The debates in advanced RCC management used to be limited to “Is high-dose IL-2 appropriate for everyone?” in the 1990’s, but in the past few years has been thrown into disarray with a dizzying number of new agents in this space. Ultimately, the goal is determining which agents allow our patients to live longer (ideally for a cure) and live better.
His talk focused on three major developments (amongst others):
1. ESMO 2017: Nivo/Ipi vs. sunitinib primary analysis (CheckMate 214 study)
2. SITC 2017: Nivo/ipi vs sunitinib secondary analysis (CheckMate 214 study)
3. GU ASCO 2018: Bevacizumab/atezolizumab vs. sunitinib primary analysis (ImMotion 151)
All of these are in the first line setting, though he did note many of the second line therapies are now moving up to first line. He also noted that all the studies are being compared to sunitinib, but there is increasing evidence this is no longer the standard of care.
The CheckMate 214 study compared Nivo/ipi (followed by nivo maintenance) against sunitinib (standard dosing) in the first line setting (treatment naïve advanced or metastatic RCC).
His key points:
1. Overall survival for IMDC intermediate/poor risk patients was significantly higher in the Nivo/Ipi arm than in the sutent arm (NR vs. 26.0 months, HR 0.63, p <0.001)
2. However, in IMDC favorable risk patients, sunitinib actually fared better (ORR 52% sutent vs. 29% Ipi/Nivo, 10 month PFS benefit to sunitinib)
3. Twice as many patients discontinued drug in the Nivo/Ipi arm than in the sunitinib arm
4. 60% of patients in the Nivo/Ipi arm required IV corticosteroids for adverse effects
5. PFS was significantly better in PD-L1+ patients (HR 0.48), but not in PD-L1- patients (HR 1.0); however, OS benefit was significantly better with Nivo/Ipi in both subsets, but more pronounced in PD-L1+ patients
In ImMotion 151, bevacizumab/atezolizumab (Bev/Atezo) was compared against standard dosing sunitinib in the first line setting (treatment naïve advanced or metastatic RCC) – however, it wasn’t just clear cell histology; sarcomatoid histology was also allowed.
Key points:
1. PFS was significantly better with bev/atezo in the PD-L1+ and intent-to-treat analysis (on independent review)
2. Objective response rate higher in the Bev/Atezo cohort – notably, CR rates double the sunitinib arm (9% vs 4%)
3. In contrast to Nivo/Ipi, Bev/Atezo was very well tolerated with less adverse effects that sunitinib – and only 16% required IV steroids
4. Interestingly, the investigator-assessed outcomes did not quite sync with independent review committee – though he noted that they trended the same direction, which was reassuring, likely suggesting a true signal. IRC and INV assessment of PFS and OS benefit were generally consistent with ITT population results.
5. OS data was immature – but suggested a trend towards favoring Bev/Atezo.
When comparing the two first-line comparators, they are relatively similar – except the adverse event profile which strongly favors bev/atezo.
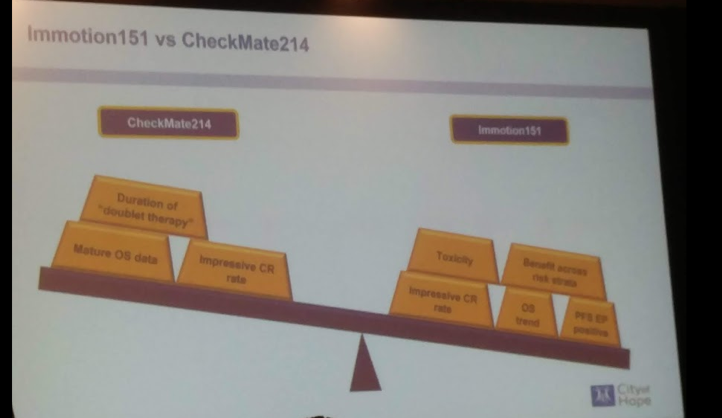
He did point out again that TKI’s should not be discounted – sunitinib is likely not the best comparator. CABOSUN, a phase III study that compared cabozantinib and sunitinib in the first line setting, clearly demonstrated that CAB had better PFS (8.6 vs. 5.3 months, HR 0.48, p<0.0001), particularly in patients with bone metastases and poor-risk disease.
His current algorithm for first and second line therapy is as follows:

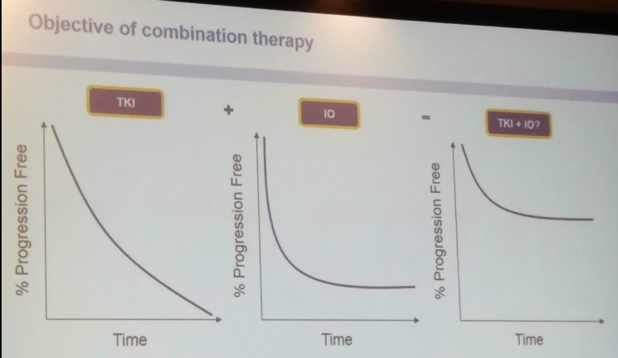
The future lies in the combination of TKI’s (as bevacizumab is an earlier targeted agent, not a TKI) and immune checkpoint blocker – there is a biologic rationale for this.
I liked this slide of his – combining the two will leverage the PFS survival benefit of TKI’s beyond the first few months and the durable response of IO’s. Hopefully to provide more patients with a long-term cure.
He then briefly reviewed the early data from early phase TKI/IO therapies, all with promising early results, including (but not limited to): axitinib/pembrolizumab, cabozantinib/nivolumab +/- Ipilimumab, tivozanib/nivolumab.
His last focus was a plug for a study that is rapidly accruing. Interestingly, it allows for adjuvant/maintenance therapy following Nivo/Ipi front-line treatment based on the response. The protocol for the study is below:

Presented by: Sumanta Pal, MD, City of Hope, Los Angeles, California
Written By: Thenappan Chandrasekar, MD, Clinical Fellow, University of Toronto Twitter: @tchandra_uromd at the 73rd Canadian Urological Association Annual Meeting - June 23 - 26, 2018 - Halifax, Nova Scotia
Read More:
Role of Cytoreductive Nephrectomy in 2018 - Ricardo Rendon, MD

