(UroToday.com) The 2023 American Urological Association (AUA) annual meeting held in Chicago, IL between April 28 and May 1st, 2023, was host to a plenary session, with Dr. Jonathan Coleman presenting the first iteration of the AUA/SUO guidelines for the diagnosis and management of non-metastatic upper tract urothelial carcinoma (UTUC).
Dr. Coleman began by noting that UTUC is a rare disease that is pathologically, genomically, biologically, and clinically distinct from urothelial carcinoma of the bladder, with a 5-year mortality rate of >50%, compared to <25% for bladder cancer.1 Furthermore, treatment of such patients is challenging due to the inherent risk of renal functional loss with current management strategies and associated patient morbidity with extirpative surgery. As such, over the last few years, there has been a push within the AUA, in collaboration with the SUO, to develop clinical guidelines framework that addressed the diagnosis and management of non-metastatic UTUC.
Next, Dr. Coleman went through the current guideline statements as follows:
Diagnosis and Evaluation- For patients with suspected UTUC, a cystoscopy and cross-sectional imaging of the upper tract with contrast, including delayed images of the collecting system and ureter, should be performed.
- Clinicians should evaluate patients with suspected UTUC with diagnostic ureteroscopy and biopsy of any identified lesion and cytologic washing from the upper tract system being inspected.
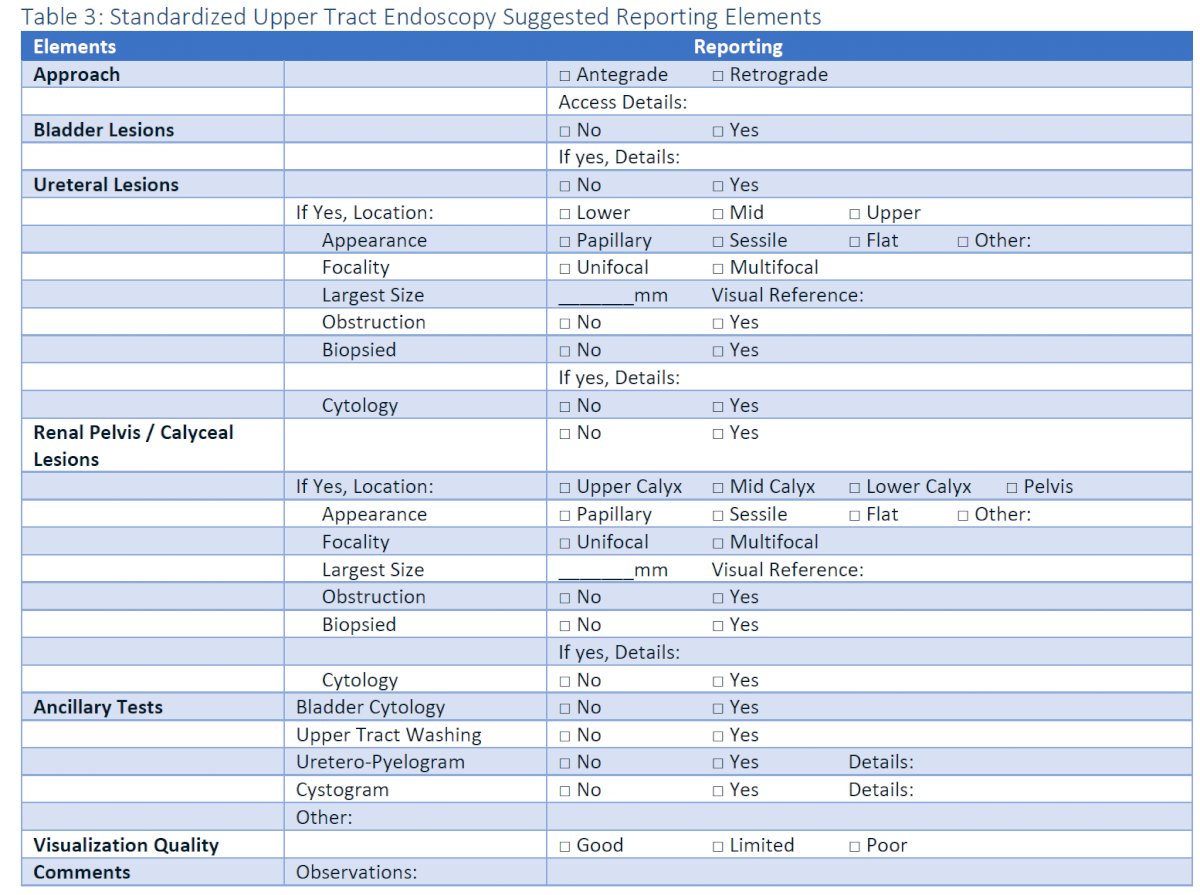
- In patients who have concomitant lower tract tumors (i.e., bladder and/or urethra) discovered at the time of ureteroscopy, the lower tract tumors should be managed in the same setting as the ureteroscopy.
- In cases of existing ureteral strictures or difficult access to the upper tract, clinicians should minimize risk of ureteral injury by using gentle dilation techniques such as temporary stenting (pre-stenting) and limit use of aggressive dilation access techniques such as ureteral access sheaths.
- In cases where ureteroscopy cannot be safely performed or is not possible, an attempt at selective upper tract washing or barbotage for cytology may be made and pyeloureterography performed in cases where good quality imaging such as CT or MR urography cannot be obtained.
- At the time of ureteroscopy for suspected UTUC, clinicians should not perform ureteroscopic inspection of a radiographically and clinically normal contralateral upper tract.
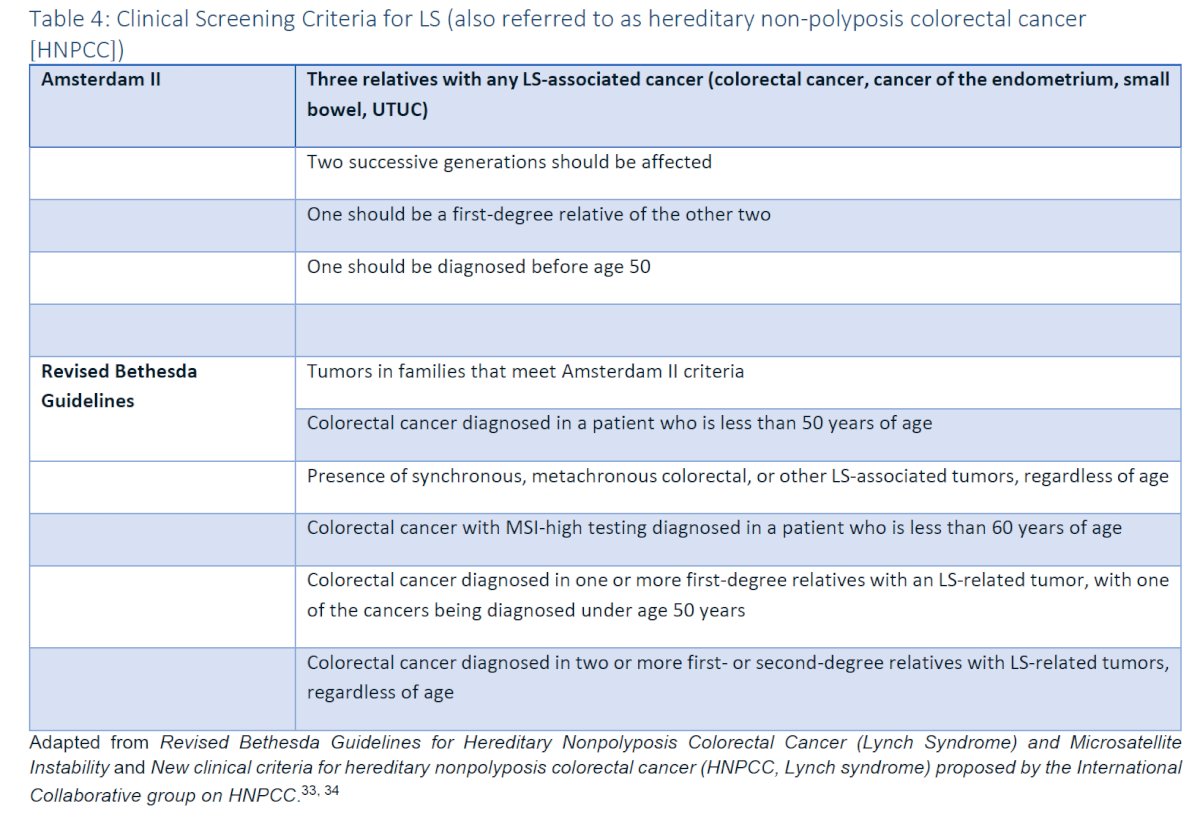
- For patients with suspected/ diagnosed UTUC, clinicians should obtain a personal and family history to identify known hereditary risk factors for familial diseases associated with Lynch Syndrome (LS) (colorectal, ovarian, endometrial, gastric, biliary, small bowel, pancreatic, prostate, skin and brain cancer) for which referral for genetic counseling should be offered.
- Universal histologic testing of UTUC with additional studies, such as immunohistochemical (IHC) or microsatellite instability (MSI), should be performed to identify patients with high probability of Lynch-related cancers whom clinicians should refer for genetic counseling and germline testing.
Risk Stratification
- At the time of identified UTUC, clinicians should perform a standardized assessment documenting clinically meaningful endoscopic (focality, location, appearance, size) and radiographic (invasion, obstruction, and lymphadenopathy) features to facilitate clinical staging and risk assessment.
- Following standardized assessment, clinicians should risk-stratify patients as “low” or “high” risk for invasive disease (pT2 or greater) based on obtained endoscopic, cytologic, pathologic, and radiographic findings. Further stratification into favorable and unfavorable risk groups should then be based on standard identified features, as summarized in the table below:

- Patients with UTUC should be assessed prior to surgery for the risk of post-NU CKD or dialysis.
- Clinicians should provide patients with a description of the short- and long-term risks associated with recommended diagnostic and therapeutic options. This includes the need for endoscopic follow-up, clinically significant strictures, toxicities associated with surgical treatment and side effects from neoadjuvant and adjuvant therapies.
- Tumor ablation should be the initial management option for patients with LR favorable UTUC.
- Tumor ablation may be the initial management option offered to patients with LR unfavorable UTUC and select patients with HR favorable disease who have low-volume tumors or cannot undergo RNU.
- Tumor ablation may be accomplished via a retrograde or antegrade percutaneous approach and repeat endoscopic evaluation should be performed within three months.
- Following ablation of UTUC tumors and after confirming there is no perforation of the bladder or upper tract, clinicians may instill adjuvant pelvicalyceal chemotherapy or intravesical chemotherapy to decrease the risk of urothelial cancer recurrence.
- Pelvicalyceal therapy with BCG may be offered to patients with HR favorable UTUC after complete tumor ablation or patients with upper tract carcinoma in situ (CIS).
- When tumor ablation is not feasible or evidence of risk group progression is identified in patients with LR UTUC, surgical resection of all involved sites either by a radical nephroureterectomy or segmental resection of the ureter should be offered.
- Clinicians may offer watchful waiting or surveillance alone to select patients with UTUC with significant comorbidities, competing risks of mortality, or at significant risk of End-Stage Renal Disease (ESRD) with any intervention resulting in dialysis.
- Clinicians should recommend a radical nephroureterectomy or segmental ureterectomy for surgically eligible patients with HR UTUC.
- For surgically eligible patients with HR and unfavorable LR cancers endoscopically confirmed as confined to the lower ureter in a functional renal unit, distal ureterectomy and ureteral reimplantation is the preferred treatment.
- When performing a nephroureterectomy or distal ureterectomy, the entire distal ureter including the intramural ureteral tunnel and ureteral orifice should be excised, and the urinary tract should be closed in a watertight fashion.
- In patients undergoing RNU or SU (including distal ureterectomy) for UTUC, a single dose of perioperative intravesical chemotherapy should be administered in eligible patients to reduce the risk of bladder recurrence.
- For patients with LR UTUC, clinicians may perform LND at time of the nephroureterectomy or segmental ureterectomy.
- For patients with HR UTUC, clinicians should perform LND at the time of NU or ureterectomy.
- Clinicians should offer cisplatin-based NAC to patients undergoing a radical nephroureterectomy or ureterectomy with HR UTUC, particularly in those patients whose post-operative eGFR is expected to be less than 60 mL/min/1.73m2 or those with other medical comorbidities that would preclude platinum-based chemotherapy in the post-operative setting.
- Clinicians should offer platinum-based adjuvant chemotherapy to patients with advanced pathological stage (pT2–T4 pN0–N3 M0 or pTany N1–3 M0) UTUC after RNU or ureterectomy who have not received neoadjuvant platinum-based therapy.
- Adjuvant nivolumab therapy may be offered to patients who received neoadjuvant platinum-based chemotherapy (ypT2–T4 or ypN+) or who are ineligible for or refuse perioperative cisplatin (pT3, pT4a, or pN+).
- In patients with metastatic UTUC, radical nephroureterectomy or ureterectomy should not be offered as initial therapy.
- Patients with clinical, regional node-positive (cN1-3, M0) UTUC should initially be treated with systemic therapy. Consolidative radical nephroureterectomy or ureterectomy with lymph-node dissection may be performed in those with a partial or complete response.
- Patients with unresectable UTUC (including those who are ineligible or refuse surgery [RNU or ureterectomy]) should be offered a clinical trial or best supportive care including palliative management (radiation, systemic approach, endoscopic, or ablative) for refractory symptoms such as hematuria.
- Low-risk patients managed with kidney sparing treatment should undergo a follow-up cystoscopy and upper tract endoscopy within one to three months to confirm successful treatment. Once confirmed, these patients should undergo continued cystoscopic surveillance of the bladder at least every six to nine months for the first two years and then at least annually thereafter. Endoscopy should be repeated at six months and one year. Upper tract imaging should be performed at least every six to nine months for two years, then annually up to five years. Surveillance after five years in the absence of recurrence should be based on shared decision-making between the patient and clinician.
- High-risk patients managed with kidney sparing treatment should undergo a follow-up cystoscopy and upper tract endoscopy with cytology within one to three months. Patients with no evidence of disease should undergo cystoscopic surveillance of the bladder and cytology at least every three to six months for the first three years and then at least annually thereafter. Endoscopy should be repeated at least at six months and one year. Upper tract imaging should be performed every three to six months for three years, then annually up to five years. Surveillance after five years in the absence of recurrence should be encouraged and based on shared decision-making between the patient and clinician.
- Patients who develop urothelial recurrence in the bladder or urethra or positive cytology following treatment for UTUC should be evaluated for possible ipsilateral recurrence or development of new contralateral upper tract disease.
- After NU, patients with <pT2 N0/M0 disease should undergo surveillance with cystoscopy and cytology within three months after surgery, then repeated based on pathologic grade. For LG this should repeated at least every six to nine months for the first two years and then at least annually thereafter. For HG, this should be repeated at least every three to six months for the first three years and then at least annually thereafter. Due to the metastasis risk and estimated 5% probability for contralateral disease, cross-sectional imaging of the abdomen and pelvis should be done within 6 months after surgery and then at least annually for a minimum of 5 years. Surveillance after five years in the absence of recurrence should be encouraged and based on shared decision-making between the patient and clinician
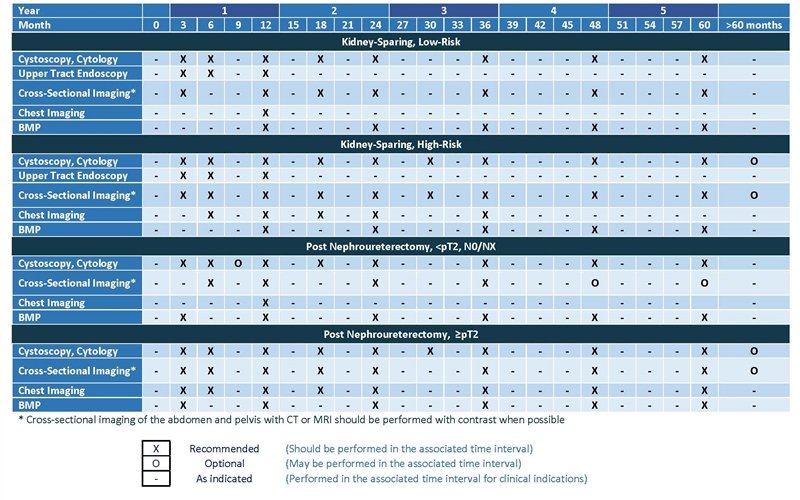
- For Patients who have undergone NU for >pT2 Nx/0 disease, a clinician should perform surveillance cystoscopy with cytology at three months after surgery, then every three to six months for 3 years, and then annually thereafter. Cross-sectional imaging of the abdomen and pelvis with multiphasic contrast-enhanced CT urography should be performed every three to six months for years one and two, every six months at year three, and annually thereafter to year five. A clinician should perform chest imaging, preferably with chest CT, every 6-12 months for the first 5 years. Beyond five years after surgery in patients without recurrence, ongoing surveillance with cystoscopy and upper tract imaging may be continued on an annual basis according to principles of shared/informed decision-making.
- For patients with reduced or deteriorating renal function following a nephroureterectomy or other intervention, clinicians should consider referral to nephrology.
- Clinicians should discuss disease-related stresses and risk factors and encourage patients with urothelial cancer to adopt healthy lifestyle habits, including smoking cessation, exercise, and a healthy diet, to promote long-term health benefits and quality of life.
Dr. Coleman highlighted that future research in this field will need to focus on:
- Urinary biomarkers that identify genetic mutations that may guide treatment decisions (e.g., FGFR3 mutations) and may have improved sensitivity/specificity performance characteristics compared to current cytology testing
- Newer instrumentation modalities to allow for improved safe and effective tissue sampling
- Ablative treatments akin to Jelmyto® or thermal ablative energy devices such as thulium:YAG laser and photodynamic treatments
Dr. Coleman concluded by emphasizing the importance of a multi-disciplinary care approach to the management of such patients.
Presented by: Jonathan A. Coleman, MD, Associate Professor, Department of Urology, Memorial Sloan Kettering Cancer Center, New York, NY
Written by: Rashid K. Sayyid, MD, MSc – Society of Urologic Oncology (SUO) Clinical Fellow at The University of Toronto, @rksayyid on Twitter during the 2023 American Urological Association (AUA) Annual Meeting, Chicago, IL, April 27 – May 1, 2023References:
- SEER: Renal pelvis seer 5-year relative survival rates, 2012-2018: National Cancer Institute, vol. 2022, 2018
- Coleman JA, Clark PE, Bixler BR, et al. Diagnosis and management of non-metastatic upper tract urothelial carcinoma: AUA/SUO guideline. J Urol. 2023;209(6):10.
Insights into the AUA Guidelines for Upper Tract Urothelial Carcinoma: Standardizing Diagnosis and Treatment for Better Patient Outcomes - Jonathan A. Coleman


