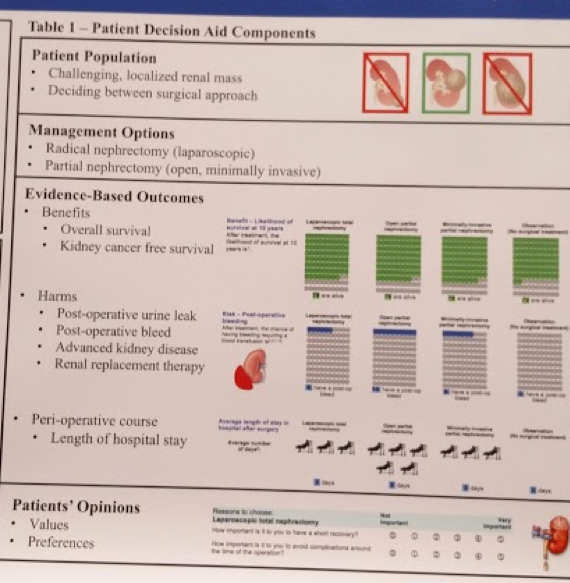
They will then use a validated screening tool (SURE test) to assess patients’ decisional conflict. Knowledge questions were included to verify patients’ understanding and to see how well the decision aid transferred information.
Initial testing has demonstrated good results – alpha testing (11 urologists, 8 patients, 3 patient advocates). They felt the length was appropriate and that the language was easy to follow. Most felt strongly that it would be useful in clinical use. One area to improve was the inclusion of robotic partial nephrectomy.
Based on this they will finalize the final decision aid and move to beta testing.
This is a very interesting study with significant clinical impact. Also presented at other major cancer conferences, it has been well-received. We look forward to the results of their prospective evaluation of the aid in clinical practice!
Presented by: McAlpine, Kristen, Resident, The Ottawa Hospital, Ottawa, Canada
Co-Authors: Kristen McAlpine, Rodney Breau1,2, Dawn Stacey2, Christopher Knee1,2, Luke Lavallee1,2.
Author Information:
1. Division of Urology, University of Ottawa, Ottawa, ON, Canada
2. The Ottawa Hospital Research Institute, Ottawa, ON, Canada
Written By: Thenappan Chandrasekar, MD, Clinical Fellow, University of Toronto Twitter: @tchandra_uromd at the 73rd Canadian Urological Association Annual Meeting - June 23 - 26, 2018 - Halifax, Nova Scotia

