(UroToday.com) On the first day of the American Society for Clinical Oncology (ASCO) Genitourinary Cancer Symposium 2023 focussing on prostate cancer, Dr. Amarnath Challapalli presented in Poster Session A on real world outcomes of patients treated with darolutamide for non-metastatic castrate resistant prostate cancer (nmCRPC).
Following approval based on data from the ARAMIS trial, the RECORD study was launched as a real world, prospective evaluation of clinical outcomes in patients with nmCRPC treated with darolutamide. The goal of this study is to increase the understanding of treatment response and management and in particular to provide information regarding the use of next generation imaging in this setting.
This prospective study collected patient data from 9 UK centres based on the recommendation of NICE for Darolutamide as an option for the treatment of non-metastatic castrate resistant prostate cancer (nmCRPC) from November 2020 onwards. While this study is ongoing, the presented data are based on a cut-off of 15 September 2022.
To date, 87 patients have been included and are eligible for analysis. The median age of the cohort was 78 (range 61-92). The median pre-treatment PSA and PSA doubling time (PSAdT) were 13 (range 1.99-110.6) mg/L and 5.05 (range 0.6 - 10) months. 42 patients (49.4%) had pre-treatment PSAdT of <6 months and 43 (50.6%) patients had PSAdT of ≥6 months (2 patients had no pre-treatment PSAdT data). Prior to the initiation of darolutamide, 6 patients (6.90%) had undergone next generation imaging.
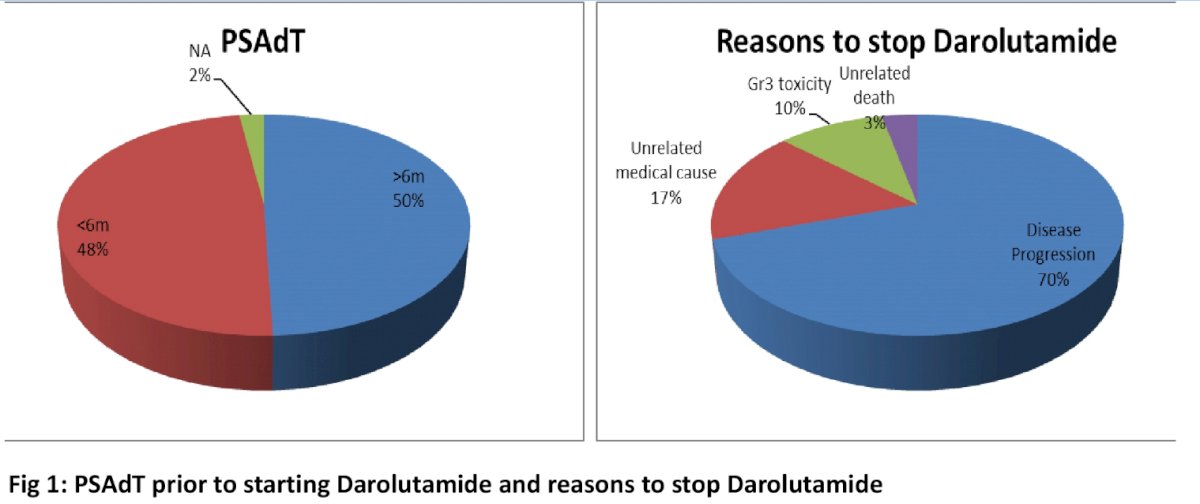
The median duration of treatment on Darolutamide was significantly shorter for patients with pre-treatment PSAdT <6 months (17 months) compared to those with pre-treatment PSAdT ≥6 (not reached; NR) (HR=0.385, 95% CI 0.17-0.88). To date, 30 patients (34.5%) have come off treatment. Of these, 21 (70%) had disease progression while 5 (16%) ceased therapy for a medical cause unrelated to the drug (e.g. COVID infection, reduced performance status secondary to pre-existing Parkinson's), 3 (10%) for unacceptable toxicity (rash, Grade3 fatigue, muscle aches, memory issues), and 1 patient died (unrelated).
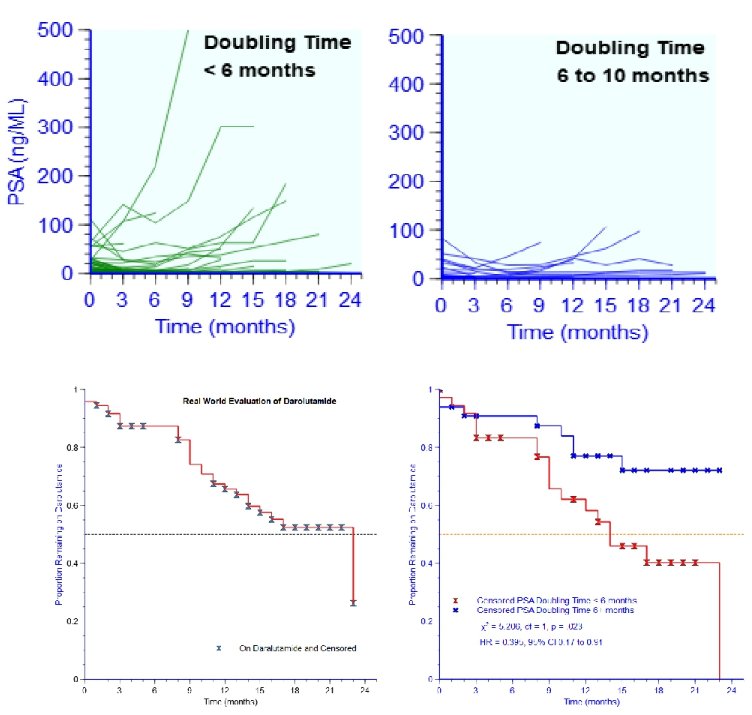
Thus, the authors conclude that these prospective real-world data from the RECORD study show that patients being managed for nmCRPC in the UK are typically staged using conventional imaging. In this population, darolutamide is well tolerated and demonstrates efficacy, in keeping with the results of the ARAMIS trial. The median duration of therapy differed significantly on the basis of PSAdT. Ongoing follow-up of this cohort will provide further data on long-term toxicity, MFS and OS.
Presented by: Amarnath Challapalli | Bristol Haematology and Oncology Centre, University Hospitals Bristol and Weston NHS Foundation Trust

