(UroToday.com) The 2022 ASTRO annual meeting featured a session on bladder and kidney preservation, including a presentation by Dr. Shankar Siva discussing long-term outcomes of stereotactic ablative body radiotherapy to primary renal cell carcinoma (RCC) from the International Radiosurgery Oncology Consortium for Kidney (IROCK). Patients with RCC not suitable for surgery have few curative treatment options, particularly as thermal ablation has diminishing efficacy in RCC masses > 3 cm or those that are adjacent to the collecting system. Stereotactic ablative body radiotherapy is a novel option limited by lack of long-term outcome data. Dr. Siva and colleagues hypothesized that stereotactic ablative body radiotherapy represents an effective and safe, nephron-sparing alternative for RCC in the long-term.
Dr. Siva notes that the incidence of RCC is rising, with an estimated 21,700 cases expected by 2035 in the United Kingdom. RCC is expected to be one of the fastest increasing cancers over the next 20 years. The total number of kidney cancer cases globally is as follows:

Additionally, incidence is increasing in North America, Asia, and Europe, with the greatest incidence in those > 70 years of age. Elderly patients are at greater risk of cancer-specific mortality (up to 3.8 fold), and age, frailty, and medical comorbidities may preclude surgical or percutaneous intervention.
Individual patient data from 12 institutions from IROCK were pooled for this analysis. Patients with M1 disease and/or upper tract urothelial carcinoma were excluded, and minimum eligible follow-up was ≥ 2 years. Demographics, treatment, oncologic and renal function outcomes were assessed using descriptive statistics. Kaplan-Meier estimates and univariable and multivariable Cox proportional hazards regression were generated for oncologic outcomes. Local failure was investigator defined using RECIST 1.1. Patterns of failure were described using a cumulative incidence function with death as competing event. Toxicity was described using CTCAE v4.0.
In 190 patients, the median follow-up was 5.0 years (95% CI 4.6 – 5.2 years). The mean ± SD tumor diameter was 4.2 ± 2.2 cm and 95 patients (50%) had ≥ T1b (≥ 4 cm) primaries. Median age was 74 years (IQR: 66-82), 73.2% were male, and 87.6% had good performance status (ECOG 0-1 or KPS ≥ 70%). In patients for whom operability details were reported, 75.0% were defined as inoperable by the referring urologist, mostly for cardiovascular comorbidities (46.9%). Baseline tumor complexity was moderate (median RENAL [1] score of 7 [IQR 5-9]), and 56 patients (29.5%) had a solitary kidney. The median biologically effective dose10 delivered was 87.5 Gy (range 33.5-180.0). Mean ± SD baseline estimated glomerular filtration rate (eGFR) was 58.9 ± 22.6 mL/min (mild-to-moderate dysfunction) with 53 patients (28.0%) of the cohort having moderate-to-severe dysfunction (eGFR < 45 mL/min):
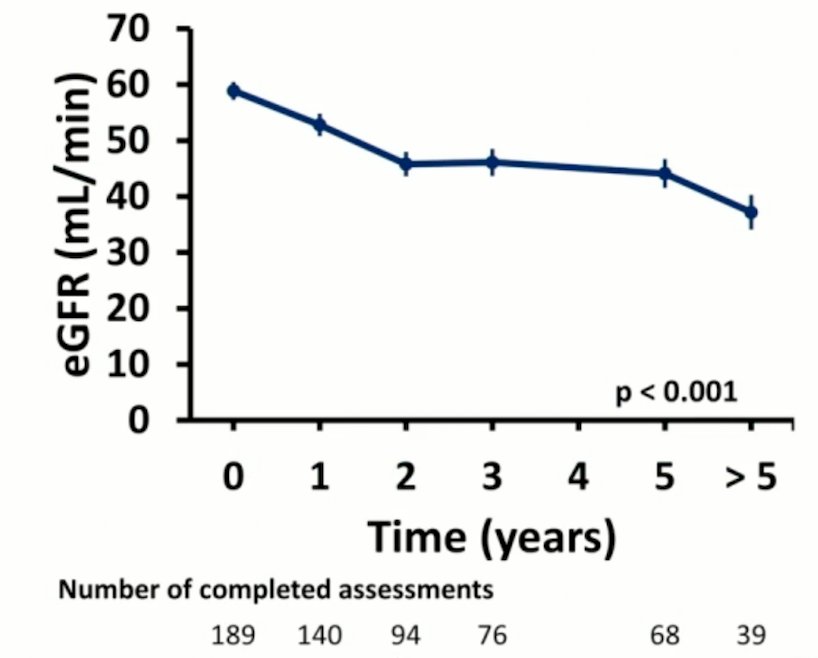
At 3- and 5-years following stereotactic ablative body radiotherapy, mean ± SD eGFR decreased by -10.8 ± 16.6 mL/min and -13.5 ± 14.9 mL/min, respectively. Seven patients (3.7%) required dialysis, with a mean +/- SD baseline eGFR of 28.1 +/- 14.9 mL/min:
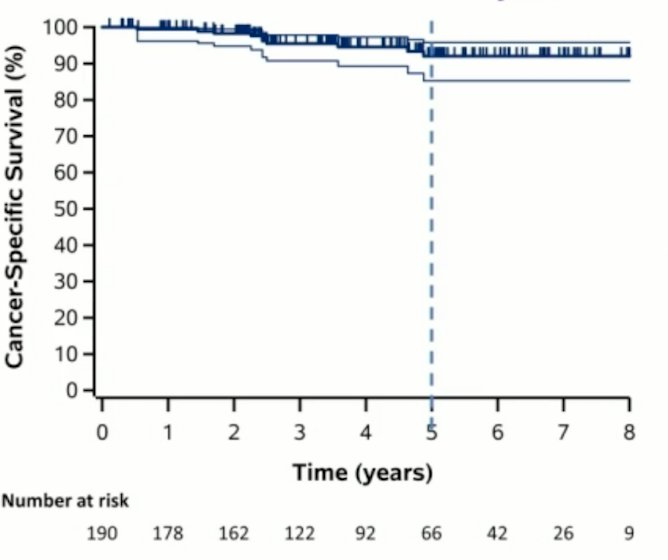
Seventy patients (36.8%) had a grade 1-2 toxicity, and one patient (0.5%) had a grade 4 gastrointestinal toxicity (at 1.4 months) and a grade 4 bowel toxicity (at 15.8 months; the patient is alive at 8.8 years without disease). Cancer-specific survival (CSS) and progression-free survival (PFS) at 3 years were 95.5% and 72.1%, and at 5 years were 92.0% and 63.6%, respectively:
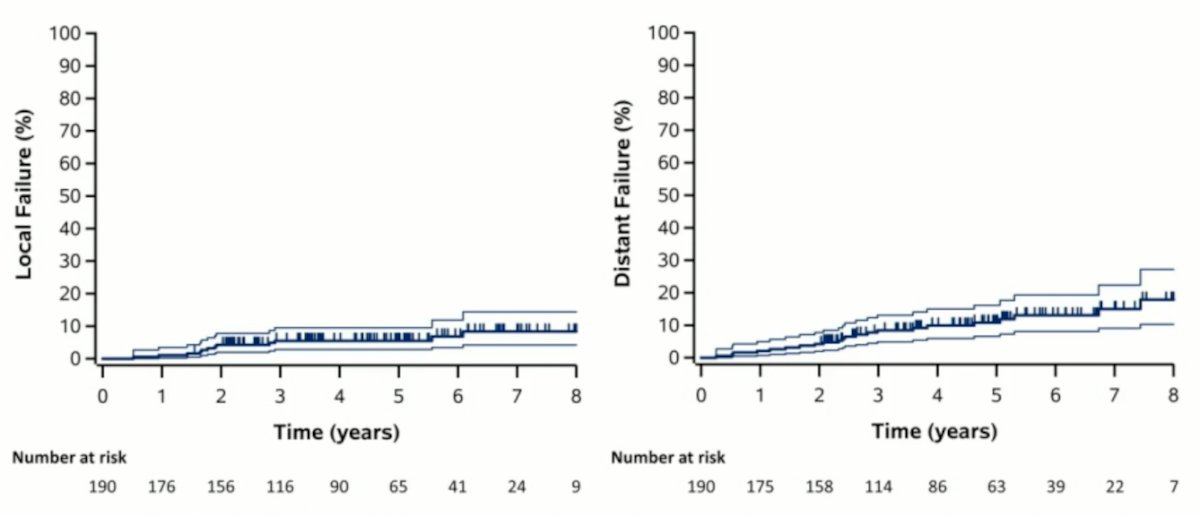
Local, distant and any failure at 5 years were 5.5%, 10.8% and 13.0%:
There were 95 patients (50%) that had < 4cm and >= 4cm at median sizes of 2.84 cm and 4.88 cm respectively. Split by T1a versus T1b+, there was no difference in CSS, PFS or local failure rate:
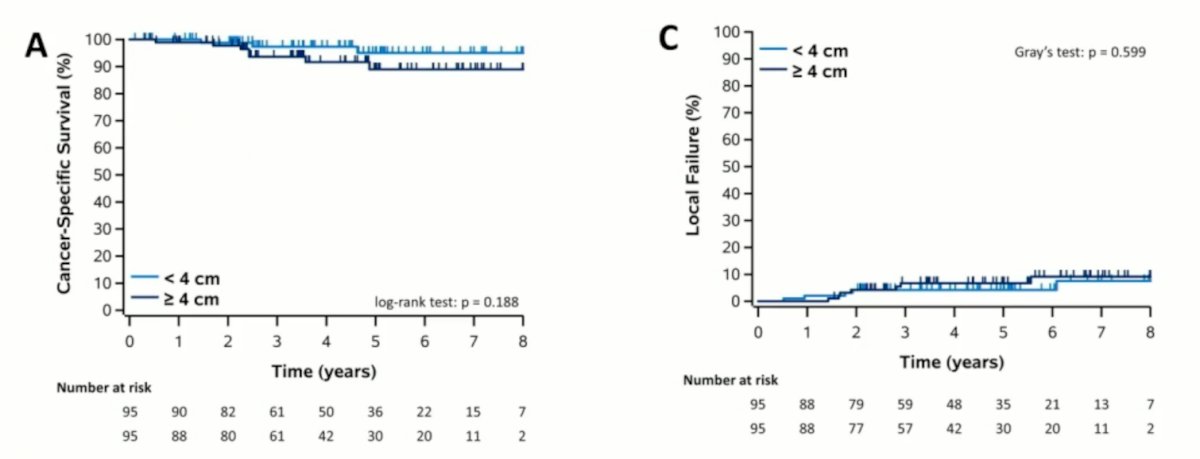
On multivariable analysis, increasing tumor size was associated with inferior CSS (HR per 1 cm increase: 1.41, 95% CI 1.15-1.71; p < 0.001), PFS (HR 1.10, 95% CI 1.01-1.19; p = 0.030), and trend to local failure (HR 1.15, 95% CI 1.10-1.32; p = 0.056).
There were 81 (42.6%) patients that received a single fraction of stereotactic body radiotherapy, most commonly as 25 Gy – 26 Gy (n = 70, 86.4%). A single fraction of stereotactic body radiotherapy resulted in decreased local failure (Gray’s p = 0.020) and decreased PFS (log-rank p = 0.004), but not CSS (p = 0.153):

Dr. Siva notes that this study observed a 5-year local failure rate of 5.5% in a large series of 190 patients with larger masses (mean 4.2 cm) than those typically treated with thermal ablation. Outcomes were comparable with other modalities for small renal masses, with 5-year control rates for partial nephrectomy, radiofrequency ablation, and cryoablation between 95-97%. In a population with baseline kidney dysfunction (average CKD class 3), there was a clinically acceptable decline at 5 years by a mean 13.5 mL/min, and only 3.7% of patients proceeded to dialysis. Dr. Siva also noted several limitations of this study including the association between single fraction stereotactic body radiotherapy and improved local control is hypothesis-generation, but not conclusive. Additional limitations include: non-uniform follow-up protocols, varied treatment techniques and dose/fractionation, and retrospective toxicity collection, which may result in under reporting of toxicity.
Dr. Siva concluded his presentation discussing long-term outcomes of stereotactic ablative body radiotherapy to primary renal cell carcinoma from IROCK with the following take home messages:
- At 5-years post-therapy, stereotactic ablative body radiotherapy for primary RCC was associated with excellent local efficacy and good oncological outcomes, with only a modest impact on renal function
- Future research includes (i) the IROCK Prospective Registry (international and launching soon), and (ii) registry-based randomized clinical trials
Presented by: Shankar Siva, Ph.D., MBBS, FRANZCR, Department of Radiation Oncology, Peter MacCallum Cancer Centre, Melbourne, VIC, Australia
Co-Authors: R. J. M. Correa2, M. Ali1, A. Muacevic3,4, L. Ponsky5, R. J. Ellis III6, S. S. Lo7, H. Onishi8, A. Swaminath9, M. McLaughlin10, S. C. Morgan11, F. Cury12, B. S. Teh13, A. Mahadevan14, I. D. Kaplan14, W. Chu15, R. Hannan16, M. Staehler17, A. Warner18, and A. V. Louie2; 1Department of Radiation Oncology, Peter MacCallum Cancer Centre, Melbourne, VIC, Australia, 2Department of Radiation Oncology, London Regional Cancer Program, London, ON, Canada, 3European Radiosurgery Center Munich, Munich, Germany, 4European Cyberknife Center Munich, Munich, Germany, 5Urology, University Hospitals Cleveland Medical Center, Cleveland, OH, 6Radiation Oncology, University Hospitals Seidman Cancer Center, Cleveland, OH, 7University of Washington School of Medicine, Seattle, WA, 8University of Yamanashi, Chuo, Japan, 9Division of Radiation Oncology, Juravinski Cancer Centre, Hamilton, ON, Canada, 10Department of Radiation Oncology, University of Texas Southwestern Medical Center, Dallas, TX, 11The Ottawa Hospital Cancer Center, Ottawa, ON, Canada, 12McGill University Health Centre, Montreal, QC, Canada, 13Department of Radiation Oncology, Houston Methodist Hospital, Houston, TX, 14Beth Israel Deaconess Medical Center, Boston, MA, 15Sunnybrook-Odette Cancer Centre, University of Toronto, Toronto, ON, Canada, 16University of Texas Southwestern Medical Center, Dallas, TX, 17University of Munich, Munich, Germany, 18London Health Sciences Centre, London, ON, Canada
Written by: Zachary Klaassen, MD, MSc – Urologic Oncologist, Assistant Professor of Urology, Georgia Cancer Center, Augusta University/Medical College of Georgia, @zklaassen_md on Twitter during the 2022 American Society of Radiation Oncology (ASTRO) Annual Hybrid Meeting, San Antonio, TX, Sat, Oct 22 – Wed, Oct 26, 2022.
References:
- Kutikov A, Uzzo RG. The RENAL nephrometry score: a comprehensive standardized system for quantitating renal tumor size, location and depth. J Urol 2009;182(3):844

