In a podium presentation at the American Urologic Association (AUA) Virtual Annual Meeting, Dr. Fred Saad and colleagues presented an exploratory analysis of the efficacy of olaparib versus either abiraterone or enzalutamide, given a potential difference in the efficacy of NHA sequencing.
While previously published, to briefly summarize, PROfound enrolled patients with mCRPC who had alterations in ≥1 of 15 genes with a direct or indirect role in HRR and had disease progression on prior NHA. Patients were randomized 2:1 to olaparib or control of physician’s choice of abiraterone or enzalutamide. At radiographic disease progression, pts could cross over to olaparib if they met the study criteria; OS is reported without adjustment for crossover.
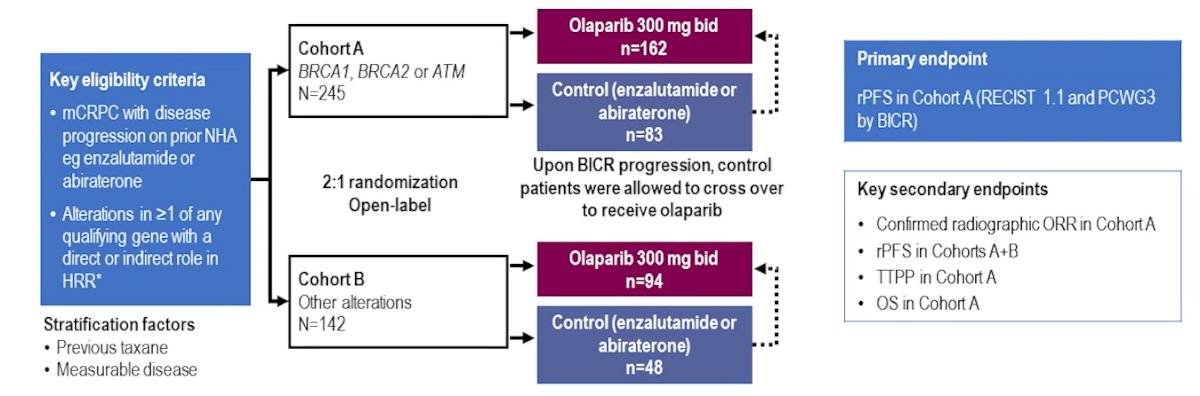
Prior to enrollment in PROfound, 40% of patients had received abiraterone only, 41% had received enzalutamide only, and 19% had received both during their treatment.
In Cohort A (defined according to mutations in BRCA1, BRCA2, or ATM), median rPFS was 7.39 months for patients in the olaparib arm versus 3.52 and 3.55 months for those receiving abiraterone or enzalutamide, respectively.
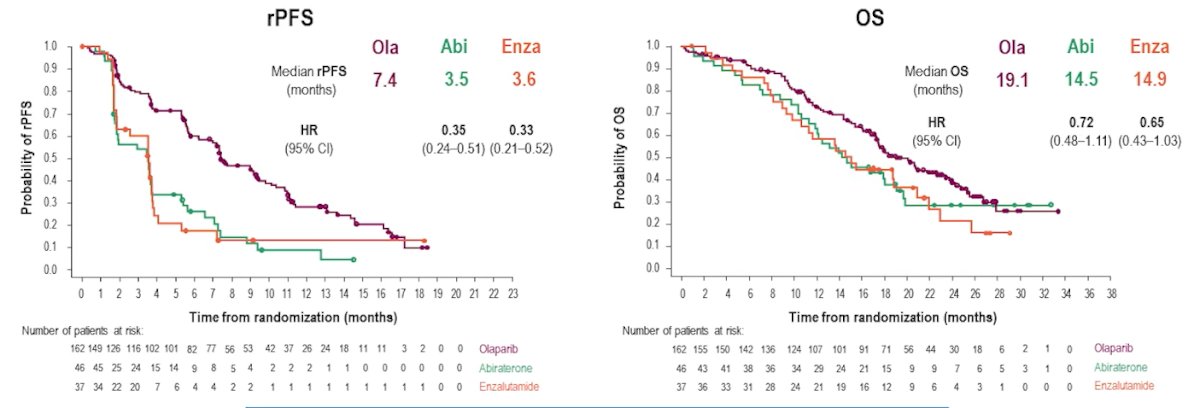
Among those patients in the control arm who received abiraterone or enzalutamide, 66% crossed over to receive olaparib at disease progression. In Cohort A, OS was 19.09 months in the olaparib arm, and 14.51 and 14.87 months for pts receiving abiraterone and enzalutamide, respectively. The efficacy of abiraterone and of enzalutamide is consistent with previous reports of the control arm in PROfound.
Dr. Saad then concluded that this exploratory analysis of the PROfound trial demonstrates improved outcomes of olaparib compared to both abiraterone and enzalutamide, without meaningful differences in outcomes in the control arm on the basis of which NHA was used. Further, he emphasized that sequential use of NHA appears to have limited benefit.
Presented by: Fred Saad, MD, FRCS, Professor and Chairman of Urology and Director of Genitourinary Oncology at the University of Montreal Hospital CenterWritten by: Christopher J.D. Wallis, University of Toronto, Twitter: @WallisCJD during the 2021 American Urological Association, (AUA) Annual Meeting, Fri, Sep 10, 2021 – Mon, Sep 13, 2021.


